

· The San Francisco Estuary can now be recognized
as the most invaded aquatic ecosystem in North America. Now recognized
in the Estuary are 212 introduced species : 69 percent of these
are invertebrates, 15 percent are fish and other vertebrates,
12 percent are vascular plants and 4 percent are protists.
· In the period since 1850, the San Francisco
Bay and Delta region has been invaded by an average of one new
species every 36 weeks. Since 1970, the rate has been at least
one new species every 24 weeks: the first collection records of
over 50 non-native species in the Estuary since 1970 thus appear
to reflect a significant new pulse of invasions.
· In addition to the 212 recognized introductions,
123 species are considered as cryptogenic (not clearly native
or introduced), and the total number of cryptogenic taxa in the
Estuary might well be twice that. Thus simply reporting the documented
introductions and assuming that all other species in a region
are nativeóas virtually all previous studies have doneóseverely
underestimates the impact of marine and aquatic invasions on a
region's biota.
· Nonindigenous aquatic animals and plants
have had a profound impact on the ecology of this region. No shallow
water habitat now remains uninvaded by exotic species and, in
some regions, it is difficult to find any native species in abundance.
In some regions of the Bay, 100% of the common species are introduced,
creating "introduced communities." In locations ranging
from freshwater sites in the Delta, through Suisun and San Pablo
Bays and the shallower parts of the Central Bay to the South Bay,
introduced species account for the majority of the species diversity.
· The major bloom-creating, dominant phytoplankton
species are cryptogenic. Because of the poor state of taxonomic
and biogeographic knowledge, it remains possible that many of
the Estuary's major primary producers that provide the phytoplankton-derived
energy for zooplankton and filter feeders, are in fact introduced.
· Introduced species are abundant and dominant
throughout the benthic and fouling communities of San Francisco
Bay. These include 10 species of introduced bivalves, most of
which are abundant to extremely abundant. Introduced filter-feeding
polychaete worms and crustaceans may occur by the thousands per
square meter. On sublittoral hard substrates, the Mediterranean
mussel Mytilus galloprovincialis is abundant,
while float fouling communities support large populations of introduced
filter feeders, including bryozoans, sponges and seasquirts. The
holistic role of the entire nonindigenous filter-feeding guildóincluding
clams, mussels, bryozoans, barnacles, seasquirts, spionid worms,
serpulid worms, sponges, hydroids, and sea anemonesóin
altering and controlling the trophic dynamics of the Bay-Delta
system remains unknown. The potential role of just one species,
the Atlantic ribbed marsh mussel Arcuatula demissa, as
a biogeochemical agent in the economy of Bay salt marshes is striking.
· Introduced clams are capable of filtering
the entire volume of the South Bay and the northern estuarine
regions (Suisun Bay) once a day: indeed, it now appears that the
primary mechanism controlling phytoplankton biomass during summer
and fall in South San Francisco Bay is "grazing" (filter
feeding) by the introduced Japanese clams Venerupis
and Musculista and the Atlantic clam Gemma. This
remarkable process has a significant impact on the standing phytoplankton
stock in the South Bay, and since this plankton is now utilized
almost entirely by introduced filter feeders, passing the energy
through a non-native benthic fraction of the biota may have fundamentally
altered the energy available for native biota
· Drought year control of phytoplankton by
introduced clamsóresulting in the failure of the summer
diatom bloom to appear in the northern reach of the Estuaryóis
a remarkable phenomenon. The introduced Atlantic soft-shell clams
(Mya) alone were estimated to be capable at times
of filtering all of the phytoplankton from the water column on
the order of once per day. Phytoplankton blooms occurred only
during higher flow years, when the populations of Mya and
other introduced benthic filter feeders retreated downstream to
saltier parts of the Estuary.
· Phytoplankton populations in the northern reaches of the Estuary may now be continuously and permanently controlled by introduced clams. Arriving by ballast water and first collected in the Estuary in 1986, by 1988 the Asian clam Potamocorbula reached and has since sustained average densities exceeding 2,000/m2. Since the appearance of Potamocorbula, the summer diatom bloom has disappeared, presumably because of increased filter feeding by this new invasion. The Potamocorbula population in the northern reaches of the Estuary can filter the entire water column over the channels more than once per day and over the shallows almost 13 times per day, a rate of filtration which exceeds the phytoplankton's specific growth rate and approaches or exceeds the bacterioplankton's specific growth rate.
· Further, the Asian clam Potamocorbula
feeds at multiple levels in the food chain, consuming bacterioplankton,
phytoplankton, and zooplankton (copepods), and so may substantially
reduce copepod populations both by depletion of the copepods'
phytoplankton food source and by direct predation. In turn, under
such conditions, the copepod-eating native opossum shrimp Neomysis
may suffer a near-complete collapse in the northern reach.
It was during one such pattern that mysid-eating juvenile striped
bass suffered their lowest recorded abundance. This example and
the linkages between introduced and native species may provide
a direct and remarkable example of the potential impact of an
introduced species on the Estuary's food webs.
· As with the guild of filter feeders, the
overall picture of the impact of introduced surface-dwelling and
shallow-burrowing grazers and deposit feeders in the Estuary is
incompletely known. The Atlantic mudsnail Ilyanassa
is likely playing a significantóif not the most importantórole
in altering the diversity, abundance, size distribution, and recruitment
of many species on the intertidal mudflats of San Francisco Bay.
· The arrival and establishment in 1989-90
of the Atlantic green crab Carcinus maenas in
San Francisco Bay signals a new level of trophic change and alteration.
The green crab is a food and habitat generalist, capable of eating
an extraordinarily wide variety of animals and plants, and capable
of inhabiting marshes, rocky substrates, and fouling communities.
European, South African, and recent Californian studies indicate
a broad and striking potential for this crab to significantly
alter the distribution, density, and abundance of prey species,
and thus to profoundly alter community structure in the Bay.
· Nearly 30 species of introduced marine,
brackish and freshwater fish are now important carnivores throughout
the Bay and Delta. Eastern and central American fish -- carp,
mosquitofish, catfish, green sunfish, bluegills, inland silverside,
largemouth and smallmouth bass, and striped bass -- are among
the most significant predators, competitors, and habitat disturbers
throughout the brackish and freshwater reaches of the Delta, with
often concomitant impacts on native fish communities. The introduced
crayfish Procambarus and Pacifastacus
may play an important role, when dense, in regulating their prey
plant and animal populations.
· Native waterfowl in the Estuary consume
some introduced aquatic plants (such as brass buttons) and native
shorebirds feed extensively on introduced benthic invertebrates.
· The Atlantic salt-marsh cordgrass Spartina
alterniflora, which has converted 100s of acres of
mudflats in Willapa Bay, Washington, into grass islands, has become
locally abundant in San Francisco Bay, and is competing with the
native cordgrass. Spartina alterniflora has broad potential
for ecosystem alteration. Its larger and more rigid stems, greater
stem density, and higher root densities may decrease habitat for
native wetland animals and infauna. Dense stands of S. alterniflora
may cause changes in sediment dynamics, decreases in benthic algal
production because of lower light levels below the cordgrass canopy,
and loss of shorebird feeding habitat through colonization of
mudflats.
· The Australian-New Zealand boring isopod Sphaeroma quoyanum creates characteristic "Sphaeroma topography" on many Bay shores, with many linear meters of fringing mud banks riddled with its half-centimeter diameter holes. This isopod may arguably play a major, if not the chief, role in erosion of intertidal soft rock terraces along the shore of San Pablo Bay, due to their boring activity that weakens the rock and facilitates its removal by wave action. Sphaeroma has been burrowing into Bay shores for over a century, and it thus may be that in certain regions the land/water margin has retreated by a distance of at least several meters due to this isopod's boring activities.
· Introduced freshwater and anadromous fish
have been directly implicated in the regional reduction and extinction,
and the global extinction, of four native California fish. The
bluegill, green sunfish, largemouth bass, striped bass, and black
bass, through predation and through competition for food and breeding
sites, have all been associated with the regional elimination
of the native Sacramento perch from the Delta. The introduced
inland silversides may be a significant predator on the larvae
and eggs of the native Delta smelt. Expansion of the introduced
smallmouth bass has been associated with the decline in the native
hardhead. Predation by largemouth bass, smallmouth black bass
and striped bass may have been a major factor in the global extinction
of the thicktail chub in California.
· The situation of the California clapper
rail may serve as a model to assess how an endangered species
may be affected by biological invasions. The rail suffers predation
by introduced Norway rats and red fox; it may both feed on and
be killed by introduced mussels; and it may find refuge in introduced
cordgrass, although this same cordgrass may compete with native
cordgrass, perhaps preferred by the rail. Other potential model
study systems include introduced crayfish and their displacement
of native crayfish; introduced gobies and their relationship to
the tidewater goby; and the combined role that introduced green
sunfish, bluegill, largemouth bass, and American bullfrog may
have played in the dramatic decline of native red-legged and yellow-legged
frogs.
· Although some of the fish intentionally
introduced into the Estuary by government agencies supported substantial
commercial food fisheries, these fisheries all declined after
a time and are now closed. The signal crayfish, Pacifastacus,
from Oregon, whose exact means of introduction is unclear, supports
the Estuary's only remaining commercial food fishery based on
an introduced species.
· The striped bass sport fishery has resulted
in a substantial transfer of funds from anglers to those who supply
anglers' needs, variously estimated, between 1962 and 1992, between
$7 million and $45 million per year. However, striped bass populations
and the striped bass sport fishery have declined dramatically
in recent years.
· Government introductions of organisms for
sport fishing, as forage fish and for biocontrol have frequently
not produced the intended benefits, and have sometimes had harmful
"side effects," such as reducing the populations of
economically important species.
· Few nonindigenous organisms that were introduced
to the Estuary by other than government intent have produced economic
benefits. The clams Mya and Venerupis,
both accidentally introduced with oysters, have supported commercial
harvesting in the Bay or elsewhere on the Pacific coast, and a
small amount of recreational harvesting in the Bay (though these
clams may have, to some extent, replaced edible native clams);
the Asian clam Corbicula is commercially harvested for
food and bait in California on a small scale; the Asian yellowfin
goby is commercially harvested for bait; muskrat are trapped for
furs; and the South African marsh plant brass buttons provides
food for waterfowl. There do not appear to be any other significant
economic benefits that derive from nongovernmental or accidental
introductions to the Estuary.
· A single introduced organism, the shipworm
Teredo navalis, caused $615 million (in 1992
dollars) of structural damage to maritime facilities in 3 years
in the early part of the 20th century.
· The economic impacts of hull fouling and
other ship fouling are clearly very large, but are not documented
or quantified for the Estuary. Most of the fouling incurred in
the Estuary is due to nonindigenous species. Indirect impacts
due to the use of toxic anti-fouling coatings may also be substantial.
· Waterway fouling by introduced water hyacinth
has become a problem in the Delta over the last fifteen years,
with other introduced plants beginning to add to the problem in
recent years. Hyacinth fouling has had significant economic impacts,
including interference with navigation.
· Perhaps the greatest economic impacts may
derive from the destabilizing of the Estuary's biota due to the
introduction and establishment of an average of one new species
every 24 weeks. This phenomenal rate of species additions has
contributed to the failure of water users and regulatory agencies
to manage the Estuary so as to sustain healthy populations of
anadromous and native fish, resulting in increasing limitations
and threats of limitations on water diversions, wastewater discharges,
channel dredging, levee maintenance, construction and other economic
activities in and near the Estuary, with implications for the
whole of California's economy.
Much remains unknown in terms of the phenomena,
patterns, and processes of invasions in the Bay and Delta, and
thus large gaps remain in the knowledge needed to establish effective
management plans. The following are examples of important research
needs and directions:
Only a few of the hundreds of invaders in the
Estuary have been the subject of quantitative experimental studies
elucidating their roles in the Estuary's ecosystem and their impacts
on native biota. Such studies should receive the highest priority.
Urgently required is a San Francisco Bay Shipping
Study which both updates the 1991 data base available and expands
that data base to all Bay and Delta ports. A biological and ecological
study of the nature of ballast water biota arriving in the Bay/Delta
system is urgently required. Equally pressing is a study of the
fouling organisms entering the Estuary on ships' hulls and in
ships' seachests, in order to assess whether this mechanism is
now becoming of increasing importance and in order to more adequately
define the unique role of ballast water. A Regional Shipping Study
would provide critical data for management plans.
Studies are required on the mechanisms and the
temporal and spatial scales of the distribution of introduced
species by human vectors after they have become established. Such
studies will be of particular value in light of any future introductions
of nuisance aquatic pests.
This study has identified a major, unregulated
vector for exotic species invasions in the Bay: the constant release
of invertebrate-laden seaweeds from New England in association
with bait worm (and lobster) importation. In addition a new trade
in exotic bait has commenced, centered around the importation
of living Vietnamese nereid worms, and both the worms and their
substrate deserve detailed study. These studies are urgently needed
to address the attendant precautionary management issues at hand.
The application of modern molecular genetic techniques
has already revealed the cryptic presence of previously unrecognized
invaders in the Bay: the Atlantic clam Macoma petalum,
the Mediterranean mussel Mytilus galloprovincialis, and
the Japanese jellyfish Aurelia "aurita." Molecular
genetic studies of the Bay's new green crab (Carcinus)
population may be of critical value in resolving the crab's geographic
origins and thus the mechanism that brought it to California.
Molecular genetic studies of worms of the genus Glycera
and Nereis in the Bay may clarify if New England populations
have or are becoming established in the region as a result of
ongoing inoculations via the bait worm industry. Molecular analysis
of other invasions will doubtless reveal, as with Macoma
and Mytilus, a number of heretofore unrecognized species.
Fishery, bait, and other utilization studies should
be conducted on developing or enlarging the scope of fisheries
for introduced bivalves (such as Mya, Venerupis,
and Corbicula), edible aquatic plants, smaller edible fish
(such as Acanthogobius), and crabs (Carcinus and
Eriocheir).
Studies are needed on the potential distribution,
abundance and impacts of zebra mussels (Dreissena polymorpha
and/or D. bugensis) in California, to support efforts to
control their introduction and to design facilities (such as water
intakes and fish screens) that will continue to function adequately
should the mussels become established.
The economic impacts of wood-boring organisms
(shipworms and gribbles) and of fouling organisms (on commercial
vessels, on recreational craft, in ports and marinas, and in water
conduits) are clearly very large in the San Francisco Estuary,
but remain largely undocumented and entirely unquantified. A modern
economic study of this phenomenon, including the economic costs
and ecological impacts of control measures now in place or forecast,
is critically needed.
Largely qualitative data suggest that the economic,
ecological, and geological impacts of the guild of burrowing organisms
that have been historically and newly introduced have been or
are forecast to potentially be extensive in the Estuary. Experimental,
quantitative studies on the impacts of burrowing and bioeroding
crustaceans and muskrats in the Estuary are clearly now needed
to assess the extent of changes that have occurred or are now
occurring, and to form the basis for predicting future alterations
in the absence of control measures.
While primary attention must be paid to preventing
future invasions, studies should begin on examining the broad
suite of potential post-invasion control mechanisms, including
biocontrol, physical containment, eradication, and related strategies.
A Regional Control Mechanisms Workshop for past and anticipated
invasions could set the foundation for future research directions.
1. Introduction 1
2. Methods 4
3. Introduced Species in the Estuary 10
4. Cryptogenic Species in the Estuary 149
5. Results 154
By Taxonomic Group 154
By Native Region 155
By Time Period 157
By Transport Mechanism 160
6. Discussion 167
Ecological Impacts 167
Economic Impacts 190
Future Invasions 202
7. Conclusions 210
Major Findings 210
Research Needs 215
References 218
Appendix 1A. Introduced Terrestrial Plants, Birds and Mammals Reported from the San Francisco Estuary
Appendix 1B. Descriptions of Introduced Terrestrial Plants Reported from the San Francisco Estuary
Appendix 1C. Descriptions of Introduced Terrestrial Mammals Reported from the San Francisco Estuary
Appendix 2. Earlier Inoculations into the San Francisco Estuary and Nearby Waters
Appendix 3. Descriptions of Introduced Plants and Invertebrates in Areas Adjacent to the San Francisco Estuary
Appendix 4. Introduced Organisms in the Northeastern Pacific Known only from the San Francisco Estuary or its Watershed
Appendix 5. Introduced Marine, Estuarine and Aquatic Organisms in Four Regional Studies
Table 1. Introduced organisms in the San Francisco Estuary
141
Table 2. Cryptogenic species in the San Francisco Estuary 150
Table 3. Treatment of introduced species as marine or continental,
for analysis by native region 156
Table 4. Associations of introduced species in the San Francisco
Estuary 170
Table 5. Patterns of invasion along the salinity gradient in the
San Francisco Estuary and the adjoining coast 180
Table 6. Positive economic impacts of marine, estuarine and aquatic
organisms introduced into the San Francisco Estuary 191
Table 7. Negative economic impacts of introduced marine, estuarine
and aquatic organisms 196
Table 8. Recent records of nonindigenous species in the San Francisco
Estuary whose establishment is uncertain 203
Table 9. Introduced species in adjacent areas with the potential
to invade the San Francisco Estuary 205
Table 10. Examples of ongoing inoculations of nonindigenous species
into the San Francisco Estuary 207
Figure 1. The San Francisco Estuary 5
Figure 2. Invasions by taxonomic group: lower-level aggregation
154
Figure 3. Invasions by taxonomic group: higher-level aggregation
155
Figure 4. Invasions by native region 157
Figure 5. Invasions into the San Francisco Estuary by period
159
Figure 6. Invasions into the Northeastern Pacific by period
159
Figure 7. Invasions by type of transport mechanism 161
Figure 8. Some examples of damage caused by the wood-boring shipworm
Teredo navalis in the San Francisco Estuary 194
Scores of individuals, scientists, agency representatives and
members of the public assisted us with the compilation of the
species records in this report. We gratefully acknowledge these
workers for their contributions in the appropriate portion of
the text. Members of the First (October 1993) and Second (July
1994) San Francisco Bay Expeditions (John Chapman, Jean Chapman,
Sarah Cohen, Terry Gosliner, Claudia Mills, Luis Solarzano and
John Rees) were of inestimable help in our field and subsequent
systematic work. John Chapman spent many hours working over recent
collections of San Francisco Bay peracarid crustaceans to resolve
the status of numerous amphipods and isopods. Gretchen Lambert
identified several sets of ascidians from the Bay, and William
Banta and Marianne DiMarco-Temkin aided with the identification
of bryozoans. Gary Gillingham (Kinnetic Laboratories, Inc., Santa
Cruz), Mike Kellogg (City and County of San Francisco), Heather
Peterson (California Department of Water Resources) and Jan Thompson
(U. S. Geological Survey) provided extensive species list from
benthic surveys under their respective aegises. James Orsi (California
Department of Fish and Game) provided assistance in our research
on zooplankton and Doris Sloan (University of California) on foraminifera.
Over the past four centuries thousands of species of fresh water, brackish water and salt water animals and plants have been introduced to the United States (Elton, 1958; Carlton, 1979a, 1989, 1992b; Moyle, 1986; Hickman, 1993; Carlton & Geller, 1993). In some regions, such as the Hawaiian Islands, aboriginal introductions date back more than two millennia (Mooney & Drake, 1986). The taxonomic, habitat and trophic range of this vast nonindigenous biota is impressiveóranging from exotic flatworms (Rectocephala exotica) in the lily ponds of Washington, D. C., to Mexican crabs (Platychirograpsus spectabilis ) in Florida rivers, to aquatic rodents such as the South American nutria (Myocaster coypu) in the southern United States.
The human role in changing the face of North America, in terms of the abundance and diversity of the animals and plants of lakes, rivers, estuaries, marshes, and coastlines, has been demonstratively profound:
· Sea lampreys (Petromyzon marinus) invaded the Great Lakes, destroying extensive native fisheries; the Eurasian carp (Cyprinus carpio), released in New York in 1831, is now a national pest; Nevada's Ash Meadows killifish (Empetrichthys merriami) became extinct at the hands of introduced mosquitofish, mollies, crayfish, and bullfrogs; and scores of exotic fish species now dominate aquatic habitats from Florida to New York and from the Atlantic drainage to California.
· Asian clams (Corbicula fluminea) spread across all of North America in only 40 years, moving from west to eastófrom the Columbia River to California and then quickly across the southern United States to the Atlantic seaboard, a dramatic and startling invasion of this canal- and pipe-fouling clam (McMahon, 1982). Fifty years later, European zebra mussels (Dreissena polymorpha and Dreissena bugensis) are similarly spreading across North Americaóthis time from east to west, from the Great Lakes to the Mississippi and into Oklahoma.
· Alien plantsóincluding the spectacularly successful purple loosestrife (Lythrum salicaria), Eurasian watermilfoil (Myriophyllum spicatum) and water chestnut (Trapa natans)óare now the dominant, and at times the only, vegetation, for hundreds of square miles of aquatic and marsh habitats in North America.
Despite these many invasions, there are with rare exception no syntheses of the spatial and temporal patterns, mechanisms or impacts of these nonindigenous aquatic and estuarine organisms. For the great majority of invasions, records are scattered among thousands of scientific papers and buried in general monographs, student theses, government reports, consultant studies and anecdotal accounts. While a comprehensive review of freshwater and marine invasions would be extraordinarily useful, an initial approach to understanding the ecological and economic impacts of nonindigenous animals and plants in U. S. aquatic and marine environments may be attained through case studies: the assessment of the role of invasions in defined geographic regions, focusing on historical and modern-day dispersal pathways, on the biological, ecological and economic consequences of invasions, and on prospects for future invasions.
We present here such a regional study, focusing on one of the
largest freshwater and estuarine ecosystems of the United States:
the San Francisco Bay and Delta region, a region known to have
sustained numerous invasions for over a century.
At the time of our study there was no synthesis available of the diversity and impacts of the nonindigenous aquatic and estuarine species of the San Francisco Bay and Delta region, an area that extends from the inland port cities of the Central Valley to the coastal waters of the Pacific Ocean at the Golden Gate.
This region includes examples of most of the common aquatic habitats found throughout the warm and cool temperate climates of the United States and, as such, represents an ideal theater for assessing the diversity and range of effects of aquatic invasions. Within the Bay-Delta Region are fresh, brackish, and salt water marshes, sandflats and mudflats, rocky shores, benthic sublittoral habitats of a wide sediment range, eelgrass beds, emergent aquatic macrophyte communities, planktonic, nektonic, and neustonic communities, extensive fouling assemblages, and communities of burrowing and boring organisms in clays and wood. Also represented is a vast range of habitat disturbance regimes. Over a 140-year period of substantial human commercial and other activitiesósince about 1850óa minimum of more than 200 plants, protists and animals from the aquatic and coastal habitats of eastern North America, Europe, Asia, Australia, and South America have invaded these ecosystems.
Prior lists or descriptions of the introduced freshwater, anadromous and estuarine fish fauna in the San Francisco Bay-Delta region were provided by Moyle (1976b) and McGinnis (1984); of freshwater mollusks by Hanna (1966) and Taylor (1981); of marine mollusks by Nichols et al. (1986); and of introduced marine and estuarine invertebrates by Carlton (1975, 1979a,b), supplemented by Carlton et al. (1990). Silva (1979) and Josselyn & West (1985) noted some introductions of marine and brackish seaweeds, but no comprehensive assessment of possibly introduced seaweeds had been made. Atwater et al. (1979) provided a list of introduced vascular plants in San Francisco Bay salt marshes, but appear not to have distinguished between aquatic plants that are characteristically found within marshes and essentially terrestrial plants that are occasionally found at the edges of or within marshes. During our study the Bay-Delta Oversight Committee of the California Department of Water Resources produced a briefing paper summarizing some of the previously published information on introduced fish, wildlife and plants of the Bay-Delta region (BDOC, 1994), and Orsi (1995) published a list of introduced estuarine copepods and mysids.
No information had been compiled on possible introductions among freshwater invertebrates (including species of freshwater sponges, jellyfish, flatworms, oligochaete and polychaete worms, snails, clams, crustaceans, insects and bryozoans), freshwater macroalgae, or fresh, brackish or salt water phytoplankton. Protozoan introductions had been similarly neglected.
Based on the information available prior to our study, and on
consideration of extant lists of aquatic or marine introductions
in other regions (Leppäkoski, 1984; den Hartog, 1987; Mills
et al., 1993, 1995; Jansson, 1994), we had estimated that the
number of aquatic and estuarine introductions in the Bay-Delta
system could exceed 150 invertebrate species, 20 fish species,
10 algal species, and 100 vascular plant species.
The present work is the first regional case study in the United States of the diversity and ecological and economic impacts of nonindigenous species in aquatic and estuarine habitats. Previous studies (Mills et al., 1993, for the Great Lakes; Mills et al., 1996, for the Hudson River) have largely concentrated on species check-lists with a minimal review of ecological or economic effects of the exotic biota. We intend the present study to be a comprehensive synthesis which may serve as a comparative model for other regional studies in U. S. waters.
The present study also sets forth detailed and clear criteria for determining which species are present and established within the study zone. Prior regional surveys of aquatic introductions have implied but rarely defined these criteria, a situation that impedes ready quantitative comparisons between regions. We include (Chapter 5) a supplemental list of vascular plant species based upon criteria which we judge to approximate the criteria in prior regional surveys of aquatic introductions in the USA, in order to facilitate such comparisons.
The present study is also the first regional survey of introductions to include a listing (although preliminary) of cryptogenic speciesóspecies which are neither demonstrably native or introduced (Chapter 4). As discussed by Carlton (1996a), the development of such lists is a necessary first step in correcting prior tendencies to profoundly underestimate the potential extent of biological invasions and in providing a more complete basis for understanding the sources, characteristics and frequency of success of biological invaders.
Both older (Elton, 1958) and newer (e. g. Mooney & Drake, 1986; Drake et al., 1989) reviews of biological invasions propose a number of theoretical models to explain the success of animal and plant invasions in regions where they did not evolve. However, for most such studies, comprehensive data sets on the diversity of invasions, temporal patterns of invasion, and ecological impacts have not been available by which to test the applicability or robustness of invasion theory. The present study provides an extensive review of an introduced biota exceeding 200 taxa in a defined geographic region, and thus provides a rare data set with which to test invasion models.
The study zone for this report is defined as the estuarine and
aquatic habitats that are within the normal range of tidal influence
in San Francisco Bay, the Sacramento-San Joaquin Delta and tributaries,
and referred to herein as the San Francisco Estuary or the Estuary
(Fig. 1). The primary data set (Chapter 3 and Table 1) contains
all demonstrably nonindigenous organisms that are characteristically
found in estuarine or aquatic habitats (including marshes, mudflats,
etc.), and for which there is significant evidence supporting
their establishment within the study zone.
Inclusion in the primary data set thus requires evidence demonstrating that the organism in question is (1) not native to the Estuary, and (2) currently established in the Estuary.
We define native organisms as those organisms present aboriginally, which for the Bay-Delta region means prior to 1769 when the first European explorers entered the area. The types of evidence that we utilized to determine the native versus introduced status of aquatic and estuarine organisms, as discussed by Carlton (1979a) and Chapman & Carlton (1991, 1994), include:
· global systematic evidence (involving taxonomic information from both morphology and molecular genetics) and biogeographic evidence, including the global distribution of closely related species;
· the existence of identifiable mechanisms of human-mediated transport;
· historical evidence of presence or absence;
· archaeological evidence of presence or absence;
· paleontological evidence of presence or absence;
· the extent to which distribution can be explained by natural dispersal mechanisms;
· rapid or sudden changes in abundance or distribution;
· highly restricted or anomalously disjunct distributions (in comparison to distributions of known native organisms);
· occurrence in assemblages with other known introduced species; and
· for parasites or commensals, occurrence on introduced organisms.
We define established organisms as those organisms present and
reproducing "in the wild" whose numbers, distribution
and persistence over time suggest that, barring unforeseen catastrophic
events or successful eradication efforts, they will continue to
be present in the future. "In the wild" implies reproduction
and persistence of the population without direct human intervention
or assistance (such
as reproductive assistance via hatcheries or periodic renewal of the population through the importation of spat), but may include dependence on human-altered or created habitats, such as water bodies warmed by the cooling-water effluent from power plants, pilings, floating docks, and salt ponds or other manipulated, semi-enclosed lagoons. The types of evidence that we used to assess establishment include:
· population size;
· persistence of the population over time;
· distribution (broad or restricted) of the population, and trends in distribution;
· for species dependent on sexual reproduction, the presence of both males and females, and the presence of ovigerous females; and
· the age structure of the population as an indicator of
successful reproduction.
Beyond the primary data set, we considered and compiled information on several additional categories of organisms, including:
· cryptogenic organisms, that is, organisms in the Estuary that are neither demonstrably native nor introduced (Table 2);
· nonindigenous organisms that have been reported from or were intentionally introduced to the Estuary, but which did not become established or for which there is inadequate evidence regarding their establishment (Table 8 and Appendix 2);
· nonindigenous organisms which are established in aquatic environments tributary to or adjacent to the Estuary, and which may in the future extend their range into the Estuary (Table 9);
· nonindigenous organisms which are not characteristically
found in estuarine or aquatic habitats but which have been occasionally
reported from or may make occasional use of the Estuary (Appendix
1).
Probably the largest and most difficult "gray zone" between the primary data set and organisms in these additional categories involves those nonindigenous plants reported from coastal or freshwater wetlands for which specific information on occurrence within the tidal boundaries of the Estuary is not available. Although previous regional studies of aquatic invasions (Mills et al., 1993, 1995) have included many such gray-zone plants, we limited inclusion in our primary data set to those that both: (a) have habitat descriptions indicating that they are primarily marsh plants, and not primarily terrestrial or moist ground plants occasionally found in or near marshes; and (b) have been reported specifically from the Delta, and not just from the Central Valley or the Bay Area generally. Similar questions arose, though less commonly, with other types of organisms, to which we applied similar logic.
Those candidate organisms which are not listed in Table 1 because
of criterion (a), are instead listed in Appendix 1. Adding the
plants in Appendix 1 to the organisms in Table 1 would produce
a list of nonindigenous organisms for the Estuary comparable those
produced for the Great Lakes (Mills et al., 1993) and the Hudson
River (Mills et al., 1995), as discussed further in Chapter 5.
Candidate organisms which failed to meet criterion (b) are listed
in Table 9. Even following these restrictive criteria, we may
have included in Table 1 some plants that are found in the Delta
region in marshes or diked ponds, but not in tidal waters.
Initial lists of taxa in the above-described categories were compiled from the prior studies discussed in the introduction and from a review of the regional biological and systematic literature including regional monographic studies, keys, field guides and checklists; from published (mainly in the gray literature) and unpublished species lists generated by public agencies and private consultants; and from discussions with taxonomists, field biologists, refuge managers and consultants familiar with the region.
Further information on the species thus identified was developed through a review of the pertinent current and historical biological literature, museum records and specimen collections, and interviews with biologists. We also undertook limited field work in order to check the presence or distribution of certain species, and to check for the presence of previously unreported species in some rarely sampled habitats. This information was used to develop the following species lists:
· Table 1, listing introduced species in the Estuary;
· Table 2, listing cryptogenic species in the Estuary;
· Table 8, listing species recently recorded from the Estuary but whose establishment is uncertain;
· Table 9 and Appendix 3, listing introduced species in adjacent aquatic habitats;
· Appendix 1, listing terrestrial species that may occasionally be found in the Estuary;
· Appendix 2, listing older inoculations of nonindigenous species that did not become established; and
· Appendix 4, listing introduced species in the northeastern
Pacific known only from the Estuary.
For each species listed in Table 1 we determined where possible:
· the date of first collection or observation or planting in the Estuary, in California and in northeastern Pacific waters or coastal states or provinces; and where this was unavailable, the date of the first written account of the organism in the area;
· the native range of the species;
· the immediate geographic source of the introduction;
· the transport mechanism;
· the organism's current taxonomic status, most frequently utilized synonyms, and common names; and
· its current spatial distribution and abundance in the Estuary.
We included common names from Turgeon et al. (1988) and Carlton (1992) for mollusks, Cairns et al. (1991) for coelenterates, Williams et al. (1989) for decapods, Gosner (1978) for other invertebrates, Robins et al. (1991) for fish and Hickman (1983) for higher plants.
The data are presented in the species descriptions in Chapter
3 and summarized (in large part) in Table 1. Some of these data
are also provided for the species listed in Tables 8 and 9 and
the appendices. We also reviewed the available information on
the ecological roles and economic impacts of individual introduced
species and of introduced species assemblages. This information
is summarized in the species descriptions in Chapter 3 and discussed
in Chapter 6.
The primary data set in Chapter 3 and Table 1 was quantitatively
analyzed with regard to taxonomic groups, native regions, timing
and transport mechanisms. The results are presented in Chapter
5.
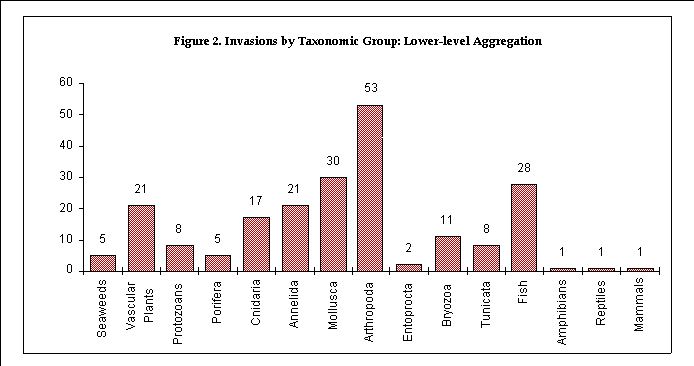
The numbers of species per taxonomic group were tabulated at two
levels of aggregation. A first tabulation was done at the taxonomic
levels of order (for vertebrates), phylum (for invertebrates),
subkingdom (for plants) and kingdom (for protozoans). A second,
more highly-aggregated, tabulation was done at the levels of class
(vertebrates), a traditional, non-phyletic grouping (invertebrates),
and kingdom (plants and protozoans).
The numbers of species per native region were tabulated with regard to eleven marine regions and five continental regions. The marine regions consist of the eastern and western portions of the North and South Atlantic oceans and the North and South Pacific oceans, the Indian Ocean, the Mediterranean Sea, and the Black and Caspian Seas. The Western South Pacific region consists primarily of waters around Australia and New Zealand. The five continental regions consist of North America, South America, Eurasia, Africa, and Australia/New Zealand. Where an organism's native range included more than one region, that organism's count was split proportionally.
We analyzed the timing of introductions in terms of both the date of first record in the Estuary, and the date of first record in the northeastern Pacific. The numbers of species were tabulated in four 30-year periods with the first beginning in 1850 and the last ending in 1969, and one 26-year period (1970-1995). In the few cases where an organism's date of first record was a period that spanned parts of two tabulation periods, that organism's count was proportionally divided between the periods.
We distinguished two different types of dates of first record. The first and preferred type is the date of initial planting or first observation or collection of the species in the area. Where this was unavailable, we reported the earliest date available (date of writing, submission or publication) of the first written account of the species in the area. In Table 1, dates of first written account are preceded by the symbol '²', meaning that the date of first planting, observation or collection was on or before (in some cases, perhaps a considerable time before) the indicated date. Dates of first written account were excluded from the quantitative analysis.
We also excluded from the analysis those dates of first record that we judged to be a clear artifact of collecting bias, or a fortuitous discovery of a species in a restricted habitat or locality, and whose inclusion would have contributed to a misleading picture of the temporal pattern of invasions in the Estuary. This is discussed further in Chapter 5 under "Results." These dates are marked by asterisks (*) in Table 1.
We analyzed the stocks of organisms that have been introduced to the Estuary in terms of the transport mechanisms (also called "transport vectors," "means of introduction" and "dispersal mechanisms") that brought them to the northeastern Pacific. We utilized thirteen categories of mechanisms, as defined in Table 1 and discussed in Chapter 5 under "Results." Where multiple possible transport mechanisms were determined for an organism, that organism's count was divided proportionally among the possible mechanisms.
Bryopsis sp. [CODIALES]
Silva (1979) reported an unidentified species of Bryopsis
which only reproduces asexually in the Bay and which he described
as exhibiting weedy behavior: developing explosively and frequently
being cast ashore in large quantities, creating a nuisance as
it decomposes. It has been observed in the Bay since at least
1951, from Alameda to Richmond on the East Bay shore and at Coyote
Point. Bryopsis occurs in ship fouling (pers. obs.) and, in concert
with the other introduced seaweeds, we tentatively suggest ship
fouling as the mechanism of introduction.
Codium fragile tomentosoides (Suringar, 1867) Hariot, 1889
[CODIALES]
DEAD MAN'S FINGERS, SPUTNIK WEED, OYSTER THIEF
Codium fragile is native to the northern Pacific, and is found in North America on exposed coasts from Alaska to Baja California (Abbot & Hollenberg, 1976). The weedy subspecies C. f. tomentosoides is native to Japan (where it is eaten) and was introduced to Europe in the nineteenth century and to New York, probably as ship fouling, around 1956, subsequently spreading north to Maine and south to North Carolina (Carlton & Scanlon, 1985; includes discussion of coastal transport mechanisms). It was first collected in San Francisco Bay in 1977, probably introduced as ship fouling (Carlton et al., 1990), and as of 1985 not reported from any other site in the northeastern Pacific (Carlton & Scanlon, 1985).
In San Francisco Bay C. f. tomentosoides is common intertidally
and subtidally attached to rocks, seawalls, piers and floating
docks. Josselyn & West (1985) report it as common (found 60-100%
of the time) at Coyote Point, and frequent (30-60%) at Redwood
City, Palo Alto. In 1993-94 we found it on floating docks in the
East Bay from Richmond to San Leandro and at Pier 39 in San Francisco.
Sargassum muticum (Yendo, 1907) Fensholt, 1955 [FUCALES]
Sargassum muticum is a Japanese species which was first collected in North
America in 1944 in British Columbia, apparently introduced in shipments of Japanese oyster spat (Crassostrea gigas), and subsequently spread both north and south into protected waters. It was reported from Coos Bay in 1947, Crescent City in 1963 and Santa Catalina Island in 1970, and is now found at scattered sites from Alaska to Baja California (Abbott & Hollenberg, 1976; Silva, 1979). It was introduced to Europe in the early 1970s, apparently also in shipments of Japanese oyster spat (Druehl, 1973; Critchley, 1983; Danek, 1984).
S. muticum was first observed in San Francisco Bay by Silva on the riprap at the entrance to the Berkeley Marina in 1973. It has been reported on the pilings of the Golden Gate Bridge, in the San Francisco Yacht Harbor, on the inside breakwater at Fort Baker, at Angel Island, Sausalito and the Tiburon Peninsula, on the east side of Yerba Buena Island, at Crown Beach in Alameda, and from Albany and Richmond (Silva, 1979; Danek, 1984). Josselyn & West (1985) found it commonly (60-100% of the time) at Tiburon Peninsula and infrequently (5-30%) at Twin Sisters.
In San Francisco Bay S. muticum appears to be restricted to low intertidal areas with hard substrate and moderate to high salinity. Germlings grow at salinities down to 10 ppt (to 20 ppt according to Norton (1977)), but maximum survival is at 25-30 ppt salinity. Low salinities and storms eliminated the Tiburon population in the winter and spring of 1983 (Danek, 1984). S. muticum was more abundant at Crown Beach, Alameda during the drought years of 1990-91 than it is at present (pers. obs.).
Both lateral branches and fertile fronds of S. muticum break
off regularly and float and disperse by currents and wind drift,
surviving afloat for up to 3 months, and can initiate new populations
(Danek, 1984). Danek (1984) reports that "in Britain S.
muticum has become the dominant species at low tide levels,
and is a successful competitor against indigenous species such
as Cystoseira and Laminaria...it forms large floating
mats (Fletcher & Fletcher, 1975) causing problems for fishermen
and small boat navigation." An eradication program in England
was "largely unsuccessful" (Silva, 1979). In Canada,
Druehl (1973) considers it to be replacing populations of Zostera
in some places, and Dudley & Collins (1995) report that it
has become a dominant intertidal species in the Channel Islands
and Santa Barbara area. However, Silva (1979) states that "there
is no evidence that S. muticum is displacing the native
biota of San Francisco Bay."
Callithamnion byssoides Arnott [CERAMIALES]
Callithamnion byssoides is native to the northwestern Atlantic
from Nova Scotia to Florida (Taylor, 1957). It was not listed
in Silva's (1979) review of Central Bay benthic algae, but Josselyn
& West (1985) found it attached to rocks "near MLLW throughout
the northern and southern reaches of the bay" in collections
between 1978 and 1983. They report it as frequent (found 30-60%
of the time) at Redwood City, Palo Alto and China Camp, and infrequent
(5-30%) at Tiburon Peninsula, Point
Pinole and Crockett. Callithamnion species are common fouling
species (WHOI, 1952). C. byssoides may have been transported
to San Francisco Bay as ship fouling, or possibly with the algae
used to pack New England bait worms or lobster.
Polysiphonia denudata (Dillwyn) Kützing [CERAMIALES]
Polysiphonia denudata is native to the Atlantic coast from Prince Edward Island to Florida and the tropics, commonly occurring in tide pools and in shallow bays attached to rocks, shells and wharves (Taylor, 1957). It was not listed by Silva (1979) in his review of Central Bay benthic algae, but Josselyn & West (1985) reported it as a "common drift algae during summer months, especially in South San Francisco Bay" (citing Cloern, pers. comm.), and as drift or epiphytic in both San Pablo Bay and South Bay in collections between 1978 and 1983. They further suggest that "the extensive decaying mats observed by Nichols (1979) in Palo Alto during the summer of 1975" may have been P. denudata. We (JTC) observed a sometimes abundant Polysiphonia, which we presume to have been P. denudata, in Lake Merritt, Oakland in 1963-64.
Polysiphonia species are common fouling species or artificial
structures, including ships (WHOI, 1952; Fletcher et al., 1984),
and a species of Polysiphonia was the organism most tolerant
of copper- and mercury-based anti-fouling compounds in tests in
Florida (Weiss, 1947), suggesting that P. denudata probably
arrived in San Francisco Bay as hull fouling, although introduction
by ballast water is possible. Josselyn & West (1985) reported
P. denudata as frequent (30-60% of the time) at Point Pinole,
and infrequent (5-30%) at stations on the western shore of the
South Bay, on the Marin shore, and at Crockett. It apparently
reproduces asexually in San Francisco Bay, and is not reported
from other Pacific coast estuaries (M. Josselyn, pers. comm.,
1985).
Chenopodium macrospermum J. D. Hooker var. halophilum
(Philippi) Standley [CHENOPODIACEAE]
SYNONYMS: Chenopodium macrospermum J. D. Hooker var. farinosum
(Watson) Howell
Probably native to South America, this plant is found in wet places
and marshes at low elevations between Orange County and Washington
state, including the coastal California (Munz, 1959) the San Francisco
Bay Area and the Delta (Hickman, 1993).
Cotula coronopifolia Linnaeus, 1753 [ASTERACEAE]
BRASS BUTTONS
Brass buttons is a native of South Africa that has become established along the Pacific coast from California to British Columbia, and is reported as adventive in New England (Peck, 1941; Muenscher, 1944; Steward et al., 1963). In 1878, Lockington (1878) reported it as an introduced plant common in wet places on the San Francisco peninsula. As it was likely to have spread to the Bay's littoral zone by around that time, we have taken 1878 as the date of first observation in the Estuary. It was probably introduced in ships' ballast (as suggested by Spicher & Josselyn, 1985).
In California brass buttons has variously been reported as common
in salt and freshwater marshes along the coast (Robbins et al.,
1941; Mason, 1957; Munz 1959; Hickman, 1993), as present in San
Francisco Bay saltmarshes (Jepson, 1951), as common in wet places
near high-tide levels in the tidal marshes around Suisun Bay (Atwater
et al., 1979), and as uncommon in the Delta (Madrone Assoc., 1980;
Herbold & Moyle, 1989). A 1981 aerial survey of Suisun Marsh
classified 3,800 acres, or 5% of the area surveyed, as Cotula
habitat (Wernette, 1986), and in 1989 it was found at 18 of 48
sites. Along with alkali bulrush, saltgrass or fat hen, brass
buttons comprised the principal vegetation at two sites in each
of 1987, 1988 and 1989 (Herrgesell, 1990). Waterfowl frequently
graze on brass button seeds, and the diked, brackish marshes around
Suisun Bay are managed in part to promote its growth (Josselyn,
1983).
Lepidium latifolium Linnaeus [BRASSICACEAE]
BROADLEAF PEPPERGRASS, PERENNIAL PEPPERWEED, TALL WHITETOP
Broadleaf peppergrass is a native of Eurasia, where it is reported from Norway to North Africa and east to the Himalayan region. It has been introduced to many parts of the United States, Mexico and Australia, and is found on beaches, tidal shores, saline soils and roadsides throughout most of California (Hickman, 1993; Young & Turner, 1995; May, 1995). Suggested mechanisms of transport to North America along the New England coast prior to 1924 include transport in gluestock (animal bones) shipped from Europe, the seeds adhering to scraps of tissue or burlap sacking (Morse, 1924, cited in May, 1995); with material shipped to a dye and licorice works (Eames, 1935, cited in May, 1995); and clinging to the wool of sheep (Rollins, 1993, cited in May, 1995).
Broadleaf peppergrass was discovered in Montana in 1935, and in California near Oakdale, Stanislaus County in 1936, possibly having been transported with beet seed (May, 1995). By 1941 it was reported from San Joaquin and Yolo counties on the edge of the Delta (Robbins et al., 1941). Herbarium specimens exist from Grizzly Island (collected in 1960), Antioch Dunes (1977) and the Bay shoreline at Martinez and Point Pinole (1978). It was reported as common in the tidal marshes of the San Francisco Estuary (Atwater et al., 1979), and uncommon in the Delta (Madrone Assoc., 1980; Herbold & Moyle, 1989). Recently it has been reported as invasive and spreading in shallow ponds and adjacent moist uplands in the Central Valley wildlife refuges, and in high tidal marsh areas and diked seasonal wetlands in Suisun Marsh (where hundreds of acres on Grizzly Island are affected) and throughout the Bay (Trumbo, 1994; Dudley & Collins, 1995; Malamud-Roam, pers. comm., 1994; May, 1995).
Broadleaf peppergrass produces large amounts of seed, can reproduce asexually by spread of rhizome sections, and is tolerant of a broad range of environmental conditions (Trumbo, 1994; May, 1995). It often becomes established on disturbed, bare soils, and was also observed in pickleweed (Salicornia) plains and among Scirpus spp. (May, 1995). May (1995) reports that it may be intolerant of frequent or prolonged flooding, and our observations suggest that it is limited to the upper edge, or often above the upper edge, of tidal inundation.
Trumbo (1994) suggests that at Suisun Marsh peppergrass first
got established in agricultural areas, then as farms closed during
the 1950s expanded rapidly "unchecked by frequent cultivations
and crop competition" and invaded wildlife areas of the marsh.
He claims that it competes with pickleweed, thereby reducing habitat
for the endangered saltmarsh harvest mouse, and that its dense
growth is unsuitable for use as nesting cover by waterfowl, although
May (1995) reports that waterfowl nests have been observed in
monotypic stands of peppergrass. BDOC (1994) states that it may
outcompete and displace certain rare native marsh plants, such
as Lilaeopsis masoni and Cordylanthus mollis mollis.
CDFG has tested burning, discing and herbicide treatments as control
measures for pepper grass, which is ranked as a "B"-level
plant pest by the California Department of Food and Agriculture
(BDOC, 1994).
Limosella subulata Ives, 1817 [SCROPHULARIACEAE]
AWL-LEAVED MUDWORT
Limosella subulata is native to Europe or the east coast
of North America, and found in southern British Columbia and in
fifteen western states. It is reported from muddy and sandy intertidal
flats in the Delta (Muenscher, 1944; Munz, 1959; Atwater et al.,
1979; Herbold & Moyle, 1989; Hickman, 1993).
Lythrum salicaria Linnaeus [LYTHRACEAE]
PURPLE LOOSESTRIFE
Native to Europe, purple loosestrife is invasive worldwide. It was introduced to North America by the early 1880s, either as seeds in solid ballast or in the wool of sheep, or as a cultivated plant. It can grow in monospecific stands, competes with cattails and other marsh plants (Mills et al., 1993), and is listed as a noxious weed in California (Hickman, 1993).
Purple loosestrife was reported by Munz (1968) in Nevada and Butte
counties, but not mentioned by Munz (1959) or Mason (1957). It
is now found in low elevation marshes, ponds, streambanks and
ditches throughout much of California, including the Sacramento
Valley and the Bay Area (Hickman, 1993).
Myriophyllum aquaticum (Velloso) [HALORAGACEAE]
PARROT'S FEATHER
SYNONYMS: Myriophyllum brasiliense Cambess.
A South American native, parrot's feather is found in ponds, ditches,
streams and lakes in warm temperate and tropical regions throughout
the world. Escaped from cultivation in California and reported
from six counties from Humboldt to San Diego ("set out in
these areas by dealers in aquatics for the purpose of market propagation;"
Mason, 1957), from the Coast and Cascade ranges and from central
western California (Hickman, 1993), and from tidal marshes and
sloughs in the Delta (Atwater et al., 1979; Madrone Assoc., 1980).
BDOC (1994) reports that parrot's feather "provides excellent
mosquito habitat," and that the USDA has investigated the
use of herbicidal and biological controls.
Myriophyllum spicatum Linnaeus [HALORAGACEAE]
EURASIAN MILFOIL
SYNONYMS: Myriophyllum exalbescens in part
Eurasian milfoil is a native of Eurasia and North Africa that has invaded lakes in the eastern United States and Canada. Its first documented occurrence in North America was in the Potomac River, Virginia in 1881, though it is thought to have arrived much earlier (Reed, 1977, cited in Mills et al., 1993). In the early 1970s it reportedly made up over 90 percent of the plant biomass in Lake Cayuga, New York, where it may have been eventually controlled by an exotic moth, Acentria niveus (Anon., 1994). Control efforts have also included cutting, water drawdown and herbicide applications (Mills et al., 1993). Eurasian milfoil reportedly can outcompete native plants through shading, clog pipes and entangle boat propellers, and foul beaches with decaying mats of dead plants. It spreads as discarded material from aquaria and entangled on boats and trailers moved between watersheds (Mills et al., 1995).
Hickman (1993) reports this plant as uncommon in ditches and lake
margins in the Bay Area and the San Joaquin Valley, and BDOC (1994)
reports it from the Delta. Munz (1959) reported Myriophyllum
spicatum ssp. exalbescens common throughout cismontane
California in quiet water below 8,000 feet, Atwater et al. (1979)
reported M. s. ssp. exalbescens in Snodgrass Slough
on the Sacramento River in the Delta in 1976, and Madrone Assoc.
(1980) reported water milfoil (as M. s. var. exalbescens
and M. exalbescens) common in the Delta. Hickman (1993)
states that M. s. ssp. exalbescens was misapplied
to M. sibiricum, which he treats as a native (but which
we consider cryptogenic (Table 2) based on its reported range
which includes Pacific coastal and eastern Northern America and
Eurasia). Based on reported distribution and abundance, we consider
Munz's (1959) exalbescens to be M. sibiricum and
the Delta reports of exalbescens since 1976 to refer, at
least in part, to M. spicatum.
Polygonum patulum Bieberstein [POLYGONACEAE]
SMARTWEED
Native to eastern Europe, Polygonum patulum is reported
as uncommon in and around salt marshes in the Bay and Delta area
(Munz 1959; Hickman, 1993). It belongs to a closely related (and
possibly hybridizing) group of introduced or cryptogenic species,
often found in or adjacent to fresh or saline wetlands, including
Polygonum aviculare (cryptogenic), argyrocoleon
(Asian), prolificum (eastern North America) and punctatum
(cryptogenic).
Rorippa nasturtium-aquaticum (Linnaeus) Hayek [BRASSICACEAE]
WATERCRESS
SYNONYMS: Nasturtium officinale R. Br.
Radicula nasturtium-aquaticum (Linnaeus) Britt. & Rendle
Rorippa nasturtium Rusby
Sisymbrium nasturtium-aquaticum
Watercress is a perennial aquatic plant native to Europe which has been widely cultivated for its edible greens, and which has escaped and become common throughout North America in marshes, in slowly flowing creeks, around seeps, on wet banks, etc. Though probably present earlier, established populations were first reported from North America near Niagara Falls in 1847 and at Ann Arbor, Michigan in 1857 (Gray, 1848; Green, 1962; Mills et al., 1993). Peck (1941) reported it widely distributed in Oregon and Muenscher (1944) reported it from 41 states including California, Oregon and Washington.
Watercress is found in the Delta (Munz, 1959; Herbold & Moyle,
1989). Most authors (e. g. Jepson, 1951; Munz, 1959; Mills et
al., 1993, 1995; BDOC, 1994) consider this plant to be an introduction
from Europe, although Hickman (1993) treats it as a native plant
of temperate world-wide distribution.
Salsola soda Linnaeus [CHENOPODIACEAE]
Native to southern Europe, Salsola soda is found on mudflats, in open areas and among pickleweed in salt marshes, and on berms, among riprap and in open areas at and above the high tide mark at scattered sites in San Francisco Bay (Hickman, 1993; pers. obs.). It was first collected in July 1968 at the west end of the Dumbarton Bridge in the South Bay (Thomas, 1975). It has since been found at several sites in the South Bay from Candlestick Park to the San Francisco Bay National Wildlife Refuge, and on the Alameda shore; from Emeryville Marina to Hoffman Marsh, Richmond and at Richardson Bay in the Central Bay; and at Chevron Marsh, Richmond, at Pinole and at Tubbs Island in San Pablo Bay (Thomas, 1975; Tamasi, 1995; pers. obs.). At the Pinole shore it appears to be successfully competing with pickleweed Salicornia virginica in the high marsh, and like pickleweed is attacked by the parasitic plant Cuscuta salina (pers. obs.). A few plants were observed on a mudflat in Bodega Harbor in the summer of 1994 but not in 1995 (Connors, 1995; C. Daehler, pers. comm., 1995).
Its mechanism of introduction is something of a mystery, as no
known modern transport vectoróexcepting the unlikely possibility
of its use (and escape) as an ornamental plantóappears
to apply.
Spergularia media (Linnaeus) Grisebach [CARYOPHYLLACEAE]
SAND SPURREY
SYNONYMS: Arenaria media
Hickman (1993) noted that "Spergularia maritima (All.)
Chiov. may prove to be the correct name" for this species.
Sand spurrey is native to coastal Europe and has been introduced
to South America, eastern North America and Oregon. It is found
on salt flats, in and bordering salt marshes, and on sandy beaches
in Marin and Contra Costa counties (Munz, 1959; Hickman, 1993).
Atwater et al. (1979) listed it as common in tidal marshes of
the San Francisco Estuary.
Egeria densa Planchon [HYDROCHARITACEAE]
ELODEA, EGERIA, BRAZILIAN WATERWEED
SYNONYMS: Elodea densa (Planchon) Caspary
Anacharis densa (Planchon) Marie-Victorin
Elodea is a highly invasive aquatic plant from South America that clogs waterways and interferes with navigation. In 1944 Muenscher reported it as a recently established introduction in six eastern states from Massachusetts to Florida and in California, Steward et al. (1963) reported it from Oregon, and it has also become established in Europe (Hickman, 1993). It is widely used in aquaria and ornamental pools, and was probably introduced as discarded material or as an escape (Muencher, 1944; Munz, 1959). In California it was reported as infrequent at scattered locations by Mason (1957), and is now found on both sides of the Sierra Nevada, in the San Joaquin Valley, and in the San Francisco Bay area (Hickman, 1993).
Elodea is reported as common in waterways throughout the Delta
and in the Contra Costa Canal (Atwater et al., 1979; Herbold &
Moyle, 1989; Holt, 1992). It was found at 8 of 10 sites in the
Delta surveyed for littoral zone vegetation in 1988-90 (IESP,
1991). In the 1990s it has spread to new areas and deeper water
in the Delta and become more abundant, perhaps due to lower summer
water levels and warmer water temperatures (Holt, 1992; Thomas,
pers. comm.). Although elodea provides shelter for newly hatched
fish, it also clogs channels and berths, gets caught in water
intake of engines, and fouls propellers. Management of this species
included the use of an aquatic weed killer on about 35 acres of
Delta waterways in 1991 (Holt, 1992). Field tests are being conducted
on the use of Komeen, a copper-based herbicide, and biocontrol
agents are being investigated (Rubissow, 1994; BDOC, 1994).
Eichhornia crassipes (Martius) Solms-Laubach, 1883 [PONTEDERIACEAE]
WATER HYACINTH
Water hyacinth, "perhaps the world's most troublesome aquatic weed" (Hickman, 1993) is a native of tropical South America that has spread to more than 50 countries on five continents, and has become a massive problem in waterways in both Africa and Southeast Asia (Barrett, 1989). Its air-filled tissue (aerenchyma) enables it to float and spread rapidly within and between connected water bodies. It reproduces asexually by breaking apart into pieces each of which develops into a separate plant. This results in a rapid increase in biomass, and continuous mats of living and decaying water hyacinth up to two meters thick covering the water surface have been reported (Barrett, 1991).
Water hyacinth was introduced to North America in 1884 via the Cotton States Exposition in New Orleans. The plant was displayed in ornamental ponds and distributed as souvenirs to visitors, with the excess dumped into nearby creeks and lakes (Barrett, 1989; Joyce, 1992). It spread across the southeastern U. S. to Florida, where a 1895 invasion of the St. Johns River produced floating mats of water hyacinth up to 40 kilometers long (Barrett, 1989), and in several southeastern sites blocked the passage of steamboats and other vessels by 1898 (Joyce, 1992). According to Joyce, these problems led to the passage of the River and Harbor Act in 1899, authorizing the U. S. Army Corps of Engineers to maintain navigation channels in these areas. Control efforts included the spraying of sodium arsenite, which poisoned applicators and livestock (Joyce, 1992).
The 1884 Cotton States Exposition was probably also the initial source of the water hyacinth that was reported from the Sacramento River near Clarksburg, California, in 1904 (Thomas & Anderson, 1983; Thomas, pers. comm., 1994). In California, water hyacinth spread gradually for many decades. Robbins et al. (1941) reported it from the Kings River in Fresno County and Warner Creek in San Bernardino County. It reached the Delta by the late 1940s or early 1950s, where the federal Bureau of Reclamation tried controlling it with herbicides around 1957 (Thomas & Anderson, 1983; L. Thomas, pers. comm., 1994). In 1959 Munz reported it as occasionally established in sloughs and sluggish water in the Sacramento and San Joaquin valleys and the Santa Ana River system. In 1972 the U. S. Army Corps of Engineers investigated water hyacinth on the Merced River and determined that it was not a flood hazard (Thomas & Anderson, 1983; L. Thomas, pers. comm., 1994). Atwater et al. (1979) listed it as common in tidal marshes, presumably in the Delta. Madrone Assoc. (1980) reported it as seasonally common in the southern and central Delta and clearing in the winter, when coot and other waterfowl fed on the dead plants.
Starting in the 1980s water hyacinth became a serious problem in the Delta watershed, blocking canals and waterways, fouling irrigation pumps, shutting down marinas, blocking salmon migration and, by 1982-83, blocking ferry boats at Bacon Island and preventing the island's produce from being shipped to market (CDBW, 1994; L. Thomas, pers. comm., 1994). The plant's abundance may have been drought-related, with plant densities building up when low river flows were unable to flush the year's growth out of the Delta. On the other hand, when a wet year arrived in 1993 the higher rainfall "washed surplus plants from the upstream channels into the Delta where it created a major problem by early summer, and it also appeared to trigger unprecedented seed growth." High flows also lowered chloride levels enabling plants to grow in parts of the western Delta that had previously been clear (CDBW, 1994).
On June 14, 1982 California Senate Bill 1344 became law, directing the California Department of Boating and Waterways (CDBW) to control water hyacinth in the Delta. CDBW set up barriers to keep large masses of floating plants out of navigation channels and sprayed the herbicides Weedar (2,4-D), Diquat and Rodeo (glyphosphate), at a cost that rose to about $400,000 annually. Program Supervisor Larry Thomas claims that if herbicides had not been used in 1986-1991, "water hyacinth would have shut the Delta down" (L. Thomas, pers. comm., 1994)
In some areas mechanical harvesting has been used to control hyacinth, but this is expensive (typically around $1,500 to $3,000 per acre) and disposal of the hyacinth can be a problem. Because of the cost, CDBW does not use mechanical harvesting (L. Thomas, pers. comm., 1994).
In 1982 and 1983 CDBW, working with the U. S. Department of Agriculture, imported and released three insects from South America as biological controls, the moth Sameodes albiguttalis (which did not survive) and the weevils Neochetina bruchi and N. eichhorniae. Although the two weevils became established in the Delta, there is no evidence that they control water hyacinth (Thomas & Anderson, 1983; L. Thomas, pers. comm., 1994).
Of the three flowering forms of water hyacinth, only medium-style plants have been found in California even though these plants are heterozygous for style length. This suggests that water hyacinth does not reproduce sexually in California. Conditions preventing sexual reproduction may include a lack of effective insect pollinators foraging in hyacinth (although honeybees Apis mellifera may be effective where they visit hyacinth), and a lack of open shallow water or saturated soil sites which are needed for germination and seedling establishment (Barrett, 1980, 1989).
Today water hyacinth is locally abundant in ponds, sloughs and waterways in the Central Valley, the Bay Area, and the southern Coast and Peninsular ranges (Hickman, 1993), and very dense in many waterways in the Delta. In 1988-1990 it was found in 4 of 10 sites in the Delta surveyed for littoral zone vegetation (IESP, 1991). In 1993 hyacinth again became very dense in parts of the Delta and the San Joaquin Valley drainage, despite herbicide treatment of around 1,500 acres (CDBW, 1994).
In the Philippines, the leaves of this troublesome weed are sold
as a market vegetable under the name of "waterlilly"
or "dahon" (Ladines & Lontoc, 1983).
Iris pseudacorus Linnaeus [IRIDACEAE]
YELLOW FLAG, YELLOW IRIS
A native of Europe, Iris pseudacorus was a popular garden flower that escaped from cultivation. The first populations reported in North America were from near Poughkeepsie, New York in 1868, from a swamp near Ithaca, New York in 1886 and from Massachusetts in 1889, and it was first reported from Canada at Ontario in 1940 (Mills et al., 1993, 1995). It is now widespread east of the Rocky Mountains (Hickman, 1993).
Jepson (1951) did not mention Iris pseudacorus, but Mason
(1957) reported that it "has escaped in Merced County and
is apparently moving down the watercourses." It has since
been found in irrigation ditches and pond margins in the San Francisco
Bay area, in the southern San Joaquin Valley, and in Sonoma County
(Munz, 1968; Hickman, 1993). Atwater (1980) found it was the only
common introduced plant on Delta islets, reporting it from the
banks of 4 out of 6 islets surveyed in 1978-79.
Polypogon elongatus Kunth, 1815 [POACEAE]
Native to South America, this plant is found in salt marshes and
on sand dunes in the Bay Area, including Contra Costa County,
and in the southern Coast Range (Munz, 1959, Hickman, 1993).
Potamogeton crispus Linnaeus, 1753 [POTAMOGETONACEAE]
CURLY-LEAF PONDWEED, CURLY PONDWEED
This pondweed is native to Europe and now found more-or-less worldwide, including Atlantic North America, California and Oregon (Steward et al., 1963). The earliest verified records in North America are from Delaware and Pennsylvania in the 1860s, although reports of it date back to 1807. It was deliberately introduced into parts of the Great Lakes basin to provide food for waterfowl, and is associated with fish hatcheries having perhaps been accidentally transported between watersheds in conjunction with fish stocking activities (Mills et al., 1993 citing Stuckey, 1979). It reportedly can grow in fresh, brackish or salt water (Mills et al., 1995).
It is uncommon in shallow water, ponds, reservoirs and streams
across most of cismontane California including the Bay Area and
the Central Valley (Munz, 1959; Hickman, 1993). In 1988-90 it
was found in 2 of 10 sites surveyed for littoral zone vegetation
in the Delta (IESP, 1991).
Spartina alterniflora Loiseleur-Deslongchamps [POACEAE]
SMOOTH CORDGRASS, SALT-WATER CORDGRASS
Spartina alterniflora is native to the coast of eastern North America from Maine to Texas (Muenscher, 1944) and has been introduced to Padilla Bay (1910), Thorndyke Bay (1930), Camano Island and Whidbey Island in Washington; the Siuslaw Estuary in Oregon; and New Zealand, England (1922) and China (1977) (Chung, 1990; Callaway, 1990; Callaway & Josselyn, 1992; Ratchford, 1995). Most literature states that S. alterniflora was first introduced to the northeastern Pacific in Willapa Bay, Washington, but both the date and mechanism of introduction to this site are unclear. In a brief note Scheffer (1945) reported first becoming aware of a cordgrass in Willapa Bay "about seven years ago"óthus about 1938óthat was identified as S. alterniflora in 1941. An oysterman reported first seeing the plants "about 1911," and Scheffer, believing that the first Atlantic oysters (shipped from Rhode Island) had been planted in Willapa Bay about 1907, concluded (apparently based on the coincidence in dates) that the cordgrass had been introduced with the oysters.
Sayce (1988) pointed out that Scheffer was mistaken about the initial date and origin of Atlantic oyster shipments to Willapa Bay, reporting that in fact the first shipment, of 80 barrels of oysters from estuaries near New York City and Chesapeake Bay, occurred in 1894, and that there were no subsequent introductions of Atlantic oysters for the next 50 years (although Carlton (1979a, p. 72) reports introductions of Atlantic oysters to Willapa Bay occurring in 1874 and 1894-1920s). Sayce did, however, continue to associate Spartina alterniflora with oyster shipments, stating that the Atlantic cordgrass was introduced with the 1894 shipment. She explained, "When the oysters were packed in barrels, in all likelihood the packing material was "salt grass" of one of two species, Spartina alterniflora or S. patens. S. patens has not been found in Willapa Bay. Either viable seeds or rhizomes of Spartina alterniflora were in the packing material." Nearly all subsequent authors have followed Sayce in reporting that S. alterniflora arrived in Willapa bay in 1894 as packing material for oysters. However, we have found no record of cordgrass ever having been used as packing material for any oyster shipments, nor is there any reason to think that hard-shelled oysters packed in barrels would need or benefit from additional packing. Thus, there is no basis for concluding that S. alterniflora was introduced to Willapa Bay in 1894.
Accordingly, we consider the first record of S. alterniflora in Willapa Bay to be "about 1911," and suggest solid ballast as the likeliest transport mechanism. Molecular genetic comparisons with east coast populations may clarify the source of the S. alterniflora stock in Willapa Bay (as has been done for San Francisco Bay S. alterniflora; C Daehler, pers. comm., 1995), providing additional information to resolve the probable means of transport.
Spartina alterniflora was separately introduced to San Francisco Bay in the early 1970s by the U. S. Army Corps of Engineers as mitigation for wetlands destroyed in the construction of the New Alameda Creek Flood Control Channel or as an experimental planting (anecdotal accounts and genetic analysis both indicating that the stock originated from Maryland; C. Daehler, pers. comm., 1995). It was planted at Pond 3 at the Coyote Hills Regional Shoreline. One source reported that after plantings of the native cordgrass S. foliosa did poorly, the area was replanted with the more robust S. alterniflora to produce a "successful" restoration.
S. alterniflora from Coyote Hills was later transplanted to San Bruno Slough near the San Francisco Airport by the Caltrans agency, either as mitigation for the Samtrans Bus Terminal or for erosion control. It may also have been planted in the Elsie Roemer Wildlife Refuge on the southwest shore of Alameda Island as part of yet another "restoration" project in 1983 or 1984, or for erosion control by the City of Alameda. It was found in Hayward Marsh in 1989 (Spicher & Josselyn, 1985; Calloway, 1990; Kelly, pers. comm., 1992; Faber, pers. comm., 1993; Taylor, pers. comm., 1993; Cohen, 1993).
In San Francisco Bay S. alterniflora is found both within existing salt marshes and extending into lower elevation mudflats. Comparing aerial photographs of the mouth of Coyote Hills Slough, Callaway (1990) saw no S. alterniflora in 1981 but counted 31 round patches in 1988 and 146 patches in 1990. Daehler & Strong (1994) found that "although some dense monocultures have formed," most S. alterniflora was growing in discrete circular patches separated by open mud, determined by isozyme analysis to consist of individual genetic clones. There are now a total of about 1,000 round or donut-shaped patches at southwestern Alameda Island and northeastern Bay Farm Island, San Leandro Bay, Hayward Marsh, Alameda Creek and Coyote Hills Slough (New Alameda Creek), and San Bruno Slough (near the San Francisco Airport). Smaller amounts are reported from the Estudillo Flood Control Channel south of the San Leandro Marina, the San Francisco Bay National Wildlife Refuge and the Cargill salt ponds near Newark, and the National Wildlife Refuge near Alviso (M. Taylor, pers. comm., 1993; J. Takekawa, pers. comm., 1994; C. Daehler, pers. comm., 1995).
New patches of S. alterniflora are established both from seed and vegetative fragments (Daehler & Strong, 1994). The cordgrass apparently arrived in Hayward as floating rhizomes (M. Taylor, pers. comm., 1993) and may be spread by dredges within the Cargill salt ponds (D. Strong, pers. comm., 1993). Daehler & Strong (1994) observed about 75 percent of patches setting very little seed in 1991-1992, and germination rates ranging from zero to 59 percent, and suggested that a few clones may be producing most of the seeds. On the other hand, Callaway (1990) found higher seed production (2,475 vs. 371 seeds/m2), higher seed viability (97% vs. 67%) and higher germination rates (average germination percentages of 77% vs. 49% in freshwater, and 37% vs. 14% in 25 ppt salinity) for S. alterniflora than for the native cordgrass Spartina foliosa in San Francisco Bay.
Spartina alterniflora grows both higher and lower in the intertidal zone than S. foliosa (Calloway, 1990; D. Strong, pers. comm., 1993; in Willapa Bay its total vertical range is at least 66 percent of the tidal range, Sayce, 1988), and can accrete sediment at a rapid rate (Sayce, 1988; Josselyn et al., 1993). By growing at a lower elevation it may reduce the area of mudflats in San Francisco Bay as it has in Willapa Bay, Washington, where it has turned an estimated 1,800-2,400 acres (5-6 percent) of Willapa Bay's mudflats into cordgrass islands (Ratchford, 1995). Callaway & Josselyn (1992) listed potential adverse impacts as: competitive replacement of native cordgrass; altered habitat for native wetland animals because of larger and more rigid stems and greater stem densities; altered habitat for infauna because of higher root densities; changed sediment dynamics; decreased benthic algal production because of lower light levels below cordgrass canopy; and loss of shorebird foraging habitat through colonization of mudflats. In British estuaries, the invasion of mudflats by Spartina anglica has produced adverse effects on shorebirds (Goss-Custard & Moser, 1990; Callaway, 1990).
The potential loss of native cordgrass is of particular concern, because it provides habitat for the severely endangered California clapper rail, Rallus longirostris obsoletus. On the other hand, S. alterniflora could possibly provide more and better cover and therefore better protection for the rail, which is threatened by predation by the introduced red fox, Vulpes vulpes (P. Kelly, pers. comm., 1992; Cohen, 1992, 1993).
In San Francisco Bay, S. alterniflora is attacked by the sap-feeding planthopper Prokelisia marginata at densities (ranging from 116 to 332 insects per inflorescence) much higher than typically observed on the Atlantic coast, and by the sap-feeding mirid bug Trigonotylus uhleri. However, this does not appear to affect growth rates, seed production or germination rates (Daehler & Strong, 1994, 1995).
The California Department of Fish and Game eliminated S. alterniflora
from Humboldt Bay in about 5 years by constructing
a dike around a clump "the size of a house" and covering
it with black plastic, at a cost of $30,000 to $40,000 (M. Taylor,
pers. comm., 1993; D. Strong, pers. comm., 1993). Burning and
herbicides have been tried in Great Britain (P. Kelly, pers. comm.,
1992). After trying weed eaters and burning, the East Bay Regional
Park District's current control strategy at Hayward Marsh is to
cover with black plastic. The herbicide Rodeo (glyphosphate) has
been used at San Bruno Slough. Smooth cordgrass has now so thoroughly
clogged the New Alameda Creek Flood Control Channel (the project
for which the plant was originally introduced as mitigation) that
the Army Corps has proposed 5 years of helicopter-spraying Rodeo
in the channel (P. Baye, pers. comm., 1994).
Spartina anglica C. E. Hubbard, 1968 [POACEAE]
ENGLISH CORDGRASS
The western Atlantic cordgrass Spartina alterniflora (2n=62)was introduced in ship ballast to Southampton Water on the south coast of England, where it was collected in 1829. S. alterniflora there hybridized with the British cordgrass S. maritima (2n=60), producing a sterile F1 hybrid known as S. townsendii or S. x townsendii (2n=62) which was first collected in 1870 near Southampton, though not recognized as a hybrid until 1956. Chromosome doubling in this hybrid produced a fertile form (2n=120-124), probably present by the late 1880s as evidenced by a marked expansion of range, and collected in 1892. S. maritima disappeared from Southampton and nearby areas as the new form multiplied (Marchant, 1967). In 1968 Hubbard recognized this form as a separate species and named it S. anglica. This new species has proved to be an effective invader of both formerly unvegetated mudflats and of salt marsh, and, through a combination of transplantings for marsh reclamation purposes, vigorous clonal growth and natural dispersal, it now occupies 10,000 hectares (25,000 acres) of the British coast (Spicher & Josselyn, 1985; Thompson, 1991).
Another dimension to this story is provided by Chevalier's suggestion (1923; reported by Marchant, 1967) that S. maritima is itself not native to Great Britain, but was introduced there with shipping (possibly in solid ballast) from Africa.
S. anglica was reported from France by 1894, where it spread rapidly (Marchant, 1967). To control shoreline erosion and create salt marshes, S. anglica has been exported from England to many parts of the world, including Germany, Denmark, the Netherlands, China (where it now occupies over 36,000 hectares, almost entirely derived from 21 plants introduced in 1963), Australia and New Zealand (in 1930, where it was later declared a "noxious weed") (Hedgpeth, 1980; Spicher & Josselyn, 1985; Chung, 1990; Callaway, 1990; Callaway & Josselyn, 1992). Chung (1990) listed as additional reasons for planting S. anglica in China the accretion of land for reclamation; the amelioration of saline soils; the production of green manure; the provision of pasture and fodder for sheep, goats, mules, donkeys, horses, pigs, cattle, dairy cows, buffalo, rabbits and geese; the production of feed for tilapia, grass carp and other farmed fish; the increased production of nereid worms for export sale and of other invertebrates; the creation of biomass for fuel production; and the production of raw material for paper-making.
In 1961 or 1962 the U. S. Department of Agriculture and Washington State University introduced what was then known as S. townsendii into Puget Sound, Washington. Ramets of these plants were introduced into San Francisco Bay at Creekside Park Marsh, Marin County, as part of a marsh restoration project in 1977. Botanists realized these plants were in fact S. anglica when they flowered in 1983 (Spicher & Josselyn, 1985; Callaway, 1990).
In England S. anglica has hampered shorebird movement and feeding and correlates with a decline in dunlin (Calidris alpina) numbers (Goss-Custard & Moser, 1990), and has reduced macroinvertebrate densities (Callaway, 1990).
S. anglica has proved to be highly invasive in many parts
of the world (e. g. southern Great Britain, new Zealand and China),
and Thompson (1991) argued that S. anglica was a more successful
invader in Europe than the similar S. alterniflora because
of greater vigor and selective advantages conferred by allopolyploidy.
However, in San Francisco Bay S. alterniflora is the aggressive
invader while S. anglica has not spread from the marsh
where it was originally planted (Spicher & Josselyn, 1985).
Daehler (pers. comm., 1994) suggests that the Bay is near the
equatorial limit of S. anglica's potential range, a supposition
supported by S. anglica's production of only 20% viable
seeds in 1983 and failure to flower in 1984 (Spicher & Josselyn,
1985).
Spartina densiflora Brongniart [POACEAE]
DENSE-FLOWERED CORDGRASS
Spartina densiflora is native to Chile and was introduced
to Humboldt Bay in the mid-nineteenth century, probably in the
shingle ballast of lumber ships returning from Chile (a mechanism
also thought to be involved in the transport of the shorehopper
Transorchestia enigmatica to San Francisco Bay). S.
densiflora was transplanted from Humboldt Bay to Corte Madera
Marsh in 1976 as part of a restoration project at a time when
it was thought to be an ecotype of the native S. foliosa.
(Spicher & Josselyn, 1985; Callaway, 1990; Faber, pers. comm.,
1993). It is currently found in salt marshes at Creekside Park,
Corte Madera Creek, Muzzi Marsh and Greenwood Cove, all in southeastern
Marin County (Spicher & Josselyn, 1985).
Spartina patens (Aiton) Muhlenberg [POACEAE]
SALTMEADOW CORDGRASS, SALT HAY
Saltmeadow cordgrass is native to the eastern United States from Maine to Texas and reported rarely from inland marshes in New York and Michigan. Meadows of this cordgrass were sometimes harvested for hay used in packing and bedding material (Muencher, 1944).
Munz (1968) listed Spartina patens as "reported from Southampton Bay in a marsh, northwest of Benicia, Solano County, Mall." Atwater et al. (1979) referred to "R. E. Mall's report of salt hay at Southampton Bay" but could not find it there or elsewhere in the estuary. In 1985 Spicher & Josselyn again found "an existing patch" of the plant in Southampton Marsh which "does not appear to have spread from its original location," and in 1993 Josselyn et al. listed it from San Bruno Slough in the South Bay. Spartina patens was also introduced to Cox Island, Siuslaw River, Oregon in 1930 (Callaway, 1990), and to China in 1977 (Chung, 1990).
Given that various Spartina species have been extensively
transplanted around the globe, and that S. patens was intentionally
planted in Oregon, it seems probable that S. patens arrived
in San Francisco Bay as a component of some marsh restoration
or erosion control project (transplanted either from Oregon or
the east coast).
Typha angustifolia Linnaeus, 1753 [TYPHACEAE]
NARROW-LEAF CATTAIL, NAIL ROD
Narrow-leaf cattail is native to Eurasia and was reported as a rare member of the coastal flora of the eastern United States in the 1820s (Mills et al., 1993). It is now common in the northeastern states and Canada, and found inland to the Great Plains, in California and in South America.
Jepson (1951) reported it from Inyo County south to cismontane southern California, and by 1959 Munz reported it from marshes in central California. Hickman (1993), who describes it as "possibly naturalized in California," reports it from the central and southern coastal region of California, including the San Francisco Bay Area, and inland to the Central Valley and Lake Tahoe. Josselyn (1983) described it as one of the dominant species in the middle elevation zone of tidal brackish marshes in San Francisco Bay.
Hybrids with the native Typha latifolia are common in central
California including San Francisco Bay tidal marshes, and are
known as Typha x glauca (Munz, 1968; Josselyn, 1983;
Hickman, 1993).
Several workers have investigated the ciliate protozoans that live with or in the introduced mollusks and boring/burrowing isopods of San Francisco Bay. We regard those species originally described from Atlantic waters as being introduced with their hosts into the Bay. Ancistrumina kofoidi, treated here as a cryptogenic species (Table 2), is an additional probable introduction.
Mechanisms of introduction of commensal and symbiotic protozoans
are the same as their hosts, and are discussed with the latter.
Mechanisms of introduction of free-living attached or errant protozoans
include ship-fouling, ship-ballast (rock, sand, and water), and
the planting of commercial oysters.
Trochammina hadai Uchio
This brackish water, benthic foraminifer is native to Japan. It has been found in sediment cores collected in 1990-93 from six stations in the South Bay and from three stations in the Central Bay near the Marin County shore. It has not been found in over 140 sediment samples collected in 1964-70 and 1980-81 from throughout the Bay (D. Sloan, pers. comm., 1995; McGann, 1995; McGann & Sloan, 1995), suggesting that the introduction occurred in the 1980s.
Furthermore, where it is present T. hadai appears to be abundant in the upper sections of cores, less abundant in lower sections, and absent at depth. For example, in a core from the South Bay, T. hadai accounts for 52.2% of the benthic foraminifera in the top 2.5 cm, 8.8% at 8-10 cm depth, 0.7% at 18-20 cm depth, and is absent from the next 33 sections examined down to 352 cm depth (McGann, 1995). In a core taken from Richardson Bay in the Central Bay, T. hadai accounts for 16% of the foraminifera at 0-2 cm from the surface, 38% at 20-22 cm, 26% at 40-42 cm, 23% at 60-62 cm, 18% at 80-82 cm, 2% at 100-102 cm and less than 1% at 120-122 cm (D. Sloan, pers. comm., 1995). This pattern of depth distribution is likely due to bioturbation or other types of sediment disturbance mixing foraminifer tests from recently-deposited, near-surface sediments downward into deeper and earlier-deposited sediments. T. hadai's depth distribution may thus provide a means of measuring the physical and biological processes that mix sediments in different parts of the Bay, which, aside from telling us something about those processes, will be critical to efforts to use sediment cores to decipher the Bay's environmental history.
Although foraminifera have sometimes been observed in some types
of fouling (WHOI, 1952; ANC, pers. obs.), transpacific transport
in ship fouling seems unlikely for this benthic organism. Bottom
sediments and presumably benthic foraminifera as well are sometimes
churned up by wind turbulence or ship activity and taken in along
with water into ballast tanks; and foraminifera have been reported
from ballast water, though rarely (Carlton & Geller, 1993).
A benthic foraminifer could readily be transported with commercial
shipments of oysters, but there have been no significant plantings
of Japanese oysters in San Francisco Bay since the 1930s (Carlton,
1979a). A possible mechanism is transport in mud on anchors or
on anchor chains in chain lockers, as discussed by Schormann et
al. (1990).
Ancistrocoma pelseneeri Chatton & Lwoff, 1926
SYNONYMS: Parachaenia myae
This ciliate was described as Parachaenia myae by Kofoid
and Bush (1936) from the pericardial region and excurrent siphons
of the introduced clam Mya arenaria in San Francisco and
Tomales bays. Kozloff (1946) subsequently reported it from another
introduced clam, Macoma balthica, and from several native
clams in San Francisco and Tomales bays, and synonymized it with
the Atlantic ciliate Ancistrocoma pelseneeri, described
from Macoma balthica in Europe.
Ancistrum cyclidioides (Issel)
Kozloff (1946) recorded this European ciliate from the introduced
clam Mya arenaria in San Francisco Bay.
Boveria teredinidi Nelson, 1923
Pickard (1927) recorded this Atlantic protozoan from the gills
(ctenidia) of the introduced Atlantic shipworm Teredo navalis
in San Francisco Bay.
Sphenophyra dosiniae Chatton & Lwoff, 1926
This European ciliate was reported by Kozloff (1946) from the
introduced clam Mya arenaria and the native clam Cryptomya
californica in San Francisco Bay.
Cothurnia limnoriae Dons, 1927
This peritrich protozoan is found on the joints of the legs of
the introduced wood-boring isopod Limnoria (Mohr, 1959)
(in San Francisco Bay, as discussed elsewhere, only non-native
species of this gribble occur). It was reported from San Francisco
Bay by Kofoid & Miller (1927, p. 330, as Cothurnia
sp.), although it may have been present since Limnoria's
introduction about 1870. Although first described from Europe,
and later reported from southern California (Mohr, 1951), its
origins, like those of its host, are not known.
Lobochona prorates Mohr, LeVeque & Matsudo, 1963
This chonotrich protozoan occurs on the bristles (setae) of the
gills (pleopods) of the introduced wood-boring gribble Limnoria;
as with other gribble associates and the host species discussed
here, the origin is not known. Lobochona prorates was reported
by Kofoid & Miller (1927, p. 330, as Spirochona sp.;
see Mohr, 1966, p. 539) from San Francisco Bay, but may have been
introduced about 1870 with the isopod itself. It is widely reported
from southern California harbors (Carlton, 1979a).
Mirofolliculina limnoriae (Girard, 1883) Dons, 1927
SYNONYMS: Folliculina sp.
This heterotrich protozoan lives on the back of the pleotelson
of the introduced gribble Limnoria. As with the other Limnoria
associated ciliates, it is undoubtedly introduced, but its origins
remain unknown. It was reported from San Francisco Bay by Kofoid
& Miller (1927, p. 330, as Folliculina sp.).
Cliona sp.
BORING SPONGE
While the species level taxonomy of this yellow, shell-boring
sponge remains unresolved, Cliona is almost certainly represented
by one or more introduced species in San Francisco Bay. Bay populations
are likely to be referable to one or more of the common Cliona
found on oysters in Atlantic estuaries; these include Cliona
celata Grant, 1826 and Cliona lobata Hancock, 1849
(Carlton, 1979a, p. 218). Japanese species (or genomes) may also
be present. Atlantic Cliona were introduced with Atlantic
oysters. The first record is that of Townsend (1893), who observed
that in 1891 large numbers of oyster shells in the Bay "were
found honeycombed by the boring sponge."
Halichondria bowerbanki Burton, 1930
BOWERBANK'S HALICHONDRIA
SYNONYMS: Halichondria coalita
This Atlantic sponge, known from both Europe and Atlantic America,
was reported from the Pacific in San Francisco Bay in the early
1950s (Carlton, 1979a), and later from other sites including Humboldt
Bay (S. Larned, pers. comm., 1989) and Coos Bay (Hewitt, 1993).
It was either introduced with Atlantic oysters, with which it
occurs (pers. obs.) or as a fouling organism. In 1993-94 we found
Halichondria on most floating docks and with other fouling
in the South, Central and San Pablo bays, though not on docks
near the Golden Gate.
Haliclona loosanoffi Hartman, 1958
LOOSANOFF'S HALICLONA
SYNONYMS: Haliclona sp. B of Hartman, 1975
Haliclona ecbasis de Laubenfels, 1930
We newly follow and extend Van Soest (1976) in designating San Francisco Bay Haliclona as the Atlantic native Haliclona loosanoffi (although the recognition of this species in the Bay does not preclude more than one species being present). This is a common tan, yellow, and orange sponge of Bay fouling communities. This is the same species referred to as Haliclona sp. B by Hartman (1975), and is also the same species reported by Fell (1970) as Haliclona ecbasis from Berkeley Yacht Harbor, St. Francis Yacht Harbor, Redwood City and Carmel. Van Soest (1976) noted that Fell's (1970) description of H. ecbasis was very close to H. loosanoffi in all characters, including details of the life cycle, but came short of designating the Bay population as the Atlantic species solely because it was in the Pacific Ocean (Van Soest not considering the possibility that it was introduced). Haliclona, possibly including this species, have been reported from Puget Sound, Coos Bay, Bodega Harbor, and several bays in southern California (Carlton, 1979a, p. 216).
Haliclona loosanoffi is a common species of oyster communities on the New England coast (pers. obs.), and may have been introduced to the Bay with Atlantic oysters, although the earliest records are only from 1950 (Hartman, pers. comm., 1977). Its presence in fouling communities, however, means that it may have been introduced by ships as well.
In 1993 we found Haliclona on most floating docks in the
Central Bay and the seaward parts of South and San Pablo bays.
We did not find it in 1994 and 1995.
Microciona prolifera (Ellis and Solander, 1786)
RED BEARD SPONGE
This large, common Atlantic sponge is known from Canada to South Carolina. It was first found in San Francisco Bay in the mid- to late-1940s by Woody Williams (it was not noted by Light, 1941), who showed photographs to M. W. de Laubenfels (who initially identified it as the native Microciona microjoanna; Hartman, pers. comm., 1977). W. Hartman (pers. comm., 1977) found large colonies at Redwood City in 1950, and transplanted some of these for experimental purposes to Berkeley Yacht Harbor where it subsequently became established. Its bright orange-red finger-like colonies are unmistakable in the fouling communities around much of the Bay. In 1993-95 we observed it on several floating docks in the South Bay, the eastern shore of the Central Bay, and the southern part of San Pablo Bay.
Only two other populations are known on the Pacific coast, from Willapa Bay (Carlton, 1979a, p. 215) and Humboldt Bay (S. Larned, pers. comm., 1989).
Microciona could have been a late introduction with Atlantic
oystersóalong with the crab Rhithropanopeus harrisii
and the whelk Busycotypus canaliculatus which were first
found in San Francisco Bay at about this time, Microciona has
been collected from Atlantic oyster beds (Wells, 1961; Maurer
& Watling, 1973). Since it is a common fouling organism (ANC
& JTC, pers. obs.), it could also have been introduced in
ship fouling.
Prosuberites sp.
This undescribed American Atlantic sponge (Hartman, pers. comm.,
1977) was first collected in the Bay in 1953 on Angel Island (Carlton,
1979a, p. 217). It may have been introduced to San Francisco Bay
with Atlantic oysters or in ship fouling.
Numerous species of hydroids have been introduced to the Bay since
the Gold Rush. We treat 13 species here. Campanularia gelatinosa
and Halocordyle disticha (=Pennaria tiarella) may
still be present in the Bay, but there are no recent records,
and we thus list them in Appendix 2.
Blackfordia virginica Mayer, 1910
This Sarmatic hydroid, native to the Black and Caspian Seas, was first collected in 1970 in the Napa River and again in 1974 in the Petaluma River. It remained misidentified (as a species of Phialidium) until 1993 (Mills & Sommer, 1995), when we collected medusae in both rivers. In San Francisco Bay Blackfordia jellyfish eat copepods, copepod nauplii, and barnacle nauplii (Mills & Sommer, 1995).
Blackfordia may have been introduced in ships' fouling
or in ships' ballast water. The presence of widely scattered populations
in the Atlantic Ocean (Chesapeake Bay, Brazil, France, and Portugal)
and in India and China means that the source of the Bay's population
is unknown, although it is possible that if other populations
have diverged genetically, candidate source regions could be identified.
The introduction into the Bay in the 1980s-1990s of the clams
Potamocorbula and Theora, the mitten crab Eriocheir,
seven species of copepods, and other crustaceans, all from Asia,
might suggest a Chinese origin. Indeed, it is possible that the
recent populations of Blackfordia in the Bay represent
a reintroduction of the species.
Cladonema uchidai Hirai, 1958
This Japanese hydroid was first collected in San Francisco Bay in 1979 (Rees, 1982), although the polyps and medusae that have been studied to date have originated from laboratory or home aquaria containing fouling organisms from San Francisco Bay. The polyps in the laboratory were small (0.5 mm height) as were the medusae (3.5 mm height), and little remains known of this hydrozoan in the Bay.
Introduction with ship fouling or ballast water is possible, although
earlier introduction with Japanese oysters may have occurred if
Cladonema's habitat in Honshu includes oyster communities.
Clava multicornis (Forskaal, 1775)
CLUB HYDROID
SYNONYMS: Clava leptostyla Agassiz, 1862 of northeastern
Pacific authors; see Austin, 1984
Rees and Hand (1975) noted that this northwestern Atlantic hydroid
forms "large pink patches on pilings in estuaries."
It was first collected in the Bay in 1895 (Carlton, 1979b, p.
229), no doubt originating from ship introductions from the New
England coast, where it is common. Fraser (1937) described its
widespread distribution throughout the Bay as documented by Albatross
collections in 1912-13.
Cordylophora caspia (Pallas, 1771)
FRESHWATER HYDROID
SYNONYMS: Cordylophora lacustris Allman, 1844
This brackish and freshwater Sarmatic hydroid, native to the Caspian
and Black Sea regions, was first found in the Bay in the San Joaquin
River at Antioch. Specimens discovered in 1950 were considered
to have been collected "20 to 40 years" previously (Hand
& Gwilliam, 1951); we choose a date of 1930 as a first record.
It was also collected at a similarly early but uncertain date
from Lake Union in Seattle, and has now been reported from several
sites between San Francisco Bay and Vancouver Island, British
Columbia (Carlton, 1979a, p. 230). It is sufficiently widespread
around the world (Hand & Gwilliam, 1951), a distribution perhaps
achieved centuries ago, as to make the origin of the Bay's populations
unknown. It was likely introduced in ship fouling (WHOI, 1952)
or ballast water. Cordylophora is common in the Delta (Hazel
& Kelly, 1966) and on the concrete sides of the Delta-Mendota
water delivery canal (Eng, 1979), and has also been collected
in San Francisco's Lake Merced (Miller, 1958).
Corymorpha sp.
This tiny estuarine, orange-tinted hydroid was collected from
soft mud bottoms on the eastern shore of the Bay at Point Richmond
(1955-56) and in Oakland's Lake Merritt (1967) (Carlton, 1979a).
It appears similar to the European Corymorpha nutans M.
Sars, 1835, but the species-level taxonomy remains unresolved
(C. Hand, pers. comm., 1967). No similar hydroid has been reported
from elsewhere on the Pacific coast. In Lake Merritt it occurs
in samples otherwise composed entirely of introduced species.
This facies, the absence of any similar Pacific taxon, and its
similarity to an Atlantic species, leads us to consider it to
be introduced, either via oyster shipments, ship fouling or ballast
water.
Garveia franciscana (Torrey, 1902)
SYNONYMS: Bimeria franciscana
This hydroid, often considered under the genus Bimeria, is common in the Bay and reported to be one of the primary food sources of the introduced Asian isopod Synidotea laevidorsalis (Carlton, 1979a). Possibly native to northern Indian Ocean estuaries, it has been introduced in ship fouling and, in later years, possibly by ballast water, to many harbors and ports around the world. It has been reported from western Africa, northwestern Europe, eastern North America, the Gulf of Mexico and Australia (Carlton, 1979a, p. 225).
Garveia was first collected by Torrey in 1901 (Torrey,
1902; Vervoort, 1964) in San Francisco Bay, its only confirmed
location on the Pacific coast. In 1993-95 we found it in dense
masses under floating docks at some sites in San Pablo Bay, coated
with the introduced bryozoan Conopeum tenuissimum and crawling
with Synidotea. We consider it a ship fouling introduction.
Gonothyraea clarki (Marktanner-Turneretscher, 1895)
This well-known North Atlantic fouling hydroid was first collected in San Francisco Bay in "Oakland Creek" in 1895 and again at various stations around the Bay by the Albatross in 1912 (both are unpublished NMNH records). Graham & Gay (1945) recorded it again in from the Oakland Estuary based upon their 1940-42 studies. Rees & Hand (1975) note that it is "often very common on harbor floats" in central California. In 1995 we collected it from floats at the Grand Street (Oakland Estuary), Emeryville and Coyote Point marinas in San Francisco Bay, and from Isthmus Slough in Coos Bay. Since Gonothyraea can be clearly distinguished from Obelia only if gonozoids are present (E. Kozloff, pers. comm., 1995), some Pacific coast records of Obelia may actually refer to Gonothyraea. Gonothyraea species have been reported from ship fouling (WHOI, 1952), and it was likely introduced either in fouling or with oysters.
Maeotias inexspectata Ostroumoff, 1896
Another Black Sea native, Maeotias was first found in the turning basin of the Petaluma River in 1992, and became sufficiently abundant by the summer of 1993 to attract public attention (Mills & Sommer, 1995). Outside of the Black Sea it was previously known from two regions on the Atlantic American coast (Chesapeake Bay and South Carolina) and France (Mills & Sommer,1995); the source of the Bay populations is as yet unknown. In the Petaluma River these jellyfish eat primarily barnacle nauplii, copepods, zoea larvae of the introduced Atlantic crab Rhithropanopeus harrisii, tanaids and other invertebrates, and in the laboratory tolerated salinities up to 13 ppt (Mills & Sommer, 1995).
Mills & Sommer (1995) concluded that the Maeotias population
in the Petaluma River appears to have been introduced as polyps
rather than medusae, since the medusae population in the River
is entirely male and therefore incapable of reproduction. A polyp
isolated from the Maeotias population, however, readily
reproduced asexually in the laboratory, creating numerous new
polyps which then produced male medusae. Both polyps (both unattached
and on floating debris) and medusae of hydroids are known from
ballast water, making this or ship fouling the probable means
of introduction.
Obelia ?dichotoma (Linnaeus, 1758) and Obelia ?bidentata
Clark, 1876
We consider these two species of Obelia, described from Europe and New England respectively, as introduced, and provisionally use the names adopted by Cornelius (1975). Obelia dichotoma was collected in 1894 and later years (identified as O. commissuralis) and in 1899 and later years (identified as O. longissima) from the Bay (unpublished NMNH records). Obelia bidentata was collected in the Bay in 1912 (identified as O. bicuspidata) (Fraser, 1925, and unpublished NMNH records). Obelia spp. occur throughout the Bay's fouling communities, although in relatively low numbers.
Kofoid (1915) early on referred to the "contamination" of Pacific coast harbors by ship-introduced "tubularian and campanularian hydroids." Obelia species have frequently been reported from ship fouling (WHOI, 1952), and there is little doubt that Obelia from around the world were a common element of ships' fouling communities brought to the Bay from the Gold Rush era on. Obelia may have commenced its world journeys on ship bottoms in the 13th century, making identification of original source regions difficult. Obelia has no doubt been introduced into the Bay continuously over the years in ship fouling, with commercial oysters both from the Atlantic (where it occurs in oyster beds; Wells, 1961; Maurer & Watling, 1973) and from Japan, and in recent times in ships' ballast water, primarily as hydromedusae.
The native nudibranch Doto kya and the introduced nudibranchs
Eubranchus misakensis and Tenellia adspersa apparently
feed upon Obelia in San Francisco Bay (Behrens, 1971, 1991;
Carlton, 1979a; Jaeckle, 1983).
Sarsia tubulosa (M. Sars, 1835)
SYNONYMS: Syncoryne mirabilis (Agassiz, 1849)
Coryne rosaria Agassiz, 1865
Redescribed from San Francisco Bay as Coryne rosaria by
Alexander Agassiz in 1865, Sarsia was one of several North
Atlantic hydroids collected by Agassiz during his visits to the
Pacific Coast in the late 1850s. He collected this hydroid at
Vancouver Island, British Columbia and in the San Juan Islands,
Washington, in 1859, and from San Francisco Bay in 1860 (Carlton,
1979a, p. 233). Ricketts & Calvin (1939), in a rare reference
to such matters, took particular note of this hydroid as a possible
"relic of the days of wooden ships;" we agree that introduction
as a ship-fouling organism is the probable means of dispersal.
It has subsequently been recorded from Alaska to southern California,
although aspects of its global distribution suggest that more
than one species may be involved.
Tubularia crocea (Agassiz, 1862)
SYNONYMS: Parypha microcephala Agassiz, 1865
Tubularia elegans Clark, 1876
Petersen (1990) proposes that Tubularia crocea be transferred
to the genus Ectopleura.
This common Atlantic fouling hydroid, known from Newfoundland to Florida and the Gulf of Mexico and frequently reported from ships' fouling communities (WHOI, 1952), was introduced by Gold Rush ships to the Bay. It was first collected in 1859 by Alexander Agassiz (who mistakenly described it as a new species, Parypha microcephala; Carlton, 1979a, p. 238) "attached to floating logs round the wharves of San Francisco." It has since been collected from the Gulf of Alaska to San Diego.
Tubularia crocea has been frequently reported from ships'
fouling communities, although some later introductions may have
occurred with Atlantic oysters, with which it occurs on the Atlantic
coast (Wells, 1961; Maurer & Watling, 1973). The introduced
nudibranchs Catriona rickettsi, Sakuraeolis enosimensis
and Tenellia adspersa reportedly feed upon Tubularia
in San Francisco Bay (Carlton, 1979a; Behrens, 1984, 1991).
Aurelia "aurita (Linnaeus, 1758)"ónorthwestern
Pacific stock
MOON JELLY
SYNONYMS: Aurelia labiata
Greenberg (1995) reports that a sometimes dense population of Aurelia aurita in Foster City Lagoon (on the San Mateo side of the South Bay), present since at least around 1989, is genetically similar (based on allozyme comparisons) to Aurelia from Tokyo Bay, Japan and unlike Aurelia from Monterey Bay and Vancouver Island. Differences in the structure of the radial canal further distinguish the Japanese and San Francisco Bay from the northeastern Pacific stocks. Aurelia has been seasonally abundant in recent years in Foster City Lagoon and Redwood Creek, both on the southwestern shore of San Francisco Bay (J. Thompson, pers. comm.). We know of no earlier reports of Aurelia in South Bay lagoons, although there are records of swarms in Tomales Bay (Ricketts et al., 1985; T. Gosliner, pers. comm., 1995) of this species which is normally found offshore in central California latitudes (Ricketts et al., 1985; E. Kozloff, pers. comm., 1995).
The San Francisco Bay population may have been introduced as larvae (known as ephyrae) in ballast water, since we have found live scyphozoan ephyrae in the ballast water of freighters arriving at Coos Bay, Oregon from Japan. Ricketts et al. (1985) describe Aurelia polyps as "extraordinarily tough and resistant," so transport across the Pacific as ship fouling would also be possible.
As Aurelia aurita was first described from North Atlantic
waters, and since there is evidence of both genetic and morphological
differentiation, the species-level taxonomy of the group may require
revision.
Diadumene ?cincta Stephenson, 1925
ORANGE ANEMONE
Between the mid-1950s (Hand, 1956) and early 1970s when it was first collected (no exact date is available as of this writing), a fourth species of Diadumene was introduced into San Francisco Bay (Carlton, 1979a). Its morphology and distribution in the Bay were extensively studied by T. Blanchard, whose work and taxonomic conclusions remain unpublished, but who felt that there was a "strong case for conspecificity" with the European (primarily British) Diadumene cincta. We tentatively use that name for this anemone, to which it is morphologically very similar. Diadumene cincta occurs in Britain both on open marine shores and in estuaries, tidal creeks, and harbors (Manuel, 1981). Blanchard also found the same species in Humboldt Bay (T. Blanchard, pers. comm., 1988).
Blanchard (pers. comm., 1988) has provided the following information about this anemone in San Francisco Bay. Diadumene ?cincta has a column diameter of about 15-20 mm and a column height of up to five or more times the width. The most common variety of Diadumene ?cincta on dock floats is solid orange, but pink forms also occur, most commonly sublittorally on pilings and in the mid to low intertidal zone in protected locations. Specimens also occur sublittorally on shells partially buried in sediment. White markings on the oral disk are common on the pink forms, but have not been observed on orange specimens. The anemone commonly forms clonal aggregations of up to 200 individuals in fouling, a character typical of the European D. cincta (Manuel, 1981); it may also occur singly. As this anemone is not described in Hand (1975) nor in other guides to Pacific coast marine life, it may be mistaken for Diadumene leucolena or stripeless Haliplanella lineata.
We tentatively assign an Atlantic origin to this species. It was probably introduced either in ship fouling or ballast water.
Diadumene franciscana Hand, 1956
SAN FRANCISCO ANEMONE
This usually white-striped introduced anemone of unknown origin has been reported from San Francisco Bay (before 1941), Morro Bay (1973) (Carlton, 1979a, p. 250) and Mission Bay (1977-78) (Dygert, 1981), and we collected it in Tomales Bay in 1995 (identified by C. Hand). Carlton (1979a) suggested that it may originate from the southern Pacific or Indian 0ceans, rather than from the Atlantic, where the anemone fauna is better known. As the anemone fauna of Japan is also relatively well studied, oyster transplantation from either the Atlantic or from Japan is not the likely mechanism of introduction. As it is a common float and piling fouling organism locally in San Francisco Bay, it may have been introduced as hull fouling, or else in ballast water. Diadumene franciscana can be very common in the warm margins of the Bay where other species, such as the tubeworm Ficopomatus enigmaticus and the barnacle Balanus amphitrite amphitrite of known warm-water origin are also common. Its presence in warm-water thermal effluents in Morro Bay (to where it was likely introduced from San Francisco Bay) is also suggestive of a warm temperate or subtropical origin.
The first record of this anemone is that of Light (1941, as a
"double-striped anemone" from Fruitvale Bridge), whose
records were based upon his field observations made in the Bay
since the 1920s.
Diadumene leucolena (Verrill, 1866)
WHITE ANEMONE
This Atlantic anemone, occurring from at least Cape Cod to South Carolina, was first reported from the Oakland Estuary by Sander (1936), although it may have been present in the Bay since the 19th century. Hand (1956) described it in detail from the Bay. It is common to abundant along the Bay margin, in fouling communities, under rocks, and on oyster shells, and may have been introduced with oyster shipments (it is recorded from Atlantic coast oyster beds; Wells, 1961), as ship fouling or in ballast water. It has also been reported from southern California bays and from Coos Bay, Oregon (Carlton, 1979a, p. 248).
Diadumene lineata (Verrill, 1873)
ORANGE-STRIPED GREEN ANEMONE
SYNONYMS: Haliplanella lineata
Haliplanella luciae (Verrill, 1898)
Diadumene luciae
Aiptasiomorpha luciae
This abundant, often orange-striped anemone, known in most literature as Haliplanella luciae (Verrill, 1898), was first collected in San Francisco Bay in 1906 (Davis, 1919), and has since been collected from bays and harbors from Newport Bay to British Columbia (Carlton, 1979a, p. 253). It is now one of the most common anemones along the margins of San Francisco Bay, occurring in habitats ranging from fouling communities to bits of shell on open mudflats to brackish marsh channels. A native of Japan, it has been widely dispersed around the world by both shipping and by the movement of commercial oysters, either or both of which mechanisms could have brought it to the Bay. That it may have arrived with the large volumes of Atlantic oysters brought to the Bay in the 1890s is suggested by its late appearance in New England (1892; Verrill, 1898) and its presence in Atlantic coast oyster beds (Wells, 1961; Maurer & Watling, 1973), and it may thus be another example of the many species whose arrival in one region (in this case San Francisco Bay) was contingent upon its introduction to another region (New England) thus interfacing with an ongoing transport vector and dispersal corridor (the commercial oyster industry).
Haliplanella has the ability, perhaps unique among the
anemones, to encyst, leaving behind upon excystment a tough capsule
(Kiener, 1972). This remarkable characteristic has likely conferred
upon Haliplanella an unusual ability to survive long-distance
transport under severe conditions (Carlton, 1979a). The introduced
nudibranch Cuthona perca feeds upon Haliplanella
in the Bay (McDonald, 1975; Carlton, 1979a).
Of all the common macroinvertebrates in San Francisco Bay, the oligochaetes are perhaps the poorest known relative to the comparative diversity of native versus introduced species. We recognize here eight introduced oligochaetes and list four others as cryptogenic (Chapter 4), although the latter are frequently abundant and embedded in communities otherwise composed of non-native species. Annelid taxonomy is widely recognized as a difficult and complex field; and although we know relatively little about the Bay's polychaetes, we know even less about its oligochaetes.
Each of the following species of oligochaetes could have been
present in San Francisco Bay for many decades, if not since the
19th century, before they were first collected in the 1950s and
1960s. We thus regard the dates of first collection of most of
the following species as artifacts of the collecting effort. The
decades- to century-long uncertainty in the actual dates of introduction
makes it hard to determine transport mechanisms. We generally
consider ships' solid ballast and water ballast, shipments of
commercial oysters, and shipments of aquatic plants to be possible
vectors.
Branchiura sowerbyi Beddard, 1892 [TUBIFICIDAE]
This oligochaete, native to tropical and subtropical Asia (India, Myanmar (Burma), Java, China, Japan), was first collected in 1892 from the mud of the Victoria regia tank in the garden of the Royal Botanic Society in Regent's Park, London. Over the next 30 years it was collected from other warm-water tanks in botanic gardens at Hamburg, Dublin, Kew and Oxford. By the late 1950s it had been found "in the wild" in the Rhone River and elsewhere in southern France, in the Thames River below Reading in water warmed by effluent from a power station, and in unheated waters in the Kennet and Avon Canal and in the Bradford River Avon in England (Mann, 1958). It has also been reported from north and west Africa (Brinkhurst, 1965).
It was first collected in North America in central Ohio in 1930
(Spencer, 1932), and spread to the Great Lakes by 1951 (Mills
et al., 1993) and to a total of eighteen states by 1966 (Brinkhurst,
1965; Cole, 1966). In California it was collected from the San
Joaquin River in 1950, from the Tuolomne River near Modesto in
1952 (Brinkhurst, 1965), and from the Delta in 1963 (specimen
at CASIZ). The California Department of Water Resources has collected
it throughout most of the Delta since sampling started in 1977
(from the western Delta upstream to the Mokelumne River, Courtland
on the Sacramento River, and Stockton on the San Joaquin River),
at densities of up to 823/m2 (Markmann, 1986;
DWR, 1995). We found no other records of Branchiura on
the Pacific coast. Branchiura could have been transported
to California in ships' solid or water ballast or on ornamental
aquatic plants.
Limnodrilus monothecus (Cook, 1974) [TUBIFICIDAE]
Although first described from Bahia de San Quintin, Baja California
based upon specimens collected in 1960 (Cook, 1974), Erseus (1982)
demonstrated that this marine and estuarine species is widely
distributed from the mid-Atlantic coast to the Gulf of Mexico,
and was only found in three stations in British Columbia, southern
California, and Bahia de San Quintin on the Pacific coast. Nichols
& Thompson (1985) record it from their south San Francisco
Bay mudflat stations, where they treated it as cryptogenic. It
appears, however, to be an Atlantic species introduced to west
coast estuaries. It could have arrived in ships' solid or water
ballast or in shipments of commercial oysters.
Paranais frici Hrabe, 1941 [NAIDIDAE]
Brinkhurst & Cook (1980) regard the fresh and brackish water
P. frici as a European (Sarmatic) species introduced into
North America. Brinkhurst & Simmons (1968) found it to be
one of two abundant oligochaetes in Suisun Bay in 1961-62. It
was collected in the eastern Delta (Mokelumne River) in 1977-79,
and in the western and central Delta in 1980-95, at concentrations
up to 1,296/m2. Brinkhurst & Coates (1985)
also report it from Newport Bay, California and Fraser River,
British Columbia, and note that it has been further reported from
Africa and South America. It could have arrived in California
in ships' solid or water ballast or on ornamental aquatic plants.
Potamothrix bavaricus (Oschman, 1913) [TUBIFICIDAE]
This freshwater Eurasian species was regarded as "possibly"
introduced to eastern North America by Brinkhurst (1965), who
further recorded a population (collected by R. Whitsel, no date
given) from Coyote Creek, in Santa Clara County. We tentatively
regard it as introduced, if the identification is correct. It
has been reported from the central and western Delta since 1991,
at concentrations up to 415/m2 (DWR, 1995).
It could have arrived in California in ships' solid or water ballast
or on ornamental aquatic plants.
Tubificoides apectinatus (Brinkhurst, 1965) [TUBIFICIDAE]
This common North Atlantic coast marine oligochaete (Brinkhurst,
1981, 1985) was found to be abundant in South San Francisco Bay
sediments in 1961-62 collections (Brinkhurst & Simmons, 1968,
as Peloscolex apectinatus). It could have arrived in ships'
solid or water ballast or in shipments of commercial oysters.
Tubificoides brownae Brinkhurst & Baker, 1979 [TUBIFICIDAE]
SYNONYMS: Peloscolex gabriellae of authors
This North Atlantic marine oligochaete (described from Delaware, and known from other Atlantic coastal sites as well as Europe) was treated by Brinkhurst & Simmons (1968) as Peloscolex gabriellae (in part), from the South Bay (Brinkhurst, 1986). It is also known from Coos Bay, Oregon (Brinkhurst, 1986). Nichols & Thompson (1985) reported it as a cryptogenic member of the South San Francisco Bay mudflat community. We regard it is as introduced based upon its broad Atlantic distribution and its apparently restricted distribution in the Pacific Ocean. It could have arrived in California in ships' solid or water ballast or in shipments of commercial oysters.
Brinkhurst & Simmons (1968) examined specimens collected in
1961-62. Brinkhurst (1965), under the name Peloscolex gabriellae,
records material from 1957 (collected by M. Jones) from Point
Richmond, but it is not clear if these specimens are referable
to T. brownae or to T. wasselli (below). The California
Department of Water Resources reports T. brownae collected
in small numbers from Grizzly Bay and Pt. Pinole since 1987 (DWR,
1995).
Tubificoides wasselli Brinkhurst & Baker, 1979 [TUBIFICIDAE]
This Atlantic marine tubificid is known from Delaware to the Gulf
of Mexico (Brinkhurst, 1986). San Francisco Bay populations collected
in 1961-62 and identified by Brinkhurst & Simmons (1968) as
a papillate form of Peloscolex gabriellae are now considered
to be this species (Brinkhurst, 1986). It is otherwise known from
Victoria, British Columbia (Brinkhurst, 1986). It could have arrived
in California in ships' solid or water ballast or in shipments
of commercial oysters.
Varichaetadrilus angustipenis (Brinkhurst & Cook, 1966)
[TUBIFICIDAE]
SYNONYMS: Limnodrilus angustipenis
This eastern United States species (Brinkhurst, 1971; Strayer, 1990; Erseus et al., 1990) occurs widely in the Sacramento-San Joaquin Delta in freshwater muddy sediments. It was collected by the California Department of Water Resources at least as early as 1982 in stations near the western end of Sherman Island. Hymanson et al. (1994) reported that it was one of the numerically dominant species at these sites from 1982-86, concluding that it and Limnodrilus hoffmeisteri (here treated as cryptogenic) "are among the few native benthic organisms that have maintained their numerical dominance and broad distribution..."
V. angustipenis could have arrived on the Pacific coast
in ballast water or on ornamental aquatic plants.
Boccardiella ligerica (Ferronnière, 1898) [SPIONIDAE]
SYNONYMS: Boccardia ligerica Ferronnière, 1898
Boccardia nr. uncata
Polydora uncata
Polydora redeki Horst
This spionid worm is native to the brackish waters and mudflats of France, Holland and Germany. A single specimen identified as Boccardiella ligerica was collected from Newport Bay in 1935 (Kudenov, 1983). B. ligerica was collected from San Francisco Bay in the San Pablo Channel by 1954 and from the Delta-Mendota Canal, in fresh water, in 1973 (Light, 1977; Carlton, 1979a, p. 305). It was also collected from freshwater in the New River and the Alamo River in Imperial County in southeastern California in 1979, and from a canal in Mar Chiquita, Argentina with the Australian serpulid worm Ficopomatus enigmaticus (Kudenov, 1983).
Boccardiella ligerica may have been introduced with ships' ballast water, perhaps during World War II or the Korean War. Spionid larvae are among the most abundant and frequently encountered groups of organisms in ballast water (Carlton & Geller, 1993).
B. ligerica was one of the most common benthic organisms
collected by CDFG near Martinez in 1975-1981, and was found upstream
as far as Collinsville in the western Delta (Markman, 1986). In
1976, a dry year, Siegfried et al. (1980) found B. ligerica
to be a dominant species at their upstream stations near Collinsville
in the late summer and fall, with peak densities of around 20,000
individuals/m2, and Markman (1986) similarly
reported an increase in B. ligerica upstream in the dry
year of 1981. Light (1978, p. 201) summarizing recent studies
showed B. ligerica collected only from the ends of the
Bay: at the southern end of the South Bay and from Martinez to
the Antioch bridge in the northern Bay.
Ficopomatus enigmaticus (Fauvel, 1923) [SERPULIDAE]
AUSTRALIAN TUBEWORM
SYNONYMS: Mercierella enigmatica
Ficopomatus enigmaticus is an Australian worm that builds and lives in a white, calcareous tube, the tubes forming large agglomerate masses when the worm is abundant. Reported from ships' hulls (WHOI, 1952) and probably transported as hull fouling, it has become established in many parts of the world including the Black, Caspian and Mediterranean seas, northern Europe, Uruguay, Argentina, Hawaii, Japan and the Gulf of Mexico. It was first reported in San Francisco Bay from Lake Merritt, a tidal lagoon on the East Bay shore, in a 1921 article in the Oakland Tribune headlined "Coral Reefs Spreading in Lake Merritt." The "reefs" had been first noticed by park officials about a year earlier.
It was also in 1921 that F. enigmaticus was discovered and described in France, and discovered at the London docks (Carlton, 1979a). F. enigmaticus apparently requires water temperatures of at least 18°C to breed (Obenat & Pezzani, 1994), and in Europe it frequently lives in water heated by the cooling water effluent from power plants (Vaas; 1978). In the Netherlands its colonies have interfered with lock operations (Vaas; 1978).
F. enigmaticus has been collected from many sites in the South, Central and San Pablo bays, sometimes in dense masses, especially from enclosed lagoons or protected waters. These sites include Aquatic Park Lagoon in Berkeley (first appeared between 1942 and 1946, and still abundant), Alameda Lagoons (abundant in 1971, scarce in the 1990s), Berkeley Yacht Harbor (1969), San Rafael and Corte Madera Creek (1970), Palo Alto Yacht Harbor and China Camp (1974), Foster City Lagoons and Belvedere Lagoons (before 1979), and the Petaluma River Turning Basin (abundant in 1993; see Carlton, 1979a, p. 331, for references on the other records). It is less abundant now in Lake Merritt than it was in the 1920s and the 1960s-70s.
Newman's (1963) report of a serpulid worm "comparable to
Mercierella enigmatica" in the seawater system of
a naval vessel docked in San Francisco Bay suggests that it may
have been introduced more than once.
Heteromastus filiformis (Claparede, 1864) [CAPITELLIDAE]
Heteromastus filiformis is native to the Atlantic coast of the United States from New England to the Gulf of Mexico, and has also been reported from Greenland, Sweden, the Mediterranean, Morocco, South Africa, the Persian Gulf, New Zealand, Japan, and the Bering and Chukchi Seas. The wide temperature range covered by these locations suggests that more than one species may be involved. In California Heteromastus was collected from San Francisco Bay in 1936, from Morro Bay in 1960, possibly from southern California by 1961, and from Bolinas Lagoon by 1969. It was collected from Vancouver Island in 1962, from Coos Bay, Oregon in 1970 (pers. obs.), and from Grays Harbor, Washington by 1977 (Carlton, 1979a, p. 322).
As with other polychaetes first collected on the Pacific Coast in the 1930s by Olga Hartman (including Polydora ligni and Streblospio benedicti in San Francisco Bay), Heteromastus filiformis may have been present but undetected for many decades due to the lack of earlier investigations of intertidal polychaetes on this coast. Thus this mud-dwelling capitellid worm may have been introduced to San Francisco Bay in the late nineteenth or early twentieth century with Atlantic oysters, (with which it occurs; Wells, 1961), or may have been an early ballast water introduction.
Heteromastus filiformis is commonly collected from the
far South Bay to the western half of Suisun Bay at concentrations
of 10 to 4000 per square meter, and has been collected upstream
to Pittsburg (Hopkins, 1986; Markmann, 1986). It is one of the
most common benthic organisms in the shallows of San Pablo Bay
and the channels of the South Bay (Nichols & Thompson, 1985a).
Manayunkia speciosa Leidy, 1858 [SABELLIDAE]
SYNONYMS: Manayunkia eriensis (Krecker, 1939)
Manayunkia speciosa is a freshwater polychaete native to eastern North America from the westernmost Great Lakes, New York and Lake Champlain in Vermont south to the Savannah River in South Carolina (Klemm, 1985). It was collected from two small, shallow lakes in northern Alaska in 1961 and 1964, and from Sevenmile Canal in Klamath County, Oregon in 1964 (Hazel, 1966; Holmquist, 1967; Croskery, 1978). It was first collected in California from the Mokelumne River near New Hope Landing in the eastern Delta in 1963 (Hazel, 1966). Hartman's (1969) report of this species from San Pablo and Suisun bays appears to be based on a misreading of earlier reports.
This tube-dwelling, colonial worm has neither a resting stage nor a planktonic or swimming stage that might aid dispersal or transport in wateróyoung worms mature within the parental tube and emerge as small, crawling adults to build tubes nearby (Holmquist, 1967; Croskery, 1978). However, transport in detritus carried in water may be possible. Hazel (1966) suggested that M. speciosa arrived in the Delta in the water in which freshwater gamefish from the eastern United States were transported. Hazel (1966), citing Smith (1896), noted as pertinent the fact that white catfish Ictalurus (now Ameiurus) catus introduced to the Delta in 1874 were taken from the Schuylkill River, Pennsylvania, the type locality for M. speciosa. However, although Smith (1896) describes these as "white catfish or Schuylkill catfish," he clearly states that the fish transported to California were taken from the Raritan River, New Jersey. Thus "Schuylkill" appears to be part of a common name for these fish, rather than the site from which they were collected.
Although most or all of the freshwater fish introduced to California from the northeastern United States appear to have been planted in the late nineteenth or early twentieth century (Table 1) and Manayunkia was not discovered in California until 1963, it is possible that this small polychaete was present and overlooked for a long time (Holmquist, 1967; Mackie & Qadri, 1971). Alternatively, it may have been transported in detritus floating in freshwater ballast.
Manayunkia is the fourth most numerous benthic invertebrate
collected by the California Department of Water Resources in the
Delta, with densities in the interior of the Delta of 2,000 to
50,000 individuals/m2. It apparently requires
fresh water and silty substrates, and is found in the eastern
portions of the Delta downstream to Frank's Tract and Rio Vista,
with questionable records from a few stations further downstream
(Markmann, 1986; Herbold & Moyle, 1989; Hymanson et al., 1994).
Marenzelleria viridis (Verrill, 1873) [SPIONIDAE]
SYNONYMS: Scolecolepis viridis
Scolecolepis tenuis
Scolecolepides viridis
Marenzelleria viridis is native to the northwestern Atlantic and was collected in Germany in 1983, probably having been introduced via ballast water (Essink & Kleef, 1993). It spread though western and northern Europe and into the Baltic Sea, where it is now extremely abundant. It was first collected on the Pacific coast in Nov. 1991 at Collinsville on the Sacramento River, at which station it has been found most consistently and abundantly at up to 1700 worms/m2. It has since been collected from Frank's Tract and the Old River in the Delta downstream to Grizzly Bay in 1992, in San Pablo Bay in 1995, and in the far South Bay (M. Kellogg, pers. comm., 1995; W. Fields, pers. comm., 1995; DWR, 1995). It probably arrived in ballast water.
Marphysa sanguinea (Montagu, 1815) [EUNICIDAE]
Marphysa sanguinea is regarded as a single cosmopolitan species, but likely consists of several difficult-to-distinguish but distinct taxa. It is reported from Europe (from Great Britain to the Mediterranean), the western Atlantic (Massachusetts to the West Indies, the Gulf of Mexico, Bermuda and the Bahamas), Japan, China, and from Australasia to the Red Sea and Africa. In the eastern Pacific it has been known from San Francisco Bay since 1969, and from various sites between Los Angeles and Panama (Carlton, 1979a, p. 302). The San Francisco Bay population may have been introduced from the Atlantic with shipments of oysters, with which it occurs on the Atlantic coast (Wells, 1961), or it may have been introduced in ballast water.
Hopkins (1969) reported M. sanguinea as common at concentrations
of 10-200 per square meter, but found only in the South Bay south
of Hunters Point, and most commonly in the channels.
Nereis succinea (Frey & Leuckart, 1847) [NEREIDAE]
PILE WORM
SYNONYMS: Neanthes succinea
Nereis saltoni Hartman, 1936
Nereis limbata Webster, 1879
This euryhaline "pile worm" lives in a variety of habitats: under rocks, in mud and sand, in oyster beds and in fouling communities. It is reported from locations around the world, including the eastern Atlantic and the Mediterranean; the western Atlantic from the Gulf of St. Lawrence to the West Indies, Gulf of Mexico and South America; West Africa and South Africa; and the tropical eastern Pacific from the Gulf of California to Colombia (Carlton, 1979a, p. 295). These reports may involve a single species transported synanthropically about the globe, or multiple, closely-related species.
In California it has been collected from San Francisco Bay (earliest records from 1896), the Salton Sea (from 1935), Tomales Bay (1941), several southern California bays (from 1952), and in Oregon from Netarts Bay (1976) (Carlton, 1979a) and Coos Bay (1986; pers. obs.). The San Francisco Bay population probably originated in the western North Atlantic and arrived in shipments of Atlantic oysters (with which it occurs on the Atlantic coast; Wells, 1961; Maurer & Watling, 1973) or in ship fouling. It may have been independently introduced to southern California bays in ballast water or as fouling, or secondarily introduced from San Francisco Bay by coastal shipping.
Nereis succinea is common in San Francisco Bay in waters of less than two meters depth, generally at concentrations of 10-400 individuals/m2. It has mainly been collected in the northern Bay from San Pablo Bay to Antioch, and in the far South Bay below the Dumbarton Bridge (Hopkins, 1986). It is one of the dominant benthic organisms in Suisun Bay (Nichols & Thompson, 1985a). As discussed by Oglesby (1965), the native worm Nereis vexillosa occupies more marine waters in the Central Bay and the native Nereis limnicola occupies fresher waters in the Delta. Nereis succinea may thus have squeezed in between two existing pile worm populations, with each population restricted by a combination of physiological limitations and competition with its neighbors.
Recher (1966) noted Nereis succinea in the diet of shorebirds
in the South Bay, and Oglesby (1965b) reported on infection by
the trematode parasite Parvatrema borealis along the East
Bay shore. Carlton (1979a) summarizes other research on the worm's
physiology and ecology.
Polydora ligni Webster, 1879 [SPIONIDAE]
MUD WORM
SYNONYMS: Polydora amarincola Hartman, 1936
Polydora ligni is native to the northern Atlantic where it is found in mudflats, fouling (including ship fouling; Hartman, 1961) and oyster beds, sometimes forming thick mud beds that cause extensive oyster mortalities. In the Pacific it was first collected in Ladysmith Harbor, British Columbia in 1932 ("on [oyster] cultch sacks"), in San Francisco Bay in 1933 (redescribed as Polydora amarincola), and in False Bay on San Juan Island, Washington in 1937. It has since been reported from other bays and harbors in British Columbia, Washington and Oregon, and from Drakes Estero, Bolinas Lagoon, Elkhorn Slough, Morro Bay, Mugu Lagoon, Santa Monica Bay, Los Angeles/Long Beach Harbors, Alamitos Bay, Anaheim Bay, Santa Catalina Island, Mission Bay and the Salton Sea in California (see Carlton, 1979a, p. 306, for references). There are a few records, questioned by Carlton (1979a), from Mexico.
As with Heteromastus filiformis, Polydora ligni could have
been transported to the Pacific coast with Atlantic oysters decades
earlier and overlooked, or transported in ballast water (larvae
of Polydora species have been found to survive transport
in ballast tanks; Carlton, 1985, p. 345), or possibly in ship
fouling. Considerable movement between embayments along the coast
may have occurred with shellfish transplants or coastal shipping.
In San Francisco Bay it has been collected from the far South
Bay to Carquinez Strait (Light, 1977, 1978), and is one of the
more common benthic organisms in the shallows of San Pablo Bay
and the channels of the South Bay (Nichols & Thompson, 1985a).
Potamilla sp. [SABELLIDAE]
This worm was first collected in June 1989 at Sherman Lake in
the western Delta by the California Department of Water Resources.
It has been found from Frank's Tract and the Old River in the
Delta downstream to Grizzly Bay, and is most common at or just
upstream of the confluence of the Sacramento and San Joaquin Rivers,
where it has reached densities of over 16,000/m2
(W. Fields, pers. comm., 1995; DWR, 1995). Its absence from Delta
samplings in previous decades suggest a relatively recent introduction.
It was probably introduced in ballast water.
Pseudopolydora kempi (Southern, 1921) [SPIONIDAE]
SYNONYMS: Neopygospio laminifera Berkeley & Berkeley, 1954
Pseudopolydora kempi californica Light, 1969
Pseudopolydora kempi japonica Imajima & Hartman, 1964
This spionid worm has been reported from Mozambique, India, Japan and the Kurile Islands, in waters ranging from marine salinities down to 6 ppt (Light, 1969). It was first collected in the eastern Pacific in 1951 at Nanaimo, British Columbia, and later from False Bay, San Juan Island (1968) in Washington and Yaquina Bay (1974), Netarts Bay (1976) and Coos Bay (1977; JTC, pers. obs.) in Oregon. In California it appeared in Morro Bay (1960), Bolinas Lagoon (1967), San Francisco Bay (1972), and Bodega Harbor, Tomales Bay and Anaheim Bay (1975) (references in Carlton, 1979a, p. 310). Many of these sites have received shipments of the oyster Crassostrea gigas from Japan, possibly containing this worm. Alternatively it could have been transported in ballast water or ship fouling.
Light (1969) found that the California specimens more closely
resembled Indian than Japanese P. kempi. In California
P. kempi occurs intertidally and subtidally on mud and
sand. It has been collected in San Francisco Bay from the far
South Bay to the western end of Carquinez Strait (Light, 1977,
1978).
Pseudopolydora paucibranchiata (Okuda, 1937) [SPIONIDAE]
SYNONYMS: Polydora paucibranchiata
P. paucibranchiata was described from Japan. It was first reported from Australia in 1973 (Carlton, 1985) may also be present in New Zealand. It was reported from Los Angeles Harbor in 1950 and thereafter from other southern California sites: Newport Bay in 1951, San Diego Bay in 1952, Alamitos Bay in 1958, Anaheim Bay and Santa Barbara in 1975, and Mission Bay (in densities up to 60,000 individuals/m2) by 1981 (Carlton, 1979a; Levin, 1981). It was collected in South San Francisco Bay (Hunters Point and Oakland Inner Harbor) in 1973, Elkhorn Slough, Bodega Harbor and Tomales Bay in 1975 (where it "may be the dominant spionid polychaete on many sand flats;" Blake, 1975), and Netarts Bay, Oregon in 1976 (Light, 1977; Carlton, 1979a, p. 312).
Summarizing recent studies, Light (1978, p. 200) showed P.
paucibranchiata collected from the South Bay to the western
end of Carquinez Strait. It may have been introduced to the northeastern
Pacific in ballast water or in fouling on ships, possibly related
to increased ship traffic during or after the Korean War, or with
Japanese oysters.
Sabaco elongatus (Verrill, 1873) [MALDANIDAE]
BAMBOO WORM
SYNONYMS: Asychis elongata
Asychis amphiglypta (Ehlers)
Maldane elongata
Maldanopsis elongata
Brachioasychis colmani
Brachioasychis americana
This common "bamboo worm" is native to the western Atlantic from Maine to Florida, the Gulf of Mexico and British Honduras (Light, 1974). It was first reported from south San Francisco Bay in 1960 (Berkeley & Berkeley, 1960) and probably collected in the 1950s (Carlton, 1979a, p. 324). It is now extremely common, typically found in concentrations of 10-1,000 individuals/m2 at most stations from the far South Bay to mid-San Pablo Bay, and in concentrations of 1,000-5,000 individuals/m2 along the eastern shore of the Central Bay. It is not found upstream of San Pablo Bay (Hopkins, 1986).
Light (1974) suggested that Sabaco was introduced with
Atlantic oysters. As there had been no systematic subtidal benthic
sampling in San Francisco Bay since the 1912-13 Albatross
survey, it is conceivable that it was a late introduction with
oysters in the 1920s or 1930s and overlooked for 30 years. Alternatively,
it may have been introduced with ballast water.
Streblospio benedicti Webster, 1879 [SPIONIDAE]
SYNONYMS: Streblospio lutincola Hartman, 1936
Streblospio benedicti is common in the western Atlantic, ranging from the Gulf of St. Lawrence to the Gulf of Mexico and Venezuela, and is also found in northern Europe and the Mediterranean and Black seas. It was collected at Berkeley in San Francisco Bay in 1932, in Tomales Bay and Bodega Harbor by 1936, and in subsequent years in several other estuaries south to Newport Bay and north to Grays Harbor, Washington (records in Carlton, 1979a, p. 314). As with Polydora ligni, the other spionid discovered in San Francisco Bay in the 1930s, Streblospio could have been introduced with Atlantic oysters (with which it occurs on the Atlantic coast; Wells, 1961; Maurer & Watling, 1973), in ballast water, or possibly in ship fouling, and moved along the Pacific coast with shellfish transplants or coastal shipping.
In San Francisco Bay Streblospio benedicti has been collected
from the far South Bay to Antioch, commonly at densities of 1-10,000
individuals/m2 in the channels and up to 50,000
or more individuals/m2 in near shore areas,
especially in constricted embayments (Light, 1978; Hopkins, 1986).
It is one of the most common benthic organisms in the shallows
of San Pablo Bay and the channels of the South Bay (Nichols &
Thompson, 1985a).
Busycotypus canaliculatus (Linnaeus, 1758) [MELONGENIDAE]
CHANNELED WHELK
SYNONYMS: Busycon canaliculatum
Busycon pyrum
The channeled whelk, a native of the western Atlantic from Massachusetts to Florida, is now by far the largest snail in San Francisco Bay. As discussed by Carlton (1979a), Stohler (1962) stated that the whelk was first collected in the Bay at Alameda in 1948, but specimens from Berkeley at the California Academy of Sciences may have been collected as early as 1938. There are records and frequent observations of the whelk on the eastern shore of the Bay from Alameda and Bay Farm Island to Berkeley, and on the western shore from Belmont Slough to Candlestick Point. One specimen was collected in 1953 from the Tiburon Peninsula in Marin County (Stohler, 1962, Carlton, 1979a, p. 397).
The channeled whelk feeds on bivalves. It produces distinctive
strings of egg cases that release crawling (nonplanktonic) snails.
Natural dispersal may be achieved by floating egg cases, one string
of which was collected at Bolinas Lagoon. The whelk may have been
introduced to San Francisco Bay with some of the later and smaller
shipments of Atlantic oysters (with which it occurs on the Atlantic
coast; Wells, 1961; Maurer & Watling 1973), but could also
have been released from a private or school aquarium.
Cipangopaludina chinensis malleata (Reeve, 1863) [VIVIPARIDAE]
CHINESE MYSTERY SNAIL
SYNONYMS: Viviparus malleatus
Cipangopaludina malleata
Viviparus stelmaphorus Bourguignat
A long history of revisions and disagreements over identification, reviewed here with regard to Bay and Delta area specimens, leaves it unclear whether one or two (or possibly more) species of Japanese or Chinese viviparids have been introduced into California.
In 1892 Wood reported buying live snails from Japan at a Chinese market in San Francisco, at a price of ten cents per dozen, and found "that each specimen contained inside, from twelve to eighteen young shells." The snails were identified by W. J. Raymond as Paludina japonica Martens. Wood's specimens were later separated by Tien-Chien Yen at the California Academy of Sciences into three lots identified as Viviparus japonicus, Viviparus japonicus inakawa and Viviparus stelmaphorus. The last of these is accompanied by Wood's business card with the notation: "Bought alive for 10 cents a dozen at a Chinese vegetable store on Wed. morning, Nov 18/91- Came from China." Stearns (1901) described Wood's snails as "being part of the first lot brought alive from Japan, where they are collected in the rice-fields near Yokohama, and are sold for a few cents a quart."
Sorenson (1950) recalled purchasing Viviparus malleatus in Fresno's Chinatown in 1895 which "had been imported from Chinese rice fields to Fresno for the thousands of Chinese vineyard workers there." In 1901 Stearns reported receiving a few snails from the San Jose or Mt. Hamilton area "a year or more ago." One living specimen was examined and identified by Pilsbry as "Vivipara stelmaphora Bgt. (=V. malleata Rve.)." Later Hannibal (1908) found no viviparids in the Mt. Hamilton area, but between San Jose and San Francisco Bay collected snails identified by Dall as Vivipara lecythoides Bensen. He reported these as "introduced by the Chinese fifteen or twenty years ago" and "common where planted, but spreads slowly." A few years later, Hannibal (1911) reported that on re-examination both these snails and Wood's snails in Raymond's collection were Viviparus malleatus Reeve, which he said were "brought from Yokohama and originally planted between Alameda and Centerville [a small town 18 miles east of Fresno] to supply the markets of San Francisco Bay...whence colonies have been distributed to a number of points in the Sacramento-San Joaquin Valley as well. This is verified by specimens from an irrigating ditch near Fresno." However, Hannibal reported that he also found Vivipara japonica, "readily distinguished from malleatus," in an irrigation ditch at Hanford, about 30 miles southeast of Fresno.
The first record of introduced viviparids within the study zone consists of five shells at the California Academy of Sciences, labeled as malleata, collected from a slough near Holt in the Delta in 1938. Other specimens from within or near the Delta include eight snails collected from a canal north of Stockton in 1933, three snails from Victoria Island in 1941, eight snails from Sycamore Slough in 1946, and two undated snails from a slough near Stockton, all labeled as malleata. Greg (1948) reported finding a few live and many broken shells of Vivipara malleata in irrigation ditches near Stockton, speculating that muskrat may have been eating the snails. Sorenson (1950) reported collecting Viviparus malleatus from an irrigation canal 60 miles northwest of Fresno in 1948. Also, the wet collections at the California Academy of Sciences include two viviparid snails labeled Bellamya japonica that were collected at Stockton in 1968.
Hanna (1966), referred all existing western North America records to Viviparus stelmaphorus, based on finding enough variation in shell morphology in specimens from a single locality to encompass records that had been reported as malleata, japonica, iwakawa or lecythoides. He reported that the snails were still for sale in San Francisco markets and very abundant throughout the Delta and in irrigation canals, and in Mountain Lake and Stow Lake in San Francisco.
Taylor (1981) assigned these various California records to two species, Bellamya japonica (including Wood's 1891 market specimens, Hannibal's 1911 Hanford record, and records from Mountain Lake) and Cipangopaludina chinensis malleata (apparently including all other California records known to him), which he listed as occurring in irrigation ditches, sloughs and ponds from the Central Valley and San Francisco Bay area to southern California. He reported both species present in California since 1891.
Based upon these records, we conclude that the Chinese mystery snail is established in the study region. The current distribution and status of the Japanese mystery snail (placed in Bellamya by Taylor (1981) and in Cipangopaludina by Turgeon et al. (1988)) remains to be determined in the Bay area.
Viviparid snails from these one or more species have been reported
from many other North American locations, including: the Chinese
market at Victoria, British Columbia (Pilsbry & Johnson, 1894);
Muddy River in Boston's Fenway (from 1914 to at least 1942); Worcester,
Massachusetts (1917); Philadelphia (1925), at St. Petersburg,
Florida and near Niagara Falls (1942); Ottawa, Sioux City, Iowa
and Seattle (1943); near Agassiz, British Columbia (collected
by 1948, but reportedly planted in 1908); Lake Erie (1940s); Jefferson
County, Washington (1964); and Hawaii (by 1976) (La Rocque, 1948;
Abbott, 1950; Mills et al., 1993; and specimens at the California
Academy of Sciences). These snails are both used as food items
and commonly sold by dealers of aquarium fish, which has undoubtedly
helped to spread them (La Rocque, 1948; Abbott, 1950). They were
reportedly introduced to Sandusky Bay, Lake Erie to feed channel
catfish in the 1940s, and became so abundant by the 1960s that
they were a nuisance to commercial seine fisherman, who reported
sometimes catching two tons in a single seine haul (Wolfert &
Hiltunen, 1968).
Crepidula convexa Say, 1822 [CALYPTRAEIDAE]
CONVEX SLIPPER SHELL
SYNONYM: Crepidula glauca Say, 1822
This slipper shell is native to the western Atlantic, where it
is found from Nova Scotia to Florida and Puerto Rico. It was first
collected in San Francisco in 1898, from oyster beds, and was
almost certainly introduced in shipments of Atlantic oysters (with
which it occurs on the Atlantic coast; Wells, 1961). In San Francisco
Bay Hopkins (1986) reported Crepidula spp. mainly from
the South Bay, where C. convexa is commonly found on shells
of the native oyster Ostrea lurida and the Atlantic mudsnail
Ilyanassa obsoleta. It is not known from any other Pacific
coast site (Carlton, 1979a, p. 370).
Crepidula plana Say, 1822 [CALYPTRAEIDAE]
EASTERN WHITE SLIPPER SHELL
Crepidula plana is native to the western Atlantic with
a recorded range from Prince Edward Island to South America. It
was first reported on the Pacific Coast from the eastern shore
of San Francisco Bay in 1901, where it was probably introduced
with shipments of Atlantic oysters (with which it occurs on the
Atlantic coast; Wells, 1961), and was found in Willapa Bay and
Puget Sound in the 1930s and 1940s (Carlton, 1979a, p. 376). C.
plana is similar to and may be mistaken for the native flat
slipper shells C. perforans and C. nummaria, and
in fact went unreported in the Bay, though occasionally collected
and misidentified or unnoticed, for many decades after its initial
sighting. It is found considerably further into the estuary than
the native slipper shells which are restricted to the outer, more
marine portions of the Central Bay. On both the Atlantic coast
and in San Francisco Bay, C. plana is common on the inside
of hermit crab-occupied snail shells.
Ilyanassa obsoleta (Say, 1822) [NASSARIIDAE]
EASTERN MUDSNAIL
SYNONYMS: Nassarius obsoletus
This mudsnail is native to the western Atlantic from the Gulf of St. Lawrence to Florida. It was introduced to the Pacific Coast with shipments of Atlantic oysters (it is reported from oyster beds on the Atlantic coast; Wells, 1961), and was first collected in San Francisco Bay in 1907 from beds of Atlantic oysters at Alameda. Carlton (1979a) suggests that it was probably introduced between 1901 and 1907, as its presence in the Bay was unlikely to have been missed for very long due to the intensive activities of shell collectors in the area beginning in the 1890s.
Ilyanassa has also established breeding populations in Willapa Bay, Washington and Boundary Bay, British Columbia, first reported in 1945 and 1952 respectively but possibly present for a considerable time earlier. It has also been reported from but apparently not established populations in five additional Pacific Coast sites, as discussed by Carlton (1979a, p. 404): Tomales Bay (1920s-1930s?), "Bolinas Bay" (1920s or earlier), Humboldt Bay (1930), Birch Bay, British Columbia (1950s), and one specimen from Bodega Bay (1968).
Ilyanassa is today the dominant mudflat gastropod in San
Francisco Bay (Nichols & Thompson, 1985b), and is also sometimes
abundant in salt marshes and marsh sloughs and on pilings. Hopkins
(1986) reported it mainly from the southern part of the South
Bay and from San Pablo Bay, and we have also seen it abundant
at Alameda. Although intensively studied in the Atlantic (with,
for example, studies demonstrating significant effects on mudflat
community structure and sediment composition (Grant, 1965; Sibert,
1968)), there has been relatively little work on the Pacific Coast.
Ilyanassa is listed or mentioned in many faunal surveys
and checklists and bird diet studies (e. g. Painter (1966) lists
it an important food of diving ducks, but Williams (1929) and
Moffitt (1941) found it to be a minor or negligible food for California
clapper rail), and a few studies contain brief notes on its ecology
(Carpelan, 1957; Filice, 1959a; Quayle, 1964a; Vassallo, 1969).
Its distributional ecology in Lake Merritt is the subject of an
unpublished master's thesis (Gilmore, 1935). Grodhaus and Keh
(1959) found it to harbor five species of trematode flatworms,
including the schistosome Austrobilharzia variglandis which
is responsible for "swimmers' itch." Race (1979, 1982)
demonstrated competitive displacement and predation of the native
hornsnail Cerithidea californica, as discussed in Chapter
6.
Littorina saxatilis (Olivi, 1792) [LITTORINIDAE]
ROUGH PERIWINKLE
This common north Atlantic snail was first collected in San Francisco Bay by J. Carlton in May of 1993 on the shore of the Emeryville Marina. This site is adjacent to a public boat ramp and dock, and L. saxatilis was likely introduced in the seaweed used to pack live marine baitworms shipped from Maine and discarded by anglers. We have repeatedly found live L. saxatilis in the seaweed (Ascophyllum nodosum and occasionally other fucoid seaweeds) packing baitworms shipped to Newport Bay and San Francisco Bay (Carlton, 1979a; Lau, 1995; ANC, pers. obs.). As many as over a million Maine baitworms are shipped to the Bay Area each year (Lau, 1995) packed in seaweed containing many millions of living invertebrates from many phyla, so that this may be a transport vector of some significance (also see Miller, 1969).
We have irregularly visited and collected a total of about 100
live Littorina saxatilis from the shore of the Emeryville
Marina, where the snails were abundant intertidally in 1993 and
1994, and scarce in 1995, in the crevices of rocky debris along
about 10 meters of shoreline. They have not been observed elsewhere
in the Marina or the Bay. They produce "crawl away"
larvae, and could spread as eggs or snails on rafting seaweed.
Melanoides tuberculata (Müller, 1774) [THIARIDAE]
RED-RIM MELANIA
SYNONYMS: Thiara tuberculata
Melanoides tuberculata is a freshwater snail native to
the region from Africa to the East Indies. It was introduced to
the United States through the aquarium trade and was first reported
from California in 1972 from a drainage ditch in Riverside County
(Taylor, 1981). The California Department of Water Resources has
collected it from several sites in the Delta since December 1988,
at densities of up to 754 snails/m2 (DWR,
1995).
Urosalpinx cinerea (Say, 1822) [MURICIDAE]
ATLANTIC OYSTER DRILL
Urosalpinx cinerea is native to the northwestern Atlantic from the Gulf of St. Lawrence to Florida. It was introduced in shipments of Atlantic oysters to San Francisco Bay, where it was first collected from oyster beds at Belmont in 1890 (Stearns, 1894). It has been collected from many other bays in the northeastern Pacific, and is currently established in Boundary Bay, British Columbia (first record 1931), southern Puget Sound (1929), Willapa Bay (1948), Tomales Bay (1935) and Newport Bay (pre-1940s?) (Carlton, 1979a, p. 384). As Urosalpinx 's larvae are not pelagic, most of these sites represent either independent introductions from the Atlantic or intracoastal, human-aided transfers from other bays, including commercial shipments of oysters and other bivalves along the coast. Within San Francisco Bay, Hopkins (1986) reported Urosalpinx only from the South Bay.
Urosalpinx eats barnacles, mussels and bryozoans as well
as oysters. Although in some studies the drill has apparently
preferred barnacles or mussels to oysters (Haydock, 1964; Carlton,
1979a), its impacts on oysters, especially on oyster spat, can
be substantial (Haydock, 1964).
Boonea bisuturalis (Say, 1821) [PYRAMIDELLIDA]
TWO-GROOVE ODOSTOME
SYNONYMS: Menestho bisuturalis
Odostomia bisuturalis
Odostomia fetella
Boonea bisuturalis is native to the western Atlantic from the Gulf of St. Lawrence to Delaware, where it is an ectoparasite both of the Atlantic oyster Crassostrea virginica and of a number of bivalves and gastropods that were transported to San Francisco Bay with shipments of Atlantic oysters. It was reported in San Francisco Bay in 1977 associated with the Atlantic mudsnail Ilyanassa obsoleta and the native hornsnail Cerithidea californica on the Fremont shore (Race, pers. comm.), and reported as common on a far South Bay mudflat (Nichols & Thompson, 1985b). Odostomia fetella reported from San Pablo Bay (Filice, 1959) and Suisun Bay (Markman, 1986) may also be this species. Carlton (1979a, p. 435) argues that Boonea bisuturalis was probably introduced with oyster shipments in the 19th or early 20th century, and remained unreported because of incomplete systematic work on the Odostomia complex in the northeastern Pacific. He predicts that early collections of Boonea bisuturalis and possibly other species of Atlantic odostomids will be found when unsorted, unidentified or misidentified material in museum collections is systematically worked up by specialists.
Although, based on its associations, Boonea was probably
an introduction with oyster shipments that remained unrecognized
for many years, it might possibly have been a later introduction
in ballast water.
Catriona rickettsi Behrens, 1984 [TERGIPEDIDAE]
SYNONYMS: Trinchesia sp. Behrens & Tuel, 1977
Catriona rickettsi was first collected in San Francisco
Bay from Pete's Harbor, San Mateo County in 1974, where it is
associated with and presumably feeds on the hydroid Tubularia
crocea (Behrens & Tuel, 1977; Behrens, 1984), and was
subsequently collected from La Jolla (Behrens, 1980). In 1995
it was collected on Tubularia marina on the ocean side
of the Umpqua River jetty in Oregon (J. Goddard, pers. comm.,
1995). The most likely means introduction is in ballast water
or transported as eggs on ship fouling. Its origin is unknown.
Cuthona perca (Marcus, 1958) [TERGIPEDIDAE]
LAKE MERRITT CUTHONA
In California, Cuthona perca is known only from Lake Merritt,
where it feeds on the introduced Japanese anemone Haliplanella
lineata (Carlton, 1979a, p. 431, as Trinchesia sp.)
It is reported from Brazil, Jamaica, Miami, Barbados, New Zealand
and Hawaii (Behrens, 1991). The most likely mechanisms of transport
are either in ballast water or as eggs on ship fouling.
Eubranchus misakiensis Baba, 1960 [EUBRANCHIDAE]
MISAKI BALLOON AEOLIS
Eubranchus misakensis was described from Japan in 1960
and collected at the San Francisco Municipal Marina in 1962 (Behrens,
1971; Gosliner, 1985). It occurs on boat floats and docks and
silty-clay bottoms throughout the Bay, where it is found with
and apparently feeds on the hydroid Obelia. (Carlton, 1979a,
p. 433; Behrens, 1971, 1991). It may have been introduced in ballast
water or as eggs on ship fouling, or possibly with shipments of
Japanese oysters and overlooked for a few decades.
Okenia plana Baba, 1960 [GONIODORIDIDAE]
FLAT OKENIA
Okenia was first reported from San Francisco Bay by Joan
Steinberg in 1960 (the same year it was described from Japan),
based on collections in the 1950s. It has also been reported from
San Onofre, Orange County (Gosliner, 1995). It occurs on floats
and pilings among fouling and with egg cases on a membraniporid
bryozoan (tentatively identified as Conopeum tenuissimum),
on rocks on mudflats, and subtidally in San Francisco Bay, where
it has been reported from the South Bay (Palo Alto Yacht Harbor,
Crown Beach in Alameda), Central Bay (Berkeley Pier and Yacht
Harbor, San Francisco Yacht Harbor) and San Pablo Bay (Point Richmond
and China Camp) (Carlton, 1979a, p. 425; ANC, pers. obs.). Carlton
(1979a) suggests that it was probably introduced with shipping
from Japan, either in ballast water or as eggs on fouling, perhaps
related to increased trans-Pacific ship traffic during and after
the Korean War. Alternatively it could have been introduced with
shipments of Japanese oysters and overlooked for a couple of decades.
Philine auriformis Suter, 1909 [PHILINIDAE]
TORTELLINI SNAIL
Philine auriformis is native to New Zealand and possibly southern Australia, and was first identified from San Francisco Bay in July, 1993. It had been collected from the South Bay for about a year prior to its recognition as an introduced species (i.e. since about the summer of 1992) in trawls by the Marine Science Institute of Redwood City, USGS and CDFG (K. Grimmer, J. Thompson and K. Hieb, pers. comm.). By 1994 it was regularly collected in otter trawls and benthic samples from the Central Bay (P. Donald, pers. comm.; ANC, pers. obs.), and snails and egg masses (which successfully hatched in the laboratory) were collected from intertidal mudflats in Bodega Harbor, 120 km north of the entrance to San Francisco Bay, in April, 1994. As it is not known from fouling, Philine was probably introduced to California via ballast water (Gosliner, 1995).
All specimens were taken from fine, silty mud. Stomachs contained
fragments of bivalve shells, Nutricula (=Transennella
)tantilla and N. confusa in Bodega Harbor and possibly
the introduced bivalve Gemma gemma in San Francisco Bay
(Gosliner, 1995).
Sakuraeolis enosimensis (Baba, 1930) [FACELINIDAE]
WHITE-TENTACLED JAPANESE AEOLIS
SYNONYMS: Coryphella sp. Behrens, 1980
Sakuraeolis enosimensis is native to Japan and was first
collected in San Francisco Bay in 1972. It is common and widespread
in the southern portions of San Francisco Bay (Gosliner, 1995),
where it feeds on the hydroid Tubularia crocea growing
on boat docks (Behrens, 1991). It could have been introduced
in ballast water or as eggs on fouling.
Tenellia adspersa (Nordmann, 1845) [TERGIPEDIDAE]
MINIATURE AEOLIS
SYNONYMS: Tenellia pallida (Alder & Hancock, 1854)
Embletonia sp. Alder & Hancock, 1851
Tenellia adspersa is widespread in European and Mediterranean waters and recently reported from Chesapeake Bay and Brazil, with a single 2 mm specimen reported from Japan (Carlton, 1979a). It was first collected from the Pacific Coast of North America at Point Richmond in San Francisco Bay in 1953, and later from the Richmond and Berkeley Yacht Harbors, Lake Merritt, San Leandro Bay, Sausalito and South Beach Harbor, San Francisco (Carlton, 1979a, p. 428; Jaeckle, 1983; ANC, pers. obs.). It is now known from Coos Bay to Long Beach (Gosliner, 1995).
In Europe it is reported to range from waters of ocean salinity
to "quite fresh water" and feeds voraciously on a variety
of hydroids including the freshwater hydroid Cordylophora caspia
(Roginskaya, 1970), which is introduced to and common in the Delta.
In San Francisco Bay Tenellia adspersa apparently feeds
on the introduced hydroids Tubularia crocea (Carlton, 1979a;
Behrens, 1991) and Obelia dichotoma (Jaeckle, 1983). Carlton
(1979b) suggested that it was probably introduced from Europe
by shipping, either in ballast water or as eggs on fouling.
Ovatella myosotis (Draparnaud, 1801) [MELAMPIDAE]
SYNONYMS: Alexia setifer Cooper, 1872
Alexia setifer var. tenuis Cooper, 1872
Phytia myosotis
Ovatella myosotis occurs on both coasts of the north Atlantic, but may have been introduced to the western Atlantic in the late 18th or early 19th century (Berman & Carlton, 1991). It was first collected from San Francisco Bay in 1871, probably introduced with Atlantic oysters, although possibly carried in wet ballast or wedged into holes or cracks in the wooden hulls of sailing vessels. Failure to find it earlier in San Francisco Bay despite intensive prior shell collecting in the area, plus the initiation of Atlantic oyster shipments with the completion of the transcontinental railway in 1869, suggests that O. myosotis was introduced not long before its discovery, probably in 1869-1871.
O. myosotis was collected in Humboldt Bay in 1876, in San Pedro Harbor in southern California in 1915, and in Washington state in 1927. It has now been recorded from numerous Pacific coast bays and estuaries from Boundary Bay, British Columbia to Scammons Lagoon, Baja California (Carlton, 1979a, p. 414). Since O. myosotis lacks planktonic larvae, these additional sites resulted from transport either on coastal shipping or in replantings of oysters, or from separate introductions from the Atlantic.
O. myosotis is absent from Pacific coast Pleistocene deposits, but there is one anomalous report by Gifford (1916) of this snail in an aboriginal shellmound on the shore of San Francisco Bay. Carlton (1979a) doubts this is Ovatella, and Gifford's material has been lost.
O. myosotis is euryhaline and lives under boards and debris
near the high-tide line of salt marshes and protected beaches
in lagoons and bays. The snail has been studied in Europe but
largely ignored in North America. On the Pacific coast it has
been reported from the stomachs of willets (Catoptrophorus
semipalmatus) (Stenzel et al., 1976). Carlton (1979a) noted
that its co-occurrence in various Pacific coast sites with several
species of native and introduced snails provided suitable systems
for the study of competitive interactions between native and introduced
species. Berman and Carlton (1991) found dietary overlap with
the native snails Assiminea californica and Littorina
subrotundata in Coos Bay, Oregon, but no evidence of competitive
superiority by O. myosotis, and concluded that its establishment
was not at the expense of the native snails.
Arcuatula demissa (Dillwyn, 1817) [MYTILIDAE]
RIBBED MUSSEL, RIBBED HORSE MUSSEL
SYNONYMS: Ischadium demissum
Modiolus demissus
Geukensia demissa
Volsella demissus
Brachidontes demissus
Modiolus plicatulus Lamarck, 1819
Arcuatula demissa (more commonly known as Ischadium demissum on the Pacific coast and as Geukensia demissa on the Atlantic coast) is native to the northwest Atlantic, commonly found in salt marshes from the Gulf of St. Lawrence to North Carolina. Southward it is replaced by a subspecies, Arcuatula demissa granossisimum. It was first collected in the Pacific from south San Francisco Bay in 1894 (Stearns, 1899), probably introduced with Atlantic oysters (small Arcuatula are commonly found on oysters in the Atlantic; Wells, 1961; Maurer & Watling, 1973). It has since been collected from three other sites: Newport Bay (first collected in 1940), Alamitos Bay (1957) and Anaheim Bay (1972) (Reish, 1968, 1972; Carlton, 1979a, p. 440). Questionable or probably adventitious specimens from other Pacific coast bays are discussed by Carlton (1979a).
Arcuatula has become one of the most abundant bivalves in San Francisco Bay. De Groot (1927) reported that "countless millions of these small mussels cover the edges and sometimes the entire bottoms of the gutters and creeks of the west Bay marshes." Pestrong (1965) found in the Palo Alto area that they "effectively rip-rap channel banks when they form in large colonies, as is often the case." Carlton (1979a,b) found Arcuatula lining the base of concrete retaining walls at Lake Merritt, a brackish lagoon in Oakland. Arcuatula is common and often abundant in salt marshes from the South Bay to San Pablo Bay, where it frequently lies embedded with its posterior margin protruding above the mud.
This "endobyssate" habit has resulted in a curious reported
effect on the endangered California clapper rail (Rallus longirostris
obsoletus). De Groot (1927) reported that the toes or probing
beaks of rails are caught and clamped between the exposed, slightly
gaping valves of the mussel. He reported that almost every rail
examined over the preceding twenty years was missing one or more
toes, presumably from this cause, that others had had their beaks
clamped shut and died of starvation, and estimated that an average
of one or two chicks per brood were caught by mussels and drowned
by the incoming tide. More recent observers note that clapper
rails in San Francisco Bay are frequently missing one or more
toes (Moffitt, 1941; Josselyn, 1983; Takekawa, 1993), and Takekawa
(1993) reported that a rail captured in the Palo Alto marshes
with a mussel clamped onto its bill subsequently lost part of
its bill. On the other hand, Moffitt (1941) found that Arcuatula
formed 57 percent by volume of the total food in 18 clapper rail
stomachs that he examined in 1939, and Recher (1966) and Anderson
(1970) recorded Arcuatula from the stomachs of willet and
dunlin in the South Bay.
Corbicula fluminea (Müller, 1774) [CORBICULIDAE]
ASIAN CLAM, ASIATIC CLAM
SYNONYMS: Corbicula fluviatalis (Müller, 1774)
Corbicula manilensis (Philippi, 1841), Corbicula leana
(Prima, 1864) and Corbicula sinensis as reported in North
America, and many other names in Asia; see Prashad (1929), Morton
(1979), Britton & Morton (1979), and Woodruff et al. (1993)
for extensive synonymies
This freshwater clam is native to China, Korea and the Ussuri Basin in southeastern Siberia (Ingram, 1948), with closely related and possibly conspecific populations in Japan (Britton & Morton, 1979). The earliest North American record consists of three shells collected on the beach at Nanaimo, British Columbia in 1924, though no further specimens have been reported from Canada (Counts, 1981). Corbicula was next collected from the mouth of the Columbia River in 1938 (McMahon, 1982). It was reported from the Delta in 1945 (Hanna, 1966) and widespread there by 1948 (Ingram, 1948), and reached the Imperial Valley in southeastern California by 1952 (McMahon, 1982).
From southern California Corbicula spread eastward to Arizona by 1954 (Ingram, 1959), and to near El Paso in west Texas by 1964 (McMahon, 1982). Meanwhile, Corbicula was collected from the Ohio River near Paducah, Kentucky in 1957, which McMahon (1982) suggests initiated a second zone of dispersal in North America. By the end of the 1960s Corbicula had spread through the lower Mississippi and Ohio river valleys, into southeast Texas and Oklahoma, and along the Gulf coast from Louisiana to southern Florida, and by the mid-1970s had spread up the Mississippi Valley to northern Iowa and along the Atlantic coast from Florida to New Jersey. By the early 1980s, Corbicula was found in 35 of the United States and in northern Mexico (McMahon, 1982). Corbicula was reported from South America, France and Portugal in 1981, and a specimen was collected from a stream in Oahu, Hawaii in 1992 (Araujo et al., 1993; Burch, 1994).
Although for many years the Corbicula in North America were described as belonging to at least three different species, in 1979 Britton & Morton argued that only one species is involved, the highly variable Corbicula fluminea, a view that has generally been accepted since. Corbicula from California, Texas, Arkansas, Tennessee and South Carolina showed no genetic variation between populations at 18 loci, 14 of which were polymorphic in some Asian Corbicula (Smith et al., 1979).
Since Corbicula are cultivated and sold as food in many Asian countries, many researchers have suggested that it was deliberately introduced to establish a food resource (e. g. Ingram, 1948; Hanna, 1966; Britton & Morton, 1979; McMahon, 1982), or possibly introduced through the aquarium trade (Ingram et al., 1964). Some researchers have suggested that it was introduced with Japanese oysters (Burch, 1944; Hill, 1951; Filice, 1959), but since Corbicula is mainly a freshwater organism, this seems unlikely.
Corbicula's spectacular spread within and between watersheds in North America may have resulted from transport for use as bait, food or aquarium pets, or in river gravels dredged for use as aggregate (Ingram et al., 1964), although McMahon (1982) argues that natural means of dispersal were paramount, including passive downstream transport of juveniles in currents, upstream transport in fish stomachs, and upstream or between-watershed transport on birds. Corbicula are fairly hardy, tolerating several months without food (Hanna, 1966) and 7-27 days out of water (McMahon, 1979). One specimen was mailed, dry, in an envelope from Pennsylvania to Washington state for identification and mailed back without ill effect (McBane, pers. comm., 1995).
The use of Corbicula in aquaculture or for wastewater clarification, in either commercial or experimental applications as on St. Croix, Virgin Islands (Haines, 1979), may serve to introduce the clam to new locations in the future.
Corbicula is today the most widespread and abundant freshwater clam in California, found throughout lower elevation waters, the dominant mollusk and the third most abundant benthic organism in the Delta, and one of the most commonly identified benthic organisms in fish stomachs (Gleason, 1984; Herbold & Moyle, 1989). Densities of 2,000 young clams/m2 are common, and range up to 20,000/m2. Spring flows carry young Corbicula down to Suisun Bay where they are sometimes collected as far west as Martinez, but high fall salinities appear to prevent the establishment of large adult populations even in the western Delta (Hazel & Kelley, 1966; Evans et al., 1979; Markmann, 1986).
Populations of Corbicula with typical densities of 10,000 to 20,000 clams/m2 (with a maximum of 131,200/m2) trapped sediment and formed extensive bars in the Central Valley Project's Delta-Mendota Canal, reducing delivery capacity and requiring expensive dewatering and the dredging of over 50,000 cubic yards of clam-bearing material. One bar was described as filling the bottom of the canal from 0.3-1.0 meter deep for 3 kilometers (Hanna, 1966; Eng, 1979). Ingram (1959) reported the clam as an economic pest of water delivery systems in California, infesting and impairing operation of underground pipes, turnout valves, laterals and agricultural sprinkler systems in the Coachella and Imperial valleys, and plugging the tubes of condenser-cooler units at the federal government's Tracy Pumping Plant in the Delta. Corbicula is frequently cited as a significant problem in fouling irrigation systems, municipal water systems, power plant steam condensers, emergency reactor cooling systems and service water systems elsewhere in the country (e. g. Ingram et al., 1964; Sinclair, 1964; Hanna, 1966; Goss & Cain, 1977; McMahon, 1977, 1982; Mattice, 1979; Goss et al., 1979; Parsons, 1980).
Corbicula is also reported to render river sand and gravel unfit for use as aggregate, and to outcompete native unionid and sphaeriid clams (McMahon, 1982). Blue catfish, Ictalurus furcatus, were introduced to some California waters in part to control Corbicula, but without success (Gleason, 1984).
Upper salinity tolerances for Corbicula fluminea have been reported at 14 ppt (Gainey, 1978), 13-17 ppt (Morton & Tong, 1985), and about 10 ppt without acclimation and 22-24 ppt with acclimation (Evans et al., 1979). Sparse populations of Corbicula have been observed in the San Francisco Estuary near Martinez at 17 ppt, and abundant populations in areas subjected to daily salinities of 10 to 12 ppt (Evans et al., 1979).
Corbicula fluminea are viviparous, releasing benthic pediveliger larvae or planktonic veligers that become benthic within 48 hours (Eng, 1979). There are typically two spawning periods per year, with one study reporting peak production of over 800 larvae/clam/day and an average of 1,140,820 larvae/m2/year. Biomass productivity rates were the highest ever recorded for a freshwater bivalve, and higher than most marine bivalves (Aldridge & McMahon, 1978).
In California there are modest market sales of Corbicula
both for bait and for food (Gleason, 1984; commercial harvesting
for food is allowed only in Lake Isabella in Kern County). It
was noncommercially harvested from the Delta for food at least
as early as 1946 (Hanna, 1966).
Gemma gemma (Totten, 1834) [VENERIDAE]
AMETHYST GEM CLAM
SYNONYMS: Gemma purpurea (Lea, 1842)
This small, viviparous clam, native to the northwestern Atlantic from Nova Scotia to Florida and Texas, was first reported from the Pacific coast as 42 specimens recovered from the crop of a duck bought in a San Francisco market in 1893. It was collected directly from the Bay in the late 1890s, from Bolinas Lagoon in 1918 and from three other nearby embaymentsóBodega Harbor, Tomales Bay and Elkhorn Sloughóin the 1960s and 1970s (Carlton, 1979a, p. 490).
Earlier observations of Gemma gemma in these embayments could have gone unremarked because of confusion with the small native venerid Transennella tantilla. The early records from San Francisco Bay noted above were originally identified as Transennella, and many later reports of Gemma gemma from various Pacific coast embayments and offshore sites were based on material that on re-examination turn out to be Transennella or one of two other native clams (Carlton, 1979a).
Gemma gemma was probably introduced with Atlantic oysters,
which it commonly occurs on the Atlantic coast (Wells, 1961; Maurer
& Watling, 1973). It is abundant on the intertidal
mudflats from the far South Bay through San Pablo Bay where it
is one of the most common benthic species, in places reaching
midsummer densities of over 400,000 individuals/m2
(Nichols & Thompson, 1985a, 1985b) and is occasionally
found up through Suisun Bay (Hopkins, 1986). It has been found
in the stomachs of ten species of shorebird in San Francisco Bay
(Recher, 1966), of white sturgeon (McKechnie & Fenner, 1971),
and possibly of the introduced nudibranch Philine auriformis
(Gosliner, 1995), is reported as an important food of diving ducks
(Painter, 1966), and is undoubtedly eaten by many other organisms.
Oglesby (1965) suggested that Gemma gemma may be the first
intermediate host of the trematode Parvatrema borealis.
The trematode makes characteristic pits in the shell of Gemma
gemma, and such pits have been found in shells from San Francisco
Bay, Bolinas Lagoon and Tomales Bay (Carlton, 1979a).
Lyrodus pedicellatus (Quatrefages, 1849) [TEREDINIDAE]
BLACKTIP SHIPWORM
SYNONYMS: Teredo diegensis Bartsch, 1916 from San Diego
Teredo townsendi Bartsch, 1922 from San Francisco Bay
many other synonyms from other parts of the world (Turner, 1966)
Lyrodus pedicellatus is a warm-temperate and subtropical wood-boring shipworm that requires temperatures of 14 to 24°C and salinities of at least 29 ppt to breed (Eckelbarger & Reish, 1972). It has been reported from many parts of the worldóthe eastern and western Atlantic, the Indo-Pacific region, Australasia, South Africa, Japan and Hawaiióand its origin is unknown, having been early and widely distributed either by drifting wood or in the hulls of ships. It has repeatedly been "discovered" and described as a new species: 12 times in the Atlantic, and 21 times in the Pacific (Turner, 1966; Carlton, 1979a, p. 551).
A shipworm, apparently Lyrodus, was reported from Wilmington
Harbor (now part of the Los Angeles-Long Beach Harbor system)
in 1871 and following years, and Lyrodus was collected
from San Diego Harbor by 1876. It was subsequently very abundant
in these harbors (Miller, 1926). It was collected from San Bruno
Slough in south San Francisco Bay in 1920, from Elkhorn Slough
in 1935, and from several southern California bays and ports beginning
in the 1940s (Carlton, 1979a).
Macoma petalum (Valenciennes, in Humbold & Bonpland,
1821) [TELLINIDAE]
BALTIC CLAM
SYNONYMS: Macoma balthica of San Francisco Bay authors
Macoma inconspicua of San Francisco Bay authors
This Macoma species in San Francisco Bay has heretofore been known as Macoma balthica. In recent decades, M. balthica has generally been regarded as a single species with a circumboreal/arctic distribution, with records from central California north to Alaska and the Bering Sea, the Okhotsk and Japan seas, the Beaufort and Siberian seas, the Barents and White seas, northern Europe, the mid-Atlantic states north to western Greenland, Hudson Strait, Hudson Bay, and Bathurst Inlet in the Canadian Archipelago. However, the analysis of shell characteristics and growth rates (Beukema & Meehan, 1985) and allozymes (Meehan, 1985; Meehan et al., 1989) clearly indicates the existence of two species, one native to the northwestern Atlantic (here called Macoma petalum), the other native to the northeastern Atlantic and northern Pacific (Macoma balthica).
Based on recent studies, the small pink Macoma of San Francisco Bay, long thought to be native Macoma balthica, appears rather to be M. petalum introduced from the northwestern Atlantic. Tested at eleven loci, the allele frequencies of San Francisco Bay specimens closely resembled those of northwestern Atlantic M. petalum (Nei's (1978) unbiased genetic identity of 0.943), and differed sharply from those of M. balthica from Alsea Bay and Coos Bay, Oregon (genetic identity of 0.394-0.461) (Meehan et al., 1989). Genetic identities >0.9 are generally thought to occur among conspecific populations, of 0.5-0.8 among sibling species, and of <0.5 among non-sibling species (Meehan et al., 1989).
The early history of Macoma balthica and petalum in San Francisco Bay remains to be worked out. Shells identified as M. balthica have been recovered from 2,000-6,000 year old sediments under San Francisco Bay. It may be that Macoma balthica then died out in the Bay, as Meehan et al. (1989) argued based on the lack of records from later sediments and aboriginal shell middens in the region. Clams, apparently referable to M. balthica or petalum, were collected in the Bay by the United States Exploring Expedition in 1841 and by various parties in the 1860s (Carpenter, 1857, 1864; E. Coan, pers. comm., 1995). They were found to be common in all parts of the Bay in the Albatross survey of 1912-13 (Packard, 1918).
Clams collected prior to 1850 could represent Macoma balthica native to the Bay, if an aboriginal population persisted despite Meehan et al.'s arguments; or could represent M. balthica from further north on the Pacific coast or M. petalum from the northwestern Atlantic introduced in solid ballast. Clams collected after 1850 could in addition represent M. balthica from northern bays introduced with transplants of the native oyster Ostrea conchaphila (=lurida). Clams collected after 1869 could in addition represent M. petalum introduced with shipments of the Atlantic oyster Crassostrea virginica. Morphologic (Beukma & Meehan, 1985) or genetic analysis of museum specimens might sort some of these possibilities out.
The current distributional pattern of Macoma balthica and Macoma petalum in the northwestern Pacific, particularly between San Francisco Bay and Coos Bay, also remains to be determined. South of San Francisco Bay, there are records of shells and possibly live specimens of "Macoma balthica" as far south as San Diego, but these appear to be sporadic occurrences, probably related to anthropogenic transport, rather than established populations.
Macoma petalum or balthica has been collected throughout San Francisco Bay upstream to Collinsville, especially in the shallows where densities have reached over 1,000 individuals/m2 (Siegfried et al., 1980; Hopkins, 1986; Markmann, 1986), and has been a dominant benthic organism in South Bay and Suisun Bay shallows (Nichols & Thompson, 1985a). It can be an important food of fish, diving ducks and clapper rail (Williams, 1929; Painter, 1966), and formed 8 percent of the volume of food in 18 clapper rail stomachs (Moffitt, 1941). In San Francisco Bay Macoma feeds on both planktonic and benthic microalgae, and Thompson & Nichols (1988) found that the timing and rate of growth of intertidal populations was controlled by food supply and high mud-flat (air) temperatures, and independent of salinity over a 0-31 ppt range.
It was recently determined that Macoma balthica from both
Vancouver Island and the Baltic Sea host the same three species
of digenean flatworms (Pekkarinen & Ching, 1994). It would
be of interest to determine whether Macoma petalum from
San Francisco Bay and the northwestern Atlantic host the same
or different parasites.
Musculista senhousia (Benson, 1842) [MYTILIDAE]
SYNONYMS: Musculus senhousia
Modiolus demissus of Filice (1959)
Native to Japan and China, this small mussel was introduced to Washington and central California with Japanese oysters (Crassostrea gigas), with which it has been found in incoming seed (Kincaid, 1949). It was collected in Samish Bay, Washington, on beds of Japanese oysters in 1924, and at Olympia in 1959. In central California it was collected from Tomales Bay in 1941, Bolinas Lagoon in 1944, San Francisco Bay in 1946, Elkhorn Slough in 1965 and Bodega Harbor in 1971. It was collected from Mission, San Diego and Newport bays in southern California, and Papilote Bay (near Ensenada) in Baja California in the 1960s and 1970s (Carlton, 1979a, p. 449), probably transported in ballast water or on ship or boat fouling. In the 1970s it appeared in New Zealand and Australia and in the 1980s in the Mediterranean.
In the western Pacific Musculista has been reported at densities of up to 28,650 juveniles/m2 settled on eelgrass or 2,500-2,800 adults/m2 just buried in the mud of the tidal flats, where the clams build nests about them of byssal thread, mucus and sediment. Musculista is used as food in China and as fish bait and as feed for cultivating shrimp and crab in Japan (Morton, 1974; Carlton, 1979a).
On the bottom of Lake Merritt, a shallow, brackish Lagoon on San
Francisco Bay, Musculista occurs in dense byssal mats that
can be pulled from the bottom in sheets, and as individuals among
the fouling on pilings and floats. At Alameda individuals are
found nesting in the sediment or attached to the base of eelgrass
plants. Musculista has been collected at densities of up
to 1,000-2,000 clams/m2 from the South Bay
to San Pablo Bay, where it has frequently been one of the most
common benthic organisms, and occasionally collected upstream
to Honker Bay (Nichols & Thompson, 1985a; Hopkins, 1986; Markmann,
1986). Crooks (1996) has investigated its ecology and biology
in Mission Bay in southern California.
Mya arenaria Linnaeus, 1758 [MYIDAE]
SOFT-SHELL CLAM
SYNONYMS: Mya hemphillii Newcomb, 1874
Mya arenaria is native to the American Atlantic coast and from Alaska north of the Aleutian Peninsula, although its distribution north of British Columbia is not well known. It has been introduced into western and northern Europe. Although recorded from Miocene and Pliocene deposits on the Pacific coast, it has not been found in Pleistocene deposits or in aboriginal shell middens south of the Bering Sea, and had not been encountered by numerous collectors on the Pacific coast prior to 1874 (Stearns, 1881). In that year it was collected in San Francisco Bay (Newcomb, 1874), almost certainly transported there in the transcontinental shipments of Atlantic oysters that began in 1869.
This large, edible clam was soon transplanted to other Pacific Coast sites (e. g. Coos Bay, Oregon by 1880, Santa Cruz, California by 1881, Willapa Bay and Puget Sound in Washington by 1884 and 1888-89; also note Stearns' (1881) exhortation that "it would be a wise, public spirited act if the captains of our coasting vessels would take the trouble and incur the slight expense attending the planting of this clam at such points as their vessels touch at in the ordinary course of business"), and may have been distributed to others with transplantings of oysters from these sites or with fresh introductions of oysters from the Atlantic. It is less likely, though possible, that Mya arenaria's appearance in some locations resulted from deliberate introductions from the Atlantic (which Rathbun (1892), Heath (1916) and Coe (1956) claim was attempted or occurred), or from the transport of small clams in ship fouling. Although some workers have suggested that some or all of Mya arenaria's northward movement was due to natural dispersal (e. g. Quayle, 1960), Carlton (1979a) concludes that "there is little hard data that Mya has ever spread naturally anywhere along the Pacific coast." Mya arenaria does not appear to have become established south of Monterey, despite a planting of about 2,000 clams in Morro Bay in 1915 and occasional, probably erroneous reports of Mya arenaria from southern California (reviewed in Carlton, 1979a).
By the 1880s Mya arenaria was reported as the most common clam sold in San Francisco Bay area markets (Stearns, 1881). But the commercial harvest declined from 500-900 tons per year in 1889-1899, to generally above 100 tons per year in 1916-1926, to nothing after 1948, possibly due to overharvesting, habitat loss, pollution or a decline in the market due to an increasing harvest of Venerupis phillipinarum (Skinner, 1962; Herbold et al., 1992). Today, noncommercial harvest of Mya continues for food and bait (Sutton, 1981; Herbold et al., 1992). It has been collected throughout the Bay as far upstream as Collinsville and Sherman Lake, frequently at densities over 100 and sometimes over 1,000 clams/m2, and has been one of the dominant benthic organisms in the shallows of the South Bay and Suisun Bay (Nichols & Thompson, 1985a; Hopkins, 1986; Markmann, 1986).
Several workers reported that Mya arenaria replaced populations
of the native clam Macoma nasuta in San Francisco Bay,
at least in regularly harvested clam beds (e. g. Fisher, 1916).
Clam beds encompassing from a few to hundreds of acres were established
from the South Bay to the Napa River and Martinez, some of them
public and some privately owned, with some fenced to keep out
bat rays and flounder (Bonnot, 1932). Predators of Mya arenaria
on the Pacific coast include rays, sharks, flounder, ducks and
shorebirds. Five species of native pinnotherid crabs are recorded
as living in Mya arenaria's mantle cavity (references in
Carlton, 1979a).
Mytilus galloprovincialis Lamarck 1819 [MYTILIDAE]
MEDITERRANEAN MUSSEL
SYNONYMS: the taxonomy of the Mytilus "edulis"
complex is reviewed by Koehn (1991) and Seed (1992)
The cosmopolitan Mytilus "edulis" species complex was variously grouped into one or several species by different authors until electrophoretic evidence published in the late 1980s and 1990s led to the general recognition of three species: M. edulis from northern Europe and eastern North America; M. galloprovincialis from the Mediterranean Sea, various sites on the Atlantic coast of Europe, South Africa, California, Japan, Hong Kong and eastern China, Australia, Tasmania and New Zealand; and M. trossulus from the northwestern Pacific, Siberia, eastern Canada and the Baltic Sea (McDonald et al., 1991; Koehn, 1991; Seed, 1992), although frequent hybridization between these forms may raise doubts about their specific status (Seed, 1992). Mussels from Chile, Argentina, and the Falkland and Kerguelen islands contain alleles characteristic of all three genotypes but have been tentatively assigned to M. edulis (McDonald et al., 1991).
The two species present in the northwest Pacific have been differentiated on the basis of morphometric analysis (Sarver & Foltz, 1993; mussels from San Francisco Bay collected in 1990), starched gel electrophoresis at 8-15 allozyme loci (McDonald & Koehn, 1988, using mussels collected in 1985-87; Sarver & Foltz, 1993), and the sequencing of mitochondrial 16S ribosomal DNA (Geller et al., 1993, 1994). All methods agree in finding predominantly or purely M. trossulus type from Eureka, California north to Alaska; a hybridization zone including Westport, Tomales Bay, San Francisco Bay and Monterey Bay where sites contained various mixtures of M. trossulus, M. galloprovincialis and their hybrids; and high proportions of M. galloprovincialis at sites south of Monterey to San Diego.
However, these methods differed in their conclusions about how dominant M. galloprovincialis is south of Monterey, with allozyme analyses showing almost pure M. galloprovincialis genotype and DNA analysis showing a roughly equal mix of M. galloprovincialis-M. trossulus genotypes. Geller et al. (1994) suggest that this could result from the introgression of the M. trossulus mitochondrial genome into individuals with M. galloprovincialis nucleic genome. Since mitochondrial DNA is mainly transmitted maternally in Mytilus species, such introgression could be produced by repeated crossings with M. galloprovincilis males with a female M. trossulus and her female descendants.
The pattern of occurrence of these species suggests that M. trossulus is a cold-temperate species native to the northern Pacific, and that M. galloprovincialis is a warm-temperate species native to the Mediterranean and introduced to California, Japan, China and South Africa (Koehn, 1991; Seed, 1992), as well as Australia, Tasmania and New Zealand. DNA analysis of museum specimens indicates that M. galloprovincialis arrived in southern California between 1900 and 1947, probably as ship fouling or as larvae in ballast water, displacing M. trossulus (J. Geller in Culotta, 1995). DNA analysis also shows that viable M. galloprovincialis larvae are continually discharged in large numbers into Coos Bay, Oregon in the ballast water from Japanese ships, though no adult M. galloprovincialis or hybrids were found in the bay (Geller et al., 1994).
In San Francisco Bay, bay mussels are found mainly from the northern South Bay to southern San Pablo Bay, and occasionally as far upstream as Martinez (Hopkins, 1986). Distribution of M. trossulus and galloprovincialis at four sites as indicated by allozyme frequencies show a heterogeneous mix of species and hybrids that follows no obvious environmental cline, with M. trossulus strongly dominating at both the most upstream and most seaward site, and M. galloprovincialis less strongly dominating at sites between (Sarver & Foltz, 1993).
On the Pacific coast these two difficult-to-distinguish species
have long been considered one species and have been frequently
used for the biomonitoring of pollutants in the California Mussel
Watch program and other studies. Recent indications that separate
species in the Mytilus "edulis" complex exhibit
different growth rates and different concentrations of various
elements when grown in the same habitat (Lobel et al., 1990) suggest
that conclusions about the relative contamination of various sites
based on comparative bioassays of bay mussel specimens incorrectly
assumed to belong to a single species may be invalid. Other studies
have found different species within the complex to have different
levels of infection by parasites, spawning periods, fecundity
and strength of byssal attachment (Seed, 1992).
Petricolaria pholadiformis (Lamarck, 1818) [PETRICOLIDAE]
FALSE ANGELWING
SYNONYMS: Petricola pholadiformis
The false angelwing is native to the northwestern Atlantic, ranging from the Gulf of St. Lawrence to the Gulf of Mexico and possibly to Uruguay, and has been introduced to Europe (Carlton, 1979a, p. 515). It was collected in south San Francisco Bay in or before 1927 (Grant & Gale, 1931), from Willapa Bay in 1943 (Kincaid, 1947) and from Newport Bay in 1972. Reports of P. pholadiformis from "near Monterey" and from Scammons Lagoon, Baja California are probably erroneous (Carlton, 1979a). It is a borer into clay, peat, mud, sand and other soft sediments, and has been recorded from oyster beds on the Atlantic coast (Wells, 1961). Though it was most likely introduced to the Pacific in shipments of Atlantic oysters, it is puzzling that it was reported from the Pacific relatively late. It is a striking shell that would not likely have been overlooked by collectors. It is possibly an early ballast water introduction.
In Willapa Bay a spionid polychaete, a Corophium amphipod
and a nereid polychaete are often associated with P. pholadiformis.
In San Francisco Bay, Bush (1937) reported that about 90 percent
of these clams collected from sandy beaches near the Oakland Airport
host the ciliate Ancistrumina kofoidi. This protozoan is
known only from P. pholadiformis from San Francisco Bay,
and is presumed to be native to the Atlantic and introduced along
with the clam.
Potamocorbula amurensis (Schrenck, 1867) [CORBULIDAE]
AMUR RIVER CORBULA, ASIAN CLAM
In October 1986, a college biology class dredged three small and unfamiliar clams from the bottom of Suisun Bay. These were subsequently identified as Potamocorbula amurensis, a native of estuaries from southern China (22° N latitude) to southern Siberia (53° N) and Japan, which was likely transported to California as larvae in ballast water. By the summer of 1987 Potamocorbula had become the most abundant benthic organism in the northern part of the Bay, carpeting the bottom at densities of over 16,000 juvenile clams (mean shell length of 1.7 mm) per square meter (Carlton et al., 1990; Nichols et al., 1990). It seems likely that Potamocorbula arrived in the Bay very shortly before its discovery, because it was not collected earlier despite regular benthic sampling, and because all specimens collected through March 1987 were less than 11 mm long, and therefore probably less than a year old (Carlton et al., 1990).
An intensive benthic survey of the northern Bay in 1990 found Potamocorbula very common from San Pablo Bay through Suisun Bay, and most abundant in the Suisun Marsh region with mean concentrations of up to 19,200 clams/m2 and a median size of 2-3 mm. Median size was 10-11 mm in San Pablo Bay, and 5-6 mm and 8-9 mm in the shoals and channel of Suisun Bay (Hymanson, 1991). Potamocorbula is now abundant in parts of the South and Central Bay, and has occasionally been collected in the western Delta as far upstream as Rio Vista, over a range of salinities from 33 ppt to less than 1 ppt. At these sites it would be exposed to temperatures ranging from 8° C on subtidal bottoms in the winter to 23° C on intertidal flats in the summer, within the temperature range of 0-28° C suggested by its latitudinal range in Asia. It lives both subtidally and intertidally on all soft-bottom substrates, where it typically sits with one-third to one-half of its length exposed above the sediment surface (Carlton et al., 1990).
Prior to 1986, the benthic species composition and abundance in the northern Bay changed markedly from year to year, with freshwater species declining during dry periods and more numerous, higher-salinity speciesódominated by the clam Mya arenaria, the amphipods Corophium acherusicum and Ampelisca abdita, and the polychaete Streblospio benedicti, all introduced organismsóinvading the area (Nichols, 1985). Potamocorbula's arrival in the Bay followed a major flood in the spring of 1986, and its increase and spread coincided with a multi-year dry period that began in mid-1986. The 1986 flood left the benthic community nearly depauperate in the Suisun Bay area, probably facilitating Potamocorbula's establishment. This community failed to return during the subsequent dry period, presumably due to Potamocorbula's presence. The mechanisms by which Potamocorbula excluded these organisms are not known, but could include the depletion of food resources (see below) or feeding by Potamocorbula on the larvae of these organisms (Nichols et al., 1990). Potamocorbula has maintained substantial populations in the northern Bay even after the end of the drought and the return of normal flows (J. Thompson, pers. comm., 1994), and thus appears to have permanently changed benthic community dynamics in this part of the Bay (Nichols et al., 1990).
Examination of feces from specimens collected in the Bay show Potamocorbula ingesting both planktonic (Coscinodiscus spp. and Skeletonema costatum) and benthic (Navicula spp.) diatoms (Carlton et al., 1990). Werner & Hollibaugh (1993) found that Potamocorbula filters bacterioplankton as well as phytoplankton, though at lower efficiency, and assimilates both with high efficiency. They calculate that at present densities in the northern Bay (>2,000 clams/m2) Potamocorbula could filter the entire water column over the channels more than once per day and over the shallows almost 13 times per day, a rate of filtration which exceeds the phytoplankton's specific growth rate and approaches or exceeds the bacterioplankton's specific growth rate. Thus Potamocorbula may permanently reduce the phytoplankton standing stock in the northern reach of the Bay. Alpine & Cloern (1992) described the pre-Potamocorbula regime as one in which phytoplankton biomass and production were regulated by river-driven transport when benthic grazers were few, but limited by grazing pressure when grazers were abundant. With Potamocorbula in the Bay, grazing pressure may be permanently high, and phytoplankton biomass and productivity permanently low.
In laboratory experiments Kimmerer (1991) found that Potamocorbula readily consumed nauplii of the copepod Eurytemora affinis, but not the introduced copepod Pseudodiaptomus sp. Kimmerer et al. (1994) argued that an observed decline in the abundance of three dominant copepod taxaóE. affinis, Sinocalanus doerrii, and Acartia spp.óthat coincided with the spread of Potamocorbula in the northern reach of the Bay resulted from direct predation on copepods by Potamocorbula rather than from food limitation due to the decline in phytoplankton.
Further trophic changes may be expected to result from the reduction in zooplankton and the build-up of Potamocorbula, including declines in the organisms that feed on zooplankton, and increases in organisms capable of feeding on Potamocorbula (Carlton et al., 1990). Potamocorbula has been found in the stomachs of diving ducks and sturgeon in the Bay (Nichols et al., 1990), and in aquaria is readily consumed by the introduced green crab Carcinus maenas (Cohen et al., 1995).
Investigating allele frequencies at eight loci, Duda (1994) found
high genetic diversity in the San Francisco Bay population (polymorphic
at 75 percent of sites with a mean direct-count heterozygosity
of 0.295), with little genetic differentiation between sites within
the Bay.
Teredo navalis Linnaeus, 1758 [TEREDINIDAE]
NAVAL SHIPWORM
SYNONYMS: Teredo beachi Bartsch, 1921
Teredo diegensis (in part)
Teredo japonica Clessin, 1893
other synonyms are reviewed by Turner (1966), and the history
of taxonomic debate regarding San Francisco Bay shipworms is reviewed
by Carlton (1979a, pp. 558-560)
The earliest northwest Pacific record of this globally-distributed, temperate-water shipworm is from San Francisco Bay in 1913, and it has also established populations in Willapa Bay, Washington (first reported in 1957), in Pendrell Sound, British Columbia (1963), and possibly in Los Angeles Harbor (1927) and other southern California bays (Barrows, 1917; Kofoid & Miller, 1927; Reish, 1972; Carlton, 1979a, p. 556). It undoubtedly arrived in the hulls of ships.
When Commodore John Sloat arrived on the Pacific coast in 1852 in search of a suitable location for the Navy Department's western shipyard, his orders directed him to pick a site that was "safe from attack by wind, wave, enemies, and marine worms" (Lott, 1954). He chose the eastern shore of Mare Island in the northern, upstream reach of San Francisco Bay, where low salinities kept the region free of marine wood-boring organisms and where marine facilities such as wharves and ferry slips could consequently be built on untreated wooden pilings. It was in such wooden structures at Mare Island that Teredo navalis, which readily tolerated much fresher water than did the existing marine borers in the Bay (thriving down to 9 ppt and surviving indefinitely down to 5 ppt; Miller, 1926), was first noticed in 1913. By 1919-1920, possibly aided by a dry spell that brought higher than average salinities, Teredo navalis was found from the South Bay to Suisun Bay and had grown so abundant as to destroy virtually all the wooden structures in the northern part of the Bay, with damage estimated at over half a billion dollars in current dollars (McNeily, 1927; this paper, Chapter 6).
This destruction led to the formation of the San Francisco Bay Marine Piling Committee which produced a series of reports (annual reports in 1921, 1922 and 1923, and the Final Report in 1927) covering the activities and management of a variety of marine wood-borers in San Francisco Bay and elsewhere in the Pacific. The participants in the Committee's investigations later published several additional papers on the biology and morphology of Teredo navalis (references in Carlton, 1979a).
The evidence that Teredo navalis is not native to San Francisco Bay is reviewed by Barrows (1917, p. 29), Kofoid (1921, pp. 43-44), Kofoid & Miller (1922, pp. 81-82; 1927, pp. 206-207, 246-247) and Carlton (1979a, pp. 560-563). This evidence includes the absence of any known damage from marine borers in the northern part of the Bay prior to 1913, the lack of any prior record of Teredo navalis on the Pacific coast despite extensive collecting by nineteenth century conchologists, and the failure to find Teredo navalis in an investigation of shipworms conducted for the United States Forest Service in 1910-1911.
Although the specific source of the shipworms introduced to San Francisco Bay is unknown, Carlton (1979a) suggests that Teredo navalis is native to the Atlantic. A shipworm, probably Teredo navalis but possibly Nototeredo norvegica (Turner, 1966), was known from Europe since at least the start of the 17th century and was apparently mentioned by Pliny, Cicero, Theophrastus and others in ancient times (Moll, 1914). Teredo navalis was reported from Europe in 1731 by a Dutch commission describing a "horrible plague" of shipworms threatening to destroy the dikes that protected the lowlands of Holland, and by Sellius in 1733. Teredo navalis was also present in Japan at least since the 1890s, though it appears to have been absent from Australia at that time (Carlton, 1979a).
Although there has been little notice taken of shipworms in San
Francisco Bay in recent years, New York City has apparently experience
a resurgence of shipworm activity reportedly resulting from a
cleaner harbor (or, less likely, from shipworms developing a tolerance
to creosote). When city officials visited the Brooklyn Army Terminal
in the spring of 1993 to inspect shipworm damage they found that
one of the piers had collapsed the previous night. The city spent
$100 million to protect its piers against woodborer damage (Gruson,
1993).
Theora fragilis A. Adams, 1855 [SEMELIDAE]
ASIAN SEMELE
SYNONYMS: Theora lubrica Gould, 1861
Theora fragilis is a small, mud-dwelling clam native to Japan, China, the Indo-West Pacific and New Zealand. It first appeared in the northeastern Pacific in southern California, where it was collected from Anaheim Bay in 1968-69, from Newport Bay in 1971-73, and in large numbers from Los Angeles Harbor in 1973 (Seapy, 1974, Carlton, 1979a, p. 517). It was probably introduced in ballast water, possibly from ships returning from Southeast Asia during the Vietnam War. Theora fragilis larvae have been collected from the ballast water of Japanese cargo ships arriving at Coos Bay, Oregon and reared to juvenile stages (Carlton et al., 1990, p. 85).
Theora was first collected in San Francisco Bay in 1982
at Islais Creek, San Francisco (Carlton et al., 1990). It occurs
in small numbers through much of the Bay, the California Department
of Water Resources has collected it at Point Pinole at densities
of up to 127/m2 since sampling began in 1991
(DWR, 1995), and it was one of the most common benthic organisms
collected at the Alameda Naval Air Station in 1993 (G. Gillingham,
pers, comm.). It is absent from Suisun Bay according to U. S.
Geological Survey sampling records (Carlton et al., 1990).
Venerupis philippinarum (Adams & Reeve, 1850) [VENERIDAE]
JAPANESE LITTLENECK CLAM, MANILA CLAM
SYNONYMS: Tapes japonica (Deshayes, 1853)
Tapes semidecussata Reeve, 1864
Tapes philippinarum
Ruditapes philippinarum
Paphia bifurcata Quayle, 1938
Venerupis philippinarum, known until recently as Tapes japonica, is an Asian clam that was introduced with shipments of Japanese oysters to the northeastern Pacific, where it has become established in numerous bays from British Columbia to central California and is the numerically dominant clam in many of them. It was first noticed in planted oyster beds in Samish Bay, Washington in 1924 (Kincaid, 1947), and in a shipment of Japanese oysters arriving at Elkhorn Slough in 1930 (Bonnot, 1935b). However, the first record of an established population on the North American coast is from Ladysmith Harbor on the eastern shore of Vancouver Island, British Columbia in 1936 (Quayle, 1938). Northward spread from that site, and later northward spread from Barkley Sound on the west side of Vancouver Island to Venerupis' northernmost record in Hecate Strait, appear to have been due to the transport of larvae by currents, but the clam's spread southward to California is probably due in large part to new introductions in oyster shipments from Japan, to the transplanting of oysters along the coast, and to intentional transplants (some probably not recorded) of Venerupis.
Venerupis was found in Puget Sound in 1943, in Willapa Bay and San Francisco Bay in 1946, in Bodega Harbor and Elkhorn Slough in 1949, in Tomales Bay in 1955, in Humboldt Bay and Grays Harbor in 1964, and in Bolinas Lagoon in 1966. It had entered the commercial market by 1941, which encouraged laboratory aquaculture efforts and reseeding and replanting programs in the Pacific northwest, some of which continue. Efforts were made to establish Venerupis in Morro Bay, Newport Harbor and the Salton Sea in 1953, in the Queen Charlotte Islands in 1962, and in Yaquina and Tillamook bays in 1965, all of which failed. However, it was successfully established in Netarts Bay, Oregon in the 1970s (Carlton, 1979a, p. 502).
In San Francisco Bay, Venerupis is commonly found at concentrations up to 2,000 clams/m2 from the South Bay through San Pablo Bay, where it is one of the most common benthic organisms, and has on occasion been found as far upstream as Chipps Island (Nichols & Thompson, 1985a; Hopkins, 1986). In the Bay it is collected noncommercially both for food and bait (Sutton, 1981; ANC, pers. obs.).
In San Francisco Bay and elsewhere, Venerupis co-occurs with various native clams, including the similar native littleneck clam Protothaca staminea. Although a few authors have stated that Venerupis displaces the native littleneck, others have seen little evidence of competition between them, with Venerupis living higher in the intertidal zone or closer to the surface than Protothaca (see Carlton, 1979a). However, the question has not been effectively studied.
A variety of organisms feed on Venerupis on the Pacific
coast, including the moonsnail Polinices lewisii, sturgeon,
willet, gulls, ducks and raccoons (Glude, 1964; Painter, 1966;
McKechnie & Fenner, 1971; Stenzel et al., 1976; Carlton, 1979a),
and undoubtedly many others.
Eusarsiella zostericola (Cushman, 1906)
SYNONYMS: Sarsiella zostericola
Sarsiella tricostata Jones, 1958
This western Atlantic ostracod occurs from Maine to Florida and in the Gulf of Mexico. It is known on the Pacific coast only from San Francisco Bay, where it was first collected in 1953 at Point Richmond (Carlton, 1979a, p. 573). It is widely distributed in the Bay on soft substrates in shallow water. It has also been introduced to England, where it occurs only in regions where Atlantic oysters were planted. Though not recorded from San Francisco Bay until the 1950s, this minute, benthic crustacean could have been long present but gone unnoticed or unrecognized, and thus may have been introduced with Atlantic oyster shipments. Since ostracods (other than holoplanktonic ostracods) have rarely been collected from ballast water samples (e. g. Carlton & Geller, 1993), ballast water seems a less likely transport mechanism.
Acartiella sinensis
This copepod, native to the subtropical to tropical waters of
the China coast, was collected in Suisun Bay in 1993, 1994 and
1995. It is found in the vicinity of the entrapment zone and does
not extend upstream as far as the eastern Delta (Orsi, 1994, 1995;
J. Orsi, pers. comm., 1995). It was probably introduced in ballast
water.
Limnoithona sinensis (Burkhardt, 1912)
SYNONYMS: Oithona sinensis
This copepod has been collected from the brackish and fresh waters of the Yangtze River (Changjiang) inland to at least 300 km and from nearby lakes and canals in 1898, in 1906 and prior to 1962. It was collected from the San Francisco Estuary for first time in 1979, by CDFG from the San Joaquin River near Stockton (Ferrari & Orsi, 1984). Herbold & Moyle (1989) suggest that a decline in zooplankton abundance in the Delta prior to 1979 may have facilitated L. sinensis' establishment. It has been collected throughout the Delta (where it is more abundant in the San-Joaquin than in the Sacramento River) and downstream to Suisun Bay, though apparently restricted to waters of less than 1.2 ppt (Herbold & Moyle, 1989). It has been most abundant in Oct./Nov. and scarcest in Mar./Apr., with a maximum recorded abundance of 71,176 individuals/m2 in Aug., 1981 near Stockton (Ferrari & Orsi, 1984). In 1993-94 it was replaced over its entire range by Limnoithona tetraspina (J. Orsi, pers. comm., 1995).
The lack of any record of this copepod in the eastern Pacific
prior to 1979, and early records of it from the Yangtze River
area, suggest that L. sinensis is a recent introduction
to the San Francisco Estuary (Ferrari & Orsi, 1984). It was
most likely transported across the Pacific in ballast water (oithonid
copepods have been found to survive transport in ballast tanks;
Carlton, 1985, p. 346).
Limnoithona tetraspina
This copepod, native to the Yangtze River, was first found in
the Estuary in 1993 at Chipps Island in Suisun Bay and at Collinsville
and Hood on the Sacramento River. By 1994 it had replaced Limnoithona
sinensis and, reaching densities greater than 40,000/m3,
had become the most abundant copepod ever seen in the Estuary
(Orsi, 1995; J. Orsi, pers. comm., 1995). It was probably introduced
in ballast water.
Mytilicola orientalis Mori, 1935
PARASITIC COPEPOD
SYNONYMS: Mytilicola ostreae Wilson, 1938
This small red copepod lives in the intestine or rectum, or rarely in the digestive diverticulae, of oysters and other mollusks. It is native to the western Pacific and was introduced to the northeastern Pacific with shipments of the Japanese oyster Crassostrea gigas. It was first collected from Willapa Bay, Washington in 1938, and subsequently from many bays and estuaries from Vancouver Island, British Columbia to Morro Bay, California, including San Francisco Bay in 1974 (where it was discovered in three out of 30 native oysters Ostrea conchaphila from the Berkeley Marina; Bradley & Siebert, 1978; Carlton, 1979a, p. 577). These various sites could have received Mytilicola directly with shipments of oysters from Japan, with oysters transplanted from other eastern Pacific bays, or with mussels fouling coastal ships.
On the Pacific coast Mytilicola has been found in (in addition to Japanese oysters) the introduced slipper shell Crepidula fornicata (one record from Puget Sound), and several native bivalves, including the oyster Ostrea conchaphila, the mussel Mytilus californianus, and the clams Protothaca staminea (one record from Puget Sound), Saxidomus giganteus and Clinocardium nuttallii (one record each from British Columbia). It has also been found in the native mussel Mytilus trossulus (northern records reported as M. edulis) and possibly the introduced mussel M. galloprovincialis or in hybrids (San Francisco Bay record reported as M. edulis; see Sarver & Folz, 1993) (Carlton, 1979a).
Carlton (1979a) notes that the data for sites and for hosts may be selective as "all bays that have been searched, and most if not all mollusks that have been examined, have been found to have Mytilicola." He also notes that due to the copepod's endoparasitic habit and a lack of exploration and early collecting, Mytilicola could have been in these bays long before it was first observed.
Katansky et al. (1967) and Bradley & Siebert (1978) summarize
the biological research on Mytilicola in the eastern Pacific.
Oithona davisae Ferrari & Orsi, 1984
This copepod was first collected in eastern Suisun Bay in 1979, and described by Ferrari & Orsi (1984). It has been collected from the South Bay to San Pablo Bay, and upstream to Chipps Island in waters of 12 ppt. Copepods that were collected from San Pablo Bay in the winter, spring and fall of 1963 and identified as Oithona sp. may also have been Oithona davisae (Ferrari & Orsi, 1984).
Ambler et al. (1985) found Oithona davisae to be one of the most common copepods in the Bay in 1980. In June to December of that year, at sites from the South Bay to Carquinez Strait it was found in 25-48 percent of the samples collected, and reached peak abundances of 22,000-44,000 individuals/m2 in the South Bay in October and November.
Ferrari & Orsi (1984) argued that the lack of any record of
this copepod in the Bay prior to 1979, and the fact that some
distinctive morphological characters are shared exclusively with
Indo-West Pacific oithonid copepods, suggests that Oithona
davisae was a recent introduction to the San Francisco Estuary
from the western Pacific. It was subsequently found in Japanese
waters, where it is frequently abundant in eutrophic embayments
(Uye & Sano, 1995), and considered to be of Asian origin (Fleminger
& Kramer, 1988). It has also been reported from southern Chile
(Carlton, 1987). Oithona species have been found to survive
transport in ballast tanks (Carlton, 1985, p. 346), and this one
was most likely transported across the Pacific in ballast water.
Pseudodiaptomus forbesi (Poppe & Richard, 1890)
Pseudodiaptomus forbesi is native to the fresh and brackish waters of the Yangtze River (Changjiang), China, usually restricted to waters of less than 8 ppt. It was first collected outside of China in 1987 in fresh water in the eastern and southern Delta. By the following year it was found throughout the Delta and downstream into Suisun Bay up to a salinity of 16 ppt, in which areas it was the most abundant calanoid copepod in the fall of 1988 and in 1989. The maximum abundance recorded was 22,408 individuals/m2 in fresh water in the San Joaquin River near Stockton in early June, 1988 (Orsi, 1989; Orsi & Walter, 1991).
Various hypotheses have been proposed to explain the recent dramatic shifts in the absolute and relative abundance of Pseudodiaptomus forbesi and other copepods in the northern reach of the Estuary, including competition between native and introduced copepods, differential predation by introduced fish and clams on different copepods, and predation by copepods on other copepods. Herbold et al. (1992), implying competition as the relevant mechanism, reported that the "invasions of the western Delta and Suisun Bay by Sinocalanus doerrii in 1978 and by Pseudodiaptomus forbesi in 1987 were followed by declines in abundance of Eurytemora affinis and the almost complete elimination of Diaptomus spp." On the other hand, Kimmerer (1991) reported that the cryptogenic copepod Eurytemora affinis was not food-limited in the Estuary so that competition with recently introduced copepods could not account for its decline.
Orsi (1989) noted that striped bass appeared to be more effective
predators on Eurytemora than on P. forbesi, and
Meng & Orsi (1991) found that striped bass larvae in laboratory
feeding experiments selected native copepods Cyclops sp.
and cryptogenic Eurytemora (present in the Estuary since
at least the 1912-13 Albatross survey; Esterly, 1924) over
the recently introduced copepods P. forbesi and Sinocalanus
doerri, and suggested that differences in copepod swimming
and escape behaviors could account for the differential predation.
Kimmerer (1991) reported that in laboratory experiments the introduced
Asian clam Potamocorbula amurensis consumed Eurytemora
but not Pseudodiaptomus species, and Kimmerer et al.
(1994) argued that the decline in Eurytemora was caused
by Potamocorbula preying on its nauplii. Orsi (1995) suggested
that, in addition to predation by Potamocorbula, the decline
may have been partly due to competition with P. forbesi,
noting that Eurytemora continues to be seasonally present
in winter and spring when P. forbesi is scarce, both within
and upstream of Potamocorbula's range. Orsi (1995) also
suggested that predation by the introduced copepod Tortanus
sp. may account for a decline in Pseudodiaptomus in
western Suisun Bay in 1994.
Pseudodiaptomus marinus (Sato, 1913)
Pseudodiaptomus marinus is native to China, Japan and Pacific Russia, and has been introduced to Hawaii and Mauritius (Jones, 1966; Grindley & Grice, 1969; Orsi et al., 1983). It was collected north of San Diego in Mission Bay in 1986 and in Aqua Hedionda Lagoon in May 1987 (Fleminger & Kramer, 1988). It was first collected in the San Francisco Estuary from western Suisun Bay in 1986, and has been collected from there upstream to Collinsville on the Sacramento River, in waters with surface salinities ranging from about 2 to 18 ppt. It has also been collected from Tomales Bay (Orsi & Walter, 1991).
Pseudodiaptomus marinus may have been introduced to San
Francisco Bay in ballast water, to the southern California bays
or Tomales Bay in oyster shipments, and moved between bays by
coastal currents (Fleminger & Kramer, 1988; Orsi & Walter,
1991). Fleminger & Kramer (1988) suggested that the native
copepod P. euryhalinus may have been displaced by P.
marinus in southern California embayments, and called for
more sampling to determine whether P. euryhalinus was in
fact absent or confined to sites where P. marinus had not
become established.
Sinocalanus doerrii (Brehm, 1909)
SYNONYMS: Sinocalanus mystrophorus Burckhardt, 1913
This calanoid copepod is native to the rivers of mainland China, and like the other pelagic copepods described here was probably introduced in ballast water. It was first collected from the Estuary near Pittsburg in 1978 and soon became (from 1979 to the early 1980s) the most abundant copepod in the Delta, with maximum densities of over 10,000 individuals/m2 and greatest densities from June to September. It has been collected from throughout the Delta upstream to Hood on the Sacramento River and Stockton on the San Joaquin River, and downstream to San Pablo Bay, generally at salinities below 5 or 6 ppt but on occasion up to nearly 15 ppt. Its downstream limit may be regulated by both salinity and the location of the entrapment zone (Orsi et al., 1983; Ambler et al., 1985; Herbold & Moyle, 1989; Orsi, 1995). It was not collected in 1994, but reappeared in 1995 (J. Orsi, pers. comm., 1995).
Five species are recognized in the genus Sinocalanus, all from the northwestern Pacific. As S. doerrii had not been collected in regular plankton surveys in the Estuary in 1963 and from 1972-78, it was probably introduced shortly before 1978 via ballast water (Orsi et al., 1983). Orsi et al. suggest, based on the apparent pattern of spread in 1978-79, that the site of introduction was in the Pittsburg-Antioch area near where S. doerrii was first collected. They further suggest that water pumped out of the Delta into the California Aqueduct will carry S. doerrii to water project reservoirs near Los Angeles, and that the Columbia River and Puget Sound are likely sites for secondary introductions via the ballast water carried by coastal ships.
Several researchers have considered interactions between Sinocalanus doerrii and other copepods in the northern estuary (some of which are discussed above under Pseudodiaptomus forbesi). Orsi et al. (1983) noted that competition between Sinocalanus and the cryptogenic copepod Eurytemora affinis was unlikely because their preferred salinity ranges differed, and suggested that competition and/or predation between Sinocalanus and the freshwater copepods Cyclops and Diaptomus was a stronger possibility and should be investigated. Ambler et al. (1985) questioned whether there is competition for food, at least in years with average river discharge and diatom blooms in Suisun Bay. Meng & Orsi (1991) found that striped bass larvae in laboratory feeding experiments selected Cyclops sp. and Eurytemora over Sinocalanus.
Herbold et al. (1992) reported that the introduction of Sinocalanus
and of Pseudodiaptomus forbesi in 1987 was followed
by declines in Eurytemora and the almost complete elimination
of Diaptomus spp., although Herbold & Moyle (1989)
had earlier suggested that declines in Delta zooplankton prior
to 1979 may have facilitated Sinocalanus' establishment.
Kimmerer (1991) reported laboratory studies indicating that although
Sinocalanus may be food limited in the estuary in some
years, Eurytemora is not and so competition with recently
introduced copepods could not account for Eurytemora's
decline. Orsi (1995) suggested that Sinocalanus had "apparently
slipped into an unoccupied niche" between Eurytemora downstream
and Diaptomus species upstream in the San Joaquin River,
but noted that Diaptomus abundance fell when Sinocalanus
spread upstream. Herbold & Moyle (1989) had noted that the
invasion of the Sacramento River by Sinocalanus coincided
with a reduction in the relative abundance of chlorophyll in the
north Delta.
Tortanus sp.
This large calanoid copepod of unknown origin was collected in
Suisun Bay in the fall of 1993 and in 1994 (Orsi, 1994, 1995;
J. Orsi, pers. comm., 1995). It preys on other copepods and Orsi
(1995) suggests that it may have caused a decline in Pseudodiaptomus
in western Suisun Bay in 1994. Its prior absence in this well-studied
region of the Bay suggests that it was introduced in ballast water.
Balanus amphitrite Darwin, 1854
STRIPED BARNACLE
SYNONYMS: Balanus amphitrite amphitrite Darwin, 1854
Balanus amphitrite hawaiiensis Broch, 1922
Balanus amphitrite denticulata Broch, 1927
Balanus amphitrite herzi Rogers, 1949
Balanus amphitrite franciscanus Rogers, 1949
Balanus amphitrite saltonensis Rogers, 1949
This subtropical and warm-temperate barnacle is native to the Indian Ocean but has been distributed widely. In perhaps the earliest scientific recognition of the phenomenon of marine introductions, Darwin (1854, pp. 162-163) noted that Balanus amphitrite, B. improvisus and a few other barnacles "which seem to range over nearly the whole world (excepting the colder seas)" may have been transported to parts of their reported range as fouling on ships.
B. amphitrite was collected in Hawaii in the early 1900s. In California it was found in La Jolla in 1921, in San Diego in 1927, in San Francisco Bay in 1938-39, and in the Los Angeles/Long Beach area in 1940 (Zullo et al., 1972; Carlton, 1979a, p. 585). In 1945 it was found in the Salton Sea, probably introduced from San Diego Bay attached to "navy planes, boats, buoys, ropes, or other marine equipment that was transferred in large quantity to the sea for training purposes" (Carlton, 1979a). It was first collected from the Gulf of California and the west coast of Mexico in 1946, and appeared on the Atlantic coast of North America after World War II.
Although Balanus amphitrite tolerates water temperatures
down to 12°C it requires at least 18°C to breed. It
may thus be restricted to warmer sites within San Francisco Bay,
where it has been collected from scattered locations in the northern
South Bay, Central Bay and San Pablo Bay (Newman, 1967). In Britain
and the Netherlands it lives in areas heated by the outflow from
power plants (Vaas, 1978; Carlton, 1979a).
Balanus improvisus Darwin, 1854
BAY BARNACLE
Balanus improvisus, a native of the North Atlantic, is the most freshwater-tolerant of the barnacles and has been widely introduced around the world. It is also the earliest known introduction to San Francisco Bay, having been identified from a mussel shell in U. C. Berkeley's Museum of Paleontology that was collected from the harbor of San Francisco in 1853 (Carlton & Zullo, 1969). This early introduction was probably the result of transport as fouling on ship hulls.
B. improvisus is next known in San Francisco Bay from specimens on the shell of an Atlantic oyster, Crassostrea virginica, collected at San Mateo in 1900, and the barnacle then appears in collections from every decade of the twentieth century, often on oyster or mussel shells (Carlton & Zullo, 1969). A second introduction (and possibly additional introductions) of B. improvisus, with shipments of Atlantic oysters that began in 1869 thus seems possible. It is not known whether the 1850s population, introduced by shipping, persisted or died out.
B. improvisus was collected from Monterey Bay in 1916, from the Los Angeles/Long Beach area in 1932, and from San Simeon Point and San Diego in 1939. Despite these records from the 1930s, B. improvisus does not appear to be established in southern California. There are other reports from the tropical or subtropical Pacific, though actual collections are few: the Gulf of California in 1889, 1941 and 1967; the west coast of Mexico in 1960-1968; Colombia in 1854; Ecuador in 1854, 1934, 1963 and 1966; and Peru in 1926. The identification of some of these populations as Balanus improvisus may bear reexamination.
B. improvisus is likely established in bays to the north of San Francisco Bay, perhaps in some from which it has not yet been reported. It was collected from Vancouver Island and Willapa Bay in 1955, from the Columbia River in 1957 (on the shell of the crayfish Pacifastacus trowbridgii), and from Coos Bay in 1978. Since World War II, it has also been reported from Japan, Singapore and Australia (Carlton, 1979a).
In San Francisco Bay its physiology and behavior were investigated
by Newman (1967) who found that it tolerated dilution to 3 percent
seawater, and that, surprisingly, it was an osmo-conformer with
its blood remaining nearly isotonic with its environment. It is
the only barnacle found upstream of Carquinez Strait in the northern
part of the estuary. At Antioch it lives in freshwater for ten
months of the year. A population was found in December 1962 living
on the concrete walls of the Delta Mendota Canal in essentially
fresh water, although there is no evidence that barnacles in the
canal reproduce successfully (Zullo et al., 1972).
Epinebalia sp.
This unidentified nebaliid was collected on muddy bottom by John
Chapman in Aquatic Park Lagoon in Berkeley in 1992, and we found
it common at Richmond in 1993 and Lake Merritt in 1993 and 1994.
G. Gillingham (pers. comm., 1995) reports "Nebalia pugettensis"
collected at the Alameda Naval Air Station in the spring of 1993.
The prior absence of reports of any nebaliid from San Francisco
Bay, and specifically the absence of a nebaliid from the East
Bay shore in the 1960s-1970s, suggests that all these specimens
are an introduced nebaliid rather than the native N. pugettensis.
Although largely benthic organisms, nebaliids could easily be
transported by ballast water in suspended sediments swept up from
the bottom while the ship is ballasting.
Acanthomysis aspera Ii, 1964
This planktonic Japanese mysid was found in the northern part
of the San Francisco Estuary in 1992 and was still present, though
not abundant in 1993-94. It was probably introduced in ballast
water (T. W. Bowman, in litt. to J. J. Orsi; Orsi, 1994, 1995).
Acanthomysis sp.
An undescribed species of Acanthomysis, resembling A.
sinensis (T. W. Bowman, in litt. 23 Mar. 1994 to J. J. Orsi),
was collected in Suisun Bay in 1992, and was more abundant than
the common native opossum shrimp Neomysis mercedis by 1994
(J. Orsi, pers. comm., 1995). Because its morphology resembles
that of western Pacific mysids and is unlike that of eastern Pacific
species, it is probably native to the western Pacific and was
transported to California in ballast water (Orsi, 1994; T. W.
Bowman, in litt.).
Deltamysis holmquistae Bowman & Orsi, 1992
Deltamysis holmquistae was first collected and described from the San Francisco Estuary in 1977. Bowman & Orsi (1992) report that it has been collected every year since, ranging from one specimen in 1984 to 39 in 1987. Most were collected from Carquinez Strait to the Delta, with one taken in San Pablo Bay during the high spring outflow of 1983. They were found mainly in salinities of 1-2 ppt at the upstream edge of the entrapment zone, but ranged from 0-19 ppt.
Deltamysis is in the tribe Heteromysini along with mysids
that are commensal or epibenthic, or that swim among sea grass
plants, and this could account for the small numbers of Deltamysis
collected in open water trawls. That Deltamysis was not
collected until 1977 despite sampling for mysids since 1963, and
that it has been collected regularly if sparsely since 1977, strongly
suggests that it is introduced, probably in ballast water. There
are no known mysid species that closely resemble it (Bowman &
Orsi, 1992), but targeted searches in western Pacific estuaries
that are the origin of other recent zooplankton introductions
could be fruitful.
Nippoleucon hinumensis (Gamo, 1967)
SYNONYMS: Hemileucon hinumensis
This cumacean is native to Japan and was introduced to the northeast
Pacific in ballast water. The California Department of Water Resources
has collected it in San Francisco Bay in the western Delta and
Grizzly Bay since 1986, and at densities of hundreds or thousands/m2
(with a maximum of over 12,000/m2) it was
one of the three numerically dominant species in these areas from
1988 to 1990. It has also been collected at Pt. Pinole in San
Pablo Bay since sampling started there in 1991 (Hymanson et al.,
1994; DWR, 1995). We collected it from the Napa River, San Pablo
Bay and the South Bay in 1993-94. It was collected in Oregon from
Coos Bay in 1979, from the Umpqua River in 1983, from Yaquina
Bay in 1988, and from the Columbia River (J. Chapman, pers. comm.;
JTC, pers. obs.).
Dynoides dentisinus Shen, 1929
We collected this isopod, known previously from Japan and Korea,
in fouling from the Oakland Estuary in 1977 and from the Richmond
Marina in 1994. It was probably transported in ship fouling or
ballast water.
Eurylana arcuata (Hale, 1925)
SYNONYMS: Cirolana arcuata
Cirolana concinna Hale
Cirolana robusta Menzies, 1962
Eurylana arcuata was collected in San Francisco Bay on eight occasions in 1978 and 1979 from the cooling water intake screen of a power plant at Rodeo in San Pablo Bay, including brooding females and juveniles (Bowman et al., 1981). We collected it from floating docks on Coast Guard Island in the Oakland Estuary in 1993 and 1994.
Eurylana arcuata was first described from Australia, but
has not been reported from there since. It was reported from New
Zealand, where it is widespread and abundant, in 1961, and from
several distant sites in Chile (as Cirolana concinna and
C. robusta) since 1962. It is not known which of these
is its native region. It was likely introduced to San Francisco
Bay in fouling or ballast water (Bowman et al., 1981).
Iais californica (Richardson, 1904)
Iais californica is a small commensal isopod that is generally found clinging to the ventral surface of the introduced burrowing isopod Sphaeroma quoyanum. It was described from San Francisco Bay in 1904, but was presumably introduced along with Sphaeroma in ship fouling by 1893. Iais was reported from New Zealand and Australia in 1956. In California, Iais has been collected in most of the bays and harbors where Sphaeroma is found, and from none where Sphaeroma is absent (Carlton, 1979a). In 1995 we found it on Sphaeroma burrowing in floating docks on Isthmus Slough in Coos Bay.
Iais scavenges food from the mouthparts and the burrow
walls of its host, and is protected from predators and adverse
conditions both by Sphaeroma's burrow and Sphaeroma's
habit of curling into a ball when disturbed. Iais is occasionally
found on the native isopod Gnorimosphaeroma oregonensis
when the latter live in Sphaeroma burrows. Unlike Sphaeroma,
Gnorimosphaeroma will actively remove Iais (Rotramel,
1975b). These commensal relations have been studied by Rotramel
(1972, 1975b) and Schneider (1976).
Limnoria quadripunctata Holthuis, 1949 and Limnoria
tripunctata Menzies, 1951
GRIBBLE
Limnoria are small wood-boring isopods that are well-known for attacking and damaging ships' hulls, pilings and other wooden structures in contact with sea water (Kofoid, 1921; Hill & Kofoid, 1927). Many species of Limnoria have been described, some of them morphologically very similar. Some reported distributions are wide to circumglobal or strikingly disjunct, and undoubtedly complicated by centuries of transoceanic and interoceanic travel in the hulls of wooden ships.
Prior to the 1950s, all Limnoria on the Pacific coast were assigned to Limnoria lignorum, a species which is possibly native from Alaska to Humboldt County, but not known from San Francisco Bay. A Limnoria species was reported from Los Angeles in 1871 and San Diego in 1876 (Carlton, 1979). Limnoria was not mentioned in 1855, 1863 and 1869 reports on shipworm damage to pilings in San Francisco Bay (Ayres & Trask, 1855; Harris & Ayres, 1863; Neily, 1927), but was described as "recently appeared" on the San Francisco waterfront (probably L. quadripunctata, based on current distribution and thermal requirements) in 1873 (Arnold, 1873), and reported from the Oakland Estuary (probably L. tripunctata) in 1875 (Merritt, 1875). L. quadripunctata has since been collected from numerous embayments from La Jolla to Humboldt Bay, and L. tripunctata from Port Hueneme in Ventura County, California to Mexico, with the tripunctata population in the warm-water margins of San Francisco Bay remaining as an isolated northern outpost (Carlton, 1979). Carlton (1979) has argued that the Limnoria reported from northern Oregon, Washington and British Columbia as tripunctata (Quayle, 1964b) is probably a different species.
The native regions of L. quadripunctata and tripunctata
are not known. They were transported to the Pacific Coast
in the hulls of wooden ships, and dispersed along the coast in
ships' hulls, log booms, log shipments or drifting wood.
Paranthura sp.
In 1993 we collected a species of Paranthura that had not
previously been reported from San Francisco Bay (J. Chapman, pers.
comm., 1995). The isopod was very common in fouling on floating
docks from the South Bay and Central Bay and north to Richmond
in 1993 and 1994, but was not observed in 1995. Initial examination
suggests strong affinities with western Pacific species (J. Chapman,
pers. comm., 1995). Introduction has likely been by ship fouling
or ballast water.
Sphaeroma quoyanum Milne-Edwards, 1840
SYNONYMS: Sphaeroma pentodon Richardson, 1904
Sphaeroma is a burrowing, filter-feeding isopod native to New Zealand, Tasmania and Australia, and was collected in San Francisco Bay in 1893, probably having been introduced via ship fouling. It spread widely in California and was collected in Humboldt Bay, Tomales Bay, Los Angeles-Long Beach Harbors, and San Diego Bay in the late 1920s and early 1930s, and in several intervening bays and in San Quintin Bay, Baja California since the 1950s (Carlton, 1979a). In 1995 we found it burrowing in floating docks on Isthmus Slough in Coos Bay.
Sphaeroma is reported as common and frequently abundant
throughout San Francisco Bay at least as far upstream as Antioch
(Kofoid & Miller, 1927), though we did not find it on docks
in the seaward portion of the Central Bay. It burrows into all
types of soft substrate, including clay, peat, mud, sandstone
and soft or decaying wood, and wood that has been bored by shipworms
and gribbles. It is frequently found riddling the styrofoam floats
underneath docks, and is sometimes abundant in fouling accumulations.
Carlton (1979a,b) suggested that Sphaeroma's burrowing
could be responsible for substantial erosion of intertidal sediments,
which he estimated as possibly amounting to the loss of tens or
scores of meters of land along many kilometers of shoreline in
San Francisco Bay. However, no measurements of Sphaeroma's
topographic impact have ever been made. Studies of its biology
in central California include those of Barrows (1919), Rotramel
(1972, 1975a,b) and Schneider (1976).
Synidotea laevidorsalis (Miers, 1881)
SYNONYMS: Synidotea laticauda Benedict, 1897
Synidotea laticauda was described from San Francisco Bay oyster beds in 1897. It is commonly found in the Bay on the bottom and on buoys, floating docks and pilings among masses of the introduced Indo-Pacific hydroid Garveia franciscana (upon which it is thought to feed) and the introduced Atlantic bryozoan Conopeum tenuissimum (Carlton, 1979a). S. laticauda was long considered to be a native species restricted to the Bay, and its distribution and that of two other northern Pacific Synidotea species was explained by a model involving Pleistocene climate changes, range constrictions and expansions, isolation and evolution, and competition (Miller, 1968; Menzies & Miller, 1972).
Chapman & Carlton (1991, 1994) identified S. laticauda from Willapa Bay and synonymized S. laticauda with S. marplatensis and S. brunnea of eastern South America (where it was first collected in 1918) under the Asian name S. laevidorsalis. They concluded that the species is native to Asia and was transported to San Francisco Bay among hydroids and bryozoans fouling the hulls of ships (probably from China), transported by similar means to South America (probably from San Francisco Bay), and transported to Willapa Bay either from San Francisco (in ship fouling or with cargoes of the native oyster Ostrea conchaphila) or Asia (in ship fouling or with cargoes of the Japanese oyster Crassostrea gigas).
Synidotea laevidorsalis is reported to be a common benthic
organism from the far South Bay to Pittsburg in Suisun Bay, and
less common in the Central Bay and upstream to Antioch. It was
collected in both the shallows and the channels, at concentrations
typically up to 100/m2 (Hopkins, 1986; Markmann,
1986). In 1993-95 we found it common to abundant on floating docks
and buoys in San Pablo Bay and the Napa River. It is said to be
an important food of diving ducks and fish (Painter, 1966).
Sinelobus sp.
This abundant tanaid was first reported from San Francisco Bay by Miller (1968, as Tanais sp.) based upon material collected from a navigation buoy in San Pablo Bay in 1943, and later by Miller (1975, as Tanais sp., cf. T. vanis) and Carlton (1979a, as Tanais sp., cf. T. vanis, and 1979b, as Tanais sp.), based upon specimens collected in Lake Merritt, Oakland by Carlton commencing in 1963. Carlton (1979a) further reported specimens collected in 1965 from Corte Madera Creek in Marin County from the stomach of the native sculpin Cottus asper.
The only other records appear to be from Humboldt Bay (as Tanais sp.; S. Larned, pers. comm., 1989), and from several estuaries in British Columbia (as Tanais stanfordi; Levings & Rafi, 1978) where it occurred in densities up to 17,400 per 0.25 square meter in muddy sediments over a salinity range of 3.7 to 22.7 ppt, and in 7 out of 21 plankton tow stations. Levings & Rafi (1978) noted that there were no previous records of stanfordi from the west coast of North America.
Sieg (1980) and Sieg & Winn (1981) considered the report and figure of Miller (1968) to belong to Sinelobus stanfordi (Richardson, 1901). They further synonymized the earlier report of Menzies & Miller (1954) of a "Tanais sp." from central California with Sinelobus stanfordi, but that record is based on material collected on the outer rocky shore (Light, 1941, p. 92) and no doubt refers to a different species.
Sinelobus stanfordi was described from the Galapagos Islands, and has subsequently been reported from "Arctic cold, north Pacific temperate, southern temperate waters, tropical warm Pacific, tropical Indo-West Pacific, tropical Indian, and tropical warm Atlantic" waters (Sieg, 1986). Localities include Brazil, West Indies, the Mediterranean, Senegal, South Africa, Tuamotu Archipelago, and Hawaii, as well as the boreal Kurile Islands, and Holdich & Jones (1983) added England. Reported habitats include fresh, brackish, marine and hypersaline water.
Given this broad distribution, it is probable that a species complex is involved (including taxa which have been dispersed synanthropically), and we are hesitant to apply the name of a warm tropical tanaid described from the Galapagos Islands to the San Francisco Bay population. Though this population was earlier identified as Tanais vanis Miller, 1940, this is an algal-dwelling species of Hawaiian fringing coral reefs (Carlton, 1979a) and thus also not likely to be the species in San Francisco Bay.
This small crustacean is widespread throughout the estuarine margin of the Bay, and has been collected upstream at least as far as Chipps Island (Siegfried et al., 1980). It is replaced by the cryptogenic and more marine tanaid Leptochelia dubia in the middle and outer bay regions. In addition to the benthic habitat noted by Levings & Rafi (1978) in British Columbia, in San Francisco Bay it occurs commonly in fouling communities among masses of the introduced tubeworm Ficopomatus and lumbering along in intertwined mats of the green algae Ulva and Cladophora, often in association with the introduced amphipods Melita and Corophium. It occurs commonly in habitats where all other peracarids are introduced or cryptogenic.
We regard Sinelobus sp. of San Francisco Bay as introduced;
the origin of these populations remains unknown. Introduction
was possibly via ship fouling or ballast water.
Ampelisca abdita Mills, 1964
SYNONYMS: Ampelisca milleri of San Francisco Bay authors, not of Barnard, 1954
Ampelisa milleri of Dickinson, 1982 (Dillon Beach record)
Ampelisca abdita is native to northwest Atlantic from Maine to the eastern Gulf of Mexico. It was collected on the Pacific coast from San Francisco Bay in 1954, from Tomales Bay in 1969, and from Bolinas Lagoon in 1971 (Carlton, 1979a, p. 645; Chapman, 1988).
On the Atlantic coast, Ampelisca abdita often occurs in oyster beds and forms extensive mats of silt tubes which provides stable substrate for numerous other organisms. As A. abdita is a small amphipod, Chapman (1988) argues that it could have been present in the Bay for a long time before the 1950s and not been noticed due to a combination of the undeveloped taxonomy of small amphipods up to that time and the use of sieves with mesh openings of at least 1 mm (which retain few A. abdita) in early surveys. Thus it could have arrived with shipments of Atlantic oysters in the late nineteenth or early twentieth century. Since A. abdita sometimes migrates into the water column (Chapman, 1988), it could also have arrived later in ballast water.
Ampelisca abdita is now a very common and abundant benthic
organism in San Francisco Bay, recorded at virtually all sites
surveyed from far South Bay to Carquinez Strait, with concentrations
commonly of 1,000-50,000/square meter. It is less abundant in
western part of Central Bay, and less common and less abundant
in Suisun Bay, although collected upstream to Antioch (Hopkins,
1986). Its abundance varies annually, peaking around October,
although Ampelisca may be eliminated from large regions
of the Bay by floods, either because of salinity changes or sedimentation.
When abundant, it may interfere with the recruitment of Macoma
petalum (Nichols & Thompson, 1985a).
Ampithoe valida Smith, 1873
Ampithoe valida is native to the northwest Atlantic from New Hampshire to Chesapeake Bay (Bousfield, 1973). It has been collected on the central California coast from San Francisco and Tomales bays (first records in 1941), Morro Bay (1960), Bodega Harbor and Bolinas Lagoon (1975) (Carlton, 1979a, p. 649), and Humboldt Bay (S. Larned, pers. comm.). There are single records from Newport Bay in southern California (1942), Coos Bay, Oregon (1950) (Carlton, 1979a) and several other records from Oregon to southern British Columbia since the late 1960s (Conlan & Bousfield, 1982; Chapman, pers. comm.).
Ampithoe valida builds and lives in tubes on algae and
eelgrass, and has been found on oyster beds on the Atlantic coast.
It could have been introduced to San Francisco Bay with Atlantic
oyster shipments and remained undetected for decades, or arrived
in hull fouling or ballast water. In 1993-94 we collected it at
several stations in San Pablo Bay, at Coyote Point in the South
Bay, and at Pier 39 in San Francisco.
Caprella mutica Schurin, 1935
SYNONYMS: Caprella acanthogaster of Pacific coast authors (e.g., Carlton, 1979a, 1979b), not of Mayer, 1890
Caprella acanthogaster humboldtiensis Martin, 1977
SKELETON SHRIMP
This caprellid shrimp, a native of the Sea of Japan, has been
collected in Humboldt Bay (about 1973-77), San Francisco Bay (1976-1977),
Elkhorn Slough (1978-1979) and Coos Bay, Oregon (1983) (Martin,
1977; Marelli, 1981; JTC, unpublished). Marelli (1981) concluded
that Martin (1977) had incorrectly described this Japanese species
from Humboldt Bay as a new subspecies of Caprella acanthogaster
(which is a species distinct from C. mutica). It was reported
as comprising 40 percent of the caprellids at Field's Landing
in Humboldt Bay (Martin, 1977) and 90 percent of the caprellids
in the Oakland Estuary (D. Cross, pers. comm., 1977). Based on
its recent date of discovery on the Pacific coast, Caprella
mutica may have been introduced to Humboldt Bay with shipments
of Japanese oysters, which occurred from 1953 through the 1970s,
and secondarily introduced to San Francisco Bay; or it may have
been introduced to either or both bays in ballast water (Caprella
species have been found to survive transport in ballast tanks;
Carlton, 1985, p. 346).
Chelura terebrans Philippi, 1839
Chelura terebrans lives in burrows in wood in association with wood-boring isopods in the genus Limnoria, and reportedly feeds upon Limnoria's fecal pellets (Kühne & Becker, 1971). It has undoubtedly been transported around the world with Limnoria in the hulls of wooden ships. It is reported from the Atlantic on both the American and European coasts, the Mediterranean and Black seas, and from French West Africa and South Africa. In the western Pacific it has been collected in Australia, New Zealand and Hong Kong. Its area of origin is unknown.
The absence of Chelura from Limnoria-bored wood
in San Francisco Bay, Monterey Bay and Santa Barbara County was
noted by the marine piling surveys of the 1920s (Kofoid, 1921;
Atwood & Johnson, 1924; Hill & Kofoid, 1927), although
Carlton (1979a) argues that due to the patchy distribution of
Chelura populations it could have been present and overlooked.
Chelura was not recorded from the northeast Pacific until
1948 at Hunters Point Naval Shipyard in San Francisco Bay (US
Navy, 1951, p. 185), followed by collections from Los Angeles
Harbor (1950) and Grays Harbor, Washington (1959-1960) (Carlton,
1979a, p. 650).
Corophium acherusicum Costa, 1857
Corophium acherusicum has been reported from bays and harbors in the Atlantic, Pacific and Indian oceans, though which of these may be its native region is unknown. On the Pacific coast it has been collected from numerous bays and harbors ranging from British Columbia (and possibly Alaska) to Baja California. Early records are from Yaquina Bay, Oregon (1905), San Francisco Bay (1912-13 Albatross survey), Puget Sound, Washington (1915), Vancouver Island, British Columbia (1928), and Newport and Anaheim bays in southern California (1935-36) (Carlton, 1979a, p. 653).
Corophium acherusicum is a common fouling organism on floats and pilings, has been reported from oysters, and reported from ship hulls on several occasions (references in Carlton, 1979a). It was probably introduced to the Pacific Coast either as ship fouling or possibly in shipments of Atlantic oysters.
In San Francisco Bay Corophium acherusicum has been collected
upstream to Collinsville, and is among the most common species
in the Department of Water Resources' benthic samples at Carquinez
Strait. In 1993-94 we collected it at stations in San Pablo Bay
and in the Petaluma River. It established high densities in Suisun
and Honker bays during the 1977 drought (Markmann, 1986).
Corophium alienense Chapman, 1988
Corophium alienense was first collected in San Francisco
Bay in 1973 and is probably native to Southeast Asia, based on
its morphological similarity to other Southeast Asian Corophium
(Chapman, 1988). It was most likely introduced to San Francisco
Bay in ballast water (Corophium are known to migrate into
the water column at night, and ballast water often contains amphipods;
Carlton & Geller, 1993), possibly in or on naval ships returning
from Vietnam (Carlton, 1979a, as Corophium sp.; Chapman,
1988). It has become abundant in many parts of the Bay from the
South Bay to the Delta, and is especially abundant on shallow
subtidal and intertidal muddy sand (Chapman, 1988). In 1993-94
we collected it at scattered sites from Tiburon upstream to Rodeo
and the Napa River. It was also found in abundance in Bodega Harbor
in 1992 (J. Chapman, pers. comm.).
Corophium heteroceratum Yu, 1938
Corophium heteroceratum was collected from San Francisco Bay at least by 1989 (Chapman & Cole, 1994) and possibly as early as 1985 or 1986 (Chapman, pers. comm., 1995), and from Los Angeles Harbor in 1990. Outside of California, the only records are the type specimens collected in 1929 from a tide pool in Tangku (Tanggu), China, in the northwestern Yellow Sea. C. heteroceratum is probably native to Asia, as it is morphologically similar to other Asian species of Corophium (Chapman & Cole, 1994).
In San Francisco Bay, Corophium heteroceratum is found on silty sediments at low intertidal or subtidal depths at salinities over 15 ppt, frequently co-occurring with the introduced Atlantic amphipod Ampelisca abdita. It is widespread and locally abundant in the Bay, especially at salinities >20 ppt and temperatures >16° C, reaching densities of up to 9,600/m2, and has been collected at least from the northern South Bay to northern San Pablo Bay (Chapman & Cole, 1994), with a few records from Grizzly Bay (DWR, 1995). We tentatively assign a first date of collection of this amphipod in San Francisco Bay as 1986, based upon the arguments presented by Chapman & Cole (1994) and upon probable circa-1986 specimens received by J. Chapman (J. Chapman, pers. comm., 1995). In 1993-94, we collected C. heteroceratum at Tiburon and at two stations in San Pablo Bay.
As Corophium heteroceratum has been found exclusively on
soft-bottom, not on hard substrates or buoy fouling in San Francisco
Bay, it is unlikely to have been transported in ship fouling (Chapman
& Cole, 1994). Ballast water transport seems likely, as Corophium
are known to migrate into the water column at night (Chapman,
1988), and ballast water often contains demersal plankton (benthic
organisms that migrate into the water column), including amphipods
(Carlton & Geller, 1993).
Corophium insidiosum Crawford, 1937
Corophium insidiosum is a North Atlantic species known from both the European and American coasts (Bousfield, 1973), and introduced to both Chile (by 1947) and Hawaii (by 1970) (Carlton, 1979a, p. 657). The first Pacific record is a specimen taken from the stomach of a bird, a greater scaup, collected at Oyster Bay, Washington in 1915. In 1931 Corophium insidiosum was collected in Lake Merritt in San Francisco Bay, where it was thought to be a new species. It was found in four southern California bays from 1949-1952, in Tomales Bay, Monterey Harbor, Bolinas Lagoon and Elkhorn Slough between 1961 and 1977, in the Strait of Georgia in British Columbia in 1975 (Carlton, 1979a), and on a wooden ship in Humboldt Bay, in 1987 (Carlton & Hodder, 1995). It is commonly found in fouling, and was probably transported to the northwestern Pacific in ship fouling or with shipments of Atlantic oysters.
Corophium insidiosum has remained abundant in Lake Merritt
where we collected it in 1993-94, as well as at several sites
from the mouth of the Bay upstream to Martinez, at Coyote Point
in the South Bay, and at Aquatic Park in Berkeley.
Gammarus daiberi Bousfield, 1969
Gammarus daiberi is native to the northwestern Atlantic in estuaries and sounds from Delaware and Chesapeake bays to South Carolina (Bousfield, 1973). In these locations it attains its highest densities in salinities of 1-5 ppt, but is found seaward to 15 ppt. It was collected in the central Delta in 1983, and since 1986 has been regularly collected in the central and western Delta and Suisun Bay (Hymanson et al., 1994). In 1993-94 we collected it from Bethel Island in the Delta and from Martinez. It is eaten by young striped bass (Hymanson et al., 1994).
On the Atlantic coast it is described as mainly pelagic, though
also commonly collected on the bottom and in fouling (E. L. Bousfield
in litt. to W. C. Fields, Jr., 1991). We consider it to be probably
a ballast water introduction, and less likely a ship fouling introduction.
Grandidierella japonica Stephensen, 1938
This tube-dwelling amphipod is native to Japan. It was collected from San Francisco Bay near Vallejo and in Lake Merritt, Oakland, in 1966, from Tomales Bay in 1969, from Bolinas Lagoon in 1971, from Drakes Estero in 1972-73 (Chapman & Dorman, 1975; Carlton, 1979a, p. 662) and from Coos Bay, Oregon since 1977 (JTC, pers. obs.). It has been established in southern California bays since at least the early 1980s (J. Chapman, pers. comm.). It is typically found on muddy or mud-sand bottom, sometimes in oyster beds, and sometimes in fouling. It was introduced with commercial oyster transplants from Japan, with ship fouling or in ballast water.
Grandidierella japonica has been collected from all parts of San Francisco Bay, from the South Bay near Redwood City upstream to Antioch. It is one of the most common benthic species in San Pablo Bay and Carquinez Strait (Chapman & Dorman, 1975; Nichols & Thompson, 1985a; Markmann, 1986). In 1993-94 we collected it from several stations in San Pablo Bay upstream to Martinez, Napa and Petaluma, from Coyote Point in the South Bay, and from Lake Merritt and Berkeley's Aquatic Park in the East Bay.
In Bolinas Lagoon it has been recorded from the stomachs of least
and western sandpipers, dunlin, black-bellied plover and willet
(Page & Stenzel, 1975; Stenzel et al., 1976).
Jassa marmorata Holmes, 1903
SYNONYM: Jassa falcata of Pacific coast authors in reference
to bay or estuary populations, not of Montagu, 1808 (see Conlan,
1990).
This Atlantic fouling amphipod is now widely spread on both sides of the North Atlantic, in the Mediterranean and on the Pacific coast of North America, and reported from other locations as well. Carlton (1979a) predicted that the bay and harbor populations of so-called "Jassa falcata" represented "an introduced taxon." Conlan (in litt., 7 Oct. 1986 to JTC and in litt., 5 Aug. 1986 to J.W. Chapman) noted that based on her systematic revision of the genus Jassa and her field work on the Pacific coast, she "found the distribution of [Jassa] to be as predicted by" Carlton (1979a): endemic species occurred on the exposed outer coast, and the Atlantic Jassa marmorata to be harbor-restricted. Conlan (in litt.; also see Conlan, 1988) states that Jassa marmorata is "the most recently derived of all species of Jassa," that it originated in the North Atlantic and specifically on the "Atlantic North American coast," and that it is introduced to Europe, the Mediterranean, the Pacific Ocean (China, Japan, USSR, Chile, and Pacific North America), the South Atlantic (Brazil, West Africa, and South Africa), the Indian Ocean (Zanzibar) and Australia and New Zealand. It ranges in the Western Atlantic from Newfoundland to Texas and Cuba. On the Pacific coast J. marmorata has been collected from Alaska (one locality, Point Slocum) and British Columbia (Victoria Harbor, Bamfield) and then from Coos Bay, Oregon to Bahia de Los Angeles, Baja California (Conlan, 1990). Additional harbor records cited by Carlton (1979a, pp. 667-668) may also include Jassa marmorata.
The earliest San Francisco Bay record appears to be material collected in the Oakland Estuary in 1977 (Carlton, 1979a). That Jassa marmorata is a 20th century rather than a 19th century introduction is suggested by the relatively late reports of estuarine members of the Jassa falcata group from the eastern Pacific (in 1941 from Estero de San Antonio, 75 km north of San Francisco, and in 1942 from Magdalena Bay, Baja California; Carlton, 1979a). Both Carlton (1979a) and Conlan (1988) have declined to accept Barnard's (1969) proposal that "Podocerus californicus," described by Boeck (1872) from California, is "Jassa falcata."
Jassa marmorata occurs in fouling communities and on ship
hulls (Bousfield, 1973) and with oysters (Wells, 1961, as "Jassa
falcata"). It has also been collected from the ballast
tanks of a cargo ship arriving in Coos Bay, Oregon after a 15
day trip from Japan, in water that had been taken aboard in Kobe
on the Inland Sea of Japan (specimens identified by K. Conlan,
in litt., 4 Aug. 1988). Lack of early reports of this now locally
common species suggests ship fouling or ballast water as the primary
mechanism of transport.
Leucothoe sp.
We regard the endocommensal amphipod found inside the introduced tunicates Ciona and Ascidia in San Francisco Bay as an introduced species. It may belong to the species complex bearing the names Leucothoe spinicarpa (Abildgaard, 1789) and Leucothoe alata Barnard, 1959 (J. Chapman, pers. comm., 1995). Nagata's (1965) illustrations of "Leucothoe alata" from Japan, which may not be the same as Barnard's original material of this species, appear close to if not identical to San Francisco Bay specimens (J. Chapman, pers. comm., 1995).
In 1993-94 we collected this amphipod in Ciona and Ascidia
at Coyote Point in the South Bay and Coast Guard Island in
the Oakland Estuary. It was likely introduced inside a tunicate
transported either in ship fouling or possibly with oyster shipments.
While the first actual collection record that we have found is
material collected in 1977 from the Oakland Estuary, this leucothoid
may have been present in the northeastern Pacific since the introduction
of Ciona (which was collected in San Diego Bay in 1897
and in San Francisco Bay in 1932).
Melita nitida Smith, 1873
Melita nitida is native to the northwestern Atlantic, ranging from the Gulf of St. Lawrence to the Yucatan Peninsula. It was first collected from San Francisco Bay in 1938, from Howe Sound in British Columbia in 1973, from Elkhorn Slough in 1975, and in Oregon from Yaquina, Coos and Alsea bays in 1986-87 (Carlton, 1979a, p. 672; Chapman, 1988).
On the Pacific coast Melita nitida is commonly found in
fouling, under intertidal rocks and debris, and in Enteromorpha
or diatom mats on mudflats, in salinities from 0 to 25 ppt (Chapman,
1988). On the Atlantic coast it has been reported from similar
habitats as well as from oyster beds. Melita nitida could
have been transported to the Pacific coast in ship fouling, in
transcontinental shipments of Atlantic oysters, or possibly in
solid ballast or ballast water. It could have been transported
between bays in fouling or ballast, or with shipments of oysters
or the introduced soft-shell clam Mya arenaria. In San
Francisco Bay it has been collected from Lake Merritt, Point Richmond,
Rodeo, Petaluma, Martinez and Grizzly Bay, and from Collinsville
on the Sacramento River at densities of up to 355/m2
(Chapman, 1988; DWR, 1995; and 1993-94 survey).
Melita sp.
In 1993 we collected an amphipod in the genus Melita, distinct
from Melita nitida, that had not been previously reported
from San Francisco Bay (J. Chapman, pers. comm., 1995). While
its origin is unknown, introduction via ship fouling or ballast
water are the most probable mechanisms.
Paradexamine sp.
In 1993-94 we collected an amphipod in the genus Paradexamine
that had not been previously reported from San Francisco Bay (J.
Chapman, pers. comm., 1995). Introduction was probably by ship
fouling or ballast water.
Parapleustes derzhavini (Gurjanova, 1938)
SYNONYMS: Neopleustes derzhavini
Parapleustes derzhavini makiki Barnard, 1970
Parapleustes derzhavini is known as a rare species from among intertidal and subtidal algae in the western Pacific in Japan and Russia. It has also been collected from Hawaii, where it is probably an introduction. In the northeastern Pacific it was collected from San Francisco Bay in 1904 (discovered among USNM campanularid hydroid specimens by J. W. Chapman), Tomales Bay in 1970, Coos Bay in 1986 and Yaquina Bay in 1987 (Carlton, 1979a; Chapman, 1988). In San Francisco Bay it has been collected from San Mateo Point in the South Bay to Grizzly Bay, and upstream as far as Collinsville on the Sacramento River in the 1977 drought (Chapman, 1988; DWR, 1995). It was probably introduced in ship fouling.
On the Pacific coast P. derzhavini has been found at salinities
of 6 to 32 ppt., abundant on hydroids in fouling but rare on algae.
Specimens from brackish water on the Pacific coast identified
as Parapleustes pugettensis may in fact be P. derzhavini.
Stenothoe valida Dana, 1852
Stenothoe valida has a widespread, mainly tropical distribution.
It has been reported from only four Pacific coast embayments:
San Francisco Bay (first collected in 1941), Los Angeles Harbor
(1950-51), Newport Bay (1951) and Bahia de San Quintin, Baja California
(1960-61) (Carlton, 1979a, p. 677). It is commonly found among
fouling, especially in hydroids, and was probably introduced either
in ship fouling or in ballast water. In 1993-94 we collected Stenothoe
valida, identified by J. W. Chapman, at sites all around the
Central Bay.
Transorchestia enigmatica (Bousfield & Carlton, 1967)
SHOREHOPPER
SYNONYMS: Orchestia enigmatica
This beach-dwelling amphipod was first collected in Lake Merritt,
Oakland (a brackish lagoon) by JTC in 1962, and is known only
from the Lake and (rarely) from the channel connecting to the
Oakland Estuary. A closely related (or possibly identical) species,
Transorchestia chilensis, is reported from Chile and New
Zealand. Like other talitrid amphipods, T. enigmatica cannot
survive long immersion in water, and its likeliest means of introduction
is in solid ballast (i. e. sand, stones and detritus from beaches)
that was in common use by wooden cargo ships up until the 1920s.
There was substantial trade between California ports and Peru
and Chile from the last half of the 19th century to the 1920s,
with ships going south carrying grain or lumber and returning
in ballast (Carlton, 1979a).
Carcinus maenas (Linnaeus, 1758)
GREEN CRAB
This common European shorecrab was introduced to the Atlantic coast of North America by 1817 (Say, 1817), to southern Australia by 1900 (Fulton & Grant, 1900) and to South Africa by 1983 (Le Roux et al., 1990). It was first collected in California in the Estero Americano, Solano County, in 1989, and in San Francisco Bay by a bait trapper in Redwood Shores Lagoon, San Mateo County in the summer of 1989 or 1990. It was probably transported to San Francisco Bay in ballast water, although other possible mechanisms include shipment in algae used to pack shipments of live New England bait worms (Nereis virens and Glycera dibranchiata) or lobsters (Homarus americanus), release as discarded research material, or transport in a ship's seawater pipe system (Cohen et al., 1995; Carlton & Cohen, 1995).
In San Francisco Bay it has been collected from the South Bay from south of the Dumbarton Bridge to Benicia in the Carquinez Strait, where it is found intertidally and subtidally to 10 meters deep, and in lagoons around the Bay. It is commonly caught in traps set for bait fish (gobies and cottids), sometimes with hundreds of crabs filling each trap, and in shrimp nets. In 1993 it was collected from Drakes Estero, Tomales Bay and Bodega Harbor (Grosholz & Ruiz, 1995), in 1994 from Elkhorn Slough (T. Grosholz, pers, comm., 1994), and in 1995 from Humboldt Bay (T. Miller, pers. comm., 1995).
Carcinus tolerates salinities from 4-52 ppt and temperatures down to around 0°C, and can reproduce at temperatures up to around 18-26°C. In favorable conditions, females can spawn up to 185,000 eggs at a time. In various parts of the world it has become common in virtually all types of protected and semiprotected marine and estuarine habitats, including habitats with mud, sand or rock substrates, eelgrass beds and cordgrass marshes. Its wide environmental tolerances suggest that on the Pacific coast it could eventually range from Baja California to Alaska (Cohen et al., 1995; Carlton & Cohen, 1995).
In field observations or laboratory experiments, Carcinus has been seen to eat an enormous variety of prey items, including organisms from at least 104 families and 158 genera in 5 plant and protist and 14 animal phyla. In analyses of stomach contents, dominant prey at different locations have included mussels, clams, snails, polychaetes, crabs, isopods, barnacles and algae (Cohen et al., 1995). In California, Carcinus was observed to significantly reduce the density of the small clams Nutricula (Transennella ) spp., the cumacean Cumella vulgaris, and the amphipod Corophium sp. (Grosholz & Ruiz, 1995), and in the lab also consumed the mussel Mytilus sp., the Asian clams Potamocorbula amurensis and Venerupis philippinarum, and the native crabs Hemigrapsus oregonensis and Cancer magister (Dungeness crab) at up to its own size (Cohen et al., 1995; Grosholz & Ruiz, 1995).
Carcinus is fished commercially for food and bait in Europe,
though its relatively small size has prevented its entering the
commercial market in the United States. Through its predatory
activities, it is generally credited with the destruction of soft-shell
clam fisheries in New England and Canada in the 1950s, where control
efforts have included fencing, trapping and poisoning, with varying
success (Cohen et al., 1995).
Eriocheir sinensis H. Milne-Edwards, 1854
CHINESE MITTEN CRAB
Chinese mitten crabs are native to Korea and China from the Yellow Sea to south of Shanghai. They spend most of their lives in the rivers and migrate to the estuaries to reproduce. Most authorities have recognized four species of mitten crabs, including Eriocheir sinensis and E. japonicus which are distinguished by clear and consistent morphological differences (Sakai, 1939; Dai & Yang, 1991). Recently Li et al. (1993) found small genetic distances between these two forms suggestive of a single species, but confirmed the existence of morphological distinctions (which they described as ecophenotypic, although the differences appear to be more simply explained as the expression of genetically different populations and their hybrids). Dai (1993) and Chan et al. (1995) have proposed other modifications to the arrangement of species within the genus. In light of this unstable taxonomy, we continue to treat the Chinese mitten crab, E. sinensis, as a distinct species.
A Chinese mitten crab was collected in the Aller River, Germany in 1912, generally presumed to have been introduced in ballast water (Panning, 1939). Mitten crabs spread through the Netherlands and Belgium to northern France by 1930 (Hoestland, 1948), eventually reaching the west coast of France and, via the Garonne River and the Canal du Midi, the Mediterranean coast by 1959 (Hoestland, 1959; Zibrowius, 1991). They became phenomenally abundant in Germany in the mid-1930s, with masses of crabs migrating up the main rivers, piling up against dams, climbing spillways and swarming over the banks onto shore, sometimes wandering onto city streets and entering houses. Government authorities operated barrel and pit traps that caught tens of millions of crabs each year in order to prevent damage to banks and levees (the crabs dig burrows over half a meter deep in mud banks) and reduce interference with trap and net fisheries (Panning, 1939). A "plague of mitten crabs" was similarly reported from the Netherlands in 1981 (Ingle, 1986).
Hundreds of adult mitten crabs have been collected along the shores of the Baltic Sea, but as the Baltic's salt content is too low for successful spawning these are generally thought to be individuals transported by ship from the North Sea (Haahtela, 1963; Rasmussen, 1987). Occasional mitten crabs, including a few ovigerous females, have been collected in England since 1976, though it is unclear whether breeding populations are established there (Ingle, 1976).
A Chinese mitten crab was collected in the North American Great Lakes in 1965 and nine or ten additional adult crabs were collected between 1973 and 1994, all but one of which were taken from western Lake Erie (Nepszy & Leach, 1973; J. Leach, pers. comm.). As in the Baltic, the Great Lakes are too fresh for mitten crabs to spawn, and each individual is thought to have arrived as a larva or juvenile in ballast water from Europe. A single adult mitten crab was collected from the Mississippi River delta in Louisiana in 1987, with none reported since (Howarth, 1989; D. Felder, pers. comm.).
In November, 1994 a crab caught in a shrimp net in the southern end of San Francisco Bay was identified as Eriocheir sinensis by Robert Van Syoc of the California Academy of Sciences. Shrimp trawlers report that they have occasionally caught such crabs, many of them carrying eggs, in the South Bay since 1992 and in San Pablo Bay since the summer of 1994. Of 75 crabs collected from San Francisco Bay, 24 were female, and all but 5 of these were carrying eggs. Several ovigerous females collected in the winter of 1994-95 were maintained in aquaria by the Marine Science Institute of Redwood City, California, and hatched active zoeae by the first week of February. In 1995 Katie Halat found juvenile mitten crabs to be common in burrows in the upper parts of sloughs at the southern end of the South Bay.
Mitten crabs could either have arrived in San Francisco Bay in ballast water from Asia or Europe, or been intentionally planted in the watershed as a food resource. In 1978 Dustin Chivers of the California Academy of Sciences noted that live mitten crabs could be imported into California from firms in Hong Kong and Macao. In 1986 the California Department of Fish and Game found live mitten crabs, bound with twine, offered for sale in Asian food markets in San Francisco and Los Angeles at prices of $27.50 to $32.00 per kilogram. Although the importing of live mitten crabs was banned by the California government in 1987 and the United States government in 1989, the high price they command has encouraged continuing efforts to import them through official or unofficial channels. On 11 occasions since 1989, U. S. Fish and Wildlife inspectors intercepted batches of 10-28 mitten crabs hand-carried by travelers from Asia disembarking at the San Francisco Airport during the winter (H. Roche, pers. comm.), and crabs have been intercepted at Los Angeles and Seattle as well (M. Osborne and M. Williams, pers. comm.). In 1994 an Asian businessman lobbied the California legislature for permission to import and raise mitten crabs in California (T. Gosliner, pers. comm., 1994).
With its establishment in San Francisco Bay, the mitten crab is one of the few catadomous organisms (living in fresh water and breeding in salt) in North America. Studies on these crabs in Asia and Europe indicate that they live in burrows dug in river banks or (in Asia) in rice paddies in coastal areas. Some migrate far upstream, and are recorded from the Changjiang (Yangtze) River over 1,250 km from the sea. In the late fall and winter adult crabs (1-2 years old in China (G. Li, pers. comm., 1995); 3-5 years old in Germany (Panning, 1939)) migrate to coastal waters where they mate, spawn and die. Each female produces from 250,000 to 1 million eggs, which hatch in late spring or early summer. The larvae develop through five increasingly stenohaline and euhaline zoeae and a more euryhaline and mesohaline megalopa. After the final larval molt the juvenile crab settles to the bottom and begins its migration upstream (Panning, 1939; Ingle, 1986; Anger, 1991).
The ban on importing live mitten crabs was enacted due to concern
over potential damage from its burrows to levees or rice fields
in the Central Valley, and because the crab is a second intermediate
host of a human parasite, the oriental lung fluke Paragonimus
westermanii. Armand Kuris and Mark Torchin of U. C. Santa
Barbara found no parasites of any kind in 25 mitten crabs from
San Francisco Bay (A. Kuris, pers. comm., 1995). However, since
suitable first intermediate snail hosts are present in California
or adjacent states (T. Gosliner, pers. comm.), establishment of
the fluke is possible, which could lead to infections of humans,
or more likely, other mammals. The potential ecosystem impacts
of large numbers of river crabs, where none now exist, are unknown.
Orconectes virilis (Hagen, 1871)
VIRILE CRAYFISH
SYNONYMS: Cambarus virilis
This crayfish is native to Indiana, Illinois and other midwestern states. It was introduced into California waters at Chico in Butte County between 1939 and 1941, from crayfish that were being held in ponds for use as laboratory specimens at Chico State College. It has since been reported at the edges of the Delta in the lower Cosumnes River, in Putah Creek and in drainage and irrigation ditches in Yolo County, and further north in Butte and Colusa counties where it digs burrows in rice fields and eats rice shoots and is considered a pest by farmers (Riegel, 1959; Herbold et al., 1992).
The U. S. Fish and Wildlife Service proposed listing the native
Shasta crayfish Pacifasctacus fortis as an endangered species
because it had been extirpated from half its range between 1978
and 1987, in large part due to competition from Orconectes
virilis and another introduced crayfish, P. leniusculus,
for food and space (Anon., 1987).
Pacifastacus leniusculus (Dana, 1852)
SIGNAL CRAYFISH
SYNONYMS: Astacus leniusculus
It is unclear when the signal crayfish Pacifastacus leniusculus, native to Oregon, Washington and British Columbia, was first introduced to California. Osborne (1977) stated that it was introduced to Lake Tahoe in the 19th century as forage for game fish. Kimsey et al. (1982; repeated by Herbold & Moyle, 1989, and Herbold et al., 1992) reported that it was found in San Francisco County in 1898. Riegel (1959), however, speaking about the introduction of this species to California, reported that in 1912 signal crayfish from the Columbia River "were shipped in large batches to the Brookdale Hatchery of the California Fish and Game Commission in Santa Cruz County [in order] to determine their depredatory effects upon young trout. Later, many were released into the San Lorenzo River near Santa Cruz, and about 200 were shipped to Nevada County, California, and released in a private pond on the Shebley Ranch between Colfax and Grass Valley. They were thriving 18 years later." Bonnot (1930) reported it as imported "in times past for culinary purposes and as biological material."
Signal crayfish are now widely distributed throughout the Delta and Bay Area and central California, north to Siskiyou County and south to Monterey County (Riegel, 1959; Hazel & Kelley, 1966). They are the main crayfish taken from the Delta, where a commercial harvest began in 1970 with a catch of 50 tons and produced annual landings of 250 tons by the 1980s (Osborne, 1977; Herbold & Moyle, 1989). Commonly found in streams, large rivers, lakes and sometimes muddy sloughs, Riegel (1959) reported it collected on one occasion from dilute brackish water, and Kimsey et al. (1982) reported that it tolerates salinities up to 17 ppt.
Pacifastacus leniusculus may have contributed to the extinction of the native sooty crayfish, Pacifastacus nigrescens, which in the 19th century had been abundant in creeks around San Francisco Bay (Riegel, 1959; Kimsey et al., 1982). In 1987 the U. S. Fish and Wildlife Service proposed listing the native Shasta crayfish Pacifasctacus fortis as an endangered species because it had been extirpated from half its range between 1978 and 1987, in large part due to competition from P. leniusculus and another introduced crayfish, Orconectes virilis, for food and space (Anon., 1987).
Pacifastacus leniusculus has also been introduced to northern
Europe, with populations established in Sweden (introduced from
Lake Tahoe in 1969; Osborne, 1977), Finland, Lithuania and Poland
(McGriff, 1983). In Sweden the introduction of P. leniusculus
and a North American crayfish fungus have been described as the
main cause of the decimation of the noble crayfish Astacus
astacus (Jansson, 1994).
Palaemon macrodactylus Rathbun, 1902
ORIENTAL SHRIMP, KOREAN SHRIMP, GRASS SHRIMP
This shrimp is native to Korea, Japan and northern China and was first collected in San Francisco Bay in 1957, in Los Angeles Harbor in 1962, in Santa Monica Bay in the 1970s, in Coos Bay in 1987, and in Humboldt Bay in 1995 (Newman, 1963; Carlton, 1979a, p. 687; T. Miller, pers. comm., 1995). It is distributed widely throughout San Francisco Bay and upstream into the Delta, especially in dry years, and has been collected in the Delta-Mendota Canal. It is frequently abundant in brackish lagoons such as Lake Merritt in Oakland and Aquatic Park in Berkeley (Carlton, 1979a). In 1993-94 we collected it from among the fouling on docks at several sites in the Bay and upstream in the Napa River to John F. Kennedy Park and in the Petaluma River to the City of Petaluma.
Palaemon's appearance in the Bay around the mid-1950s may be related to increased shipping with South Korean and Japanese ports related to the Korean War. It was likely transported in ballast water or possibly, as Newman (1963) argued, within the fouled seawater system of a ship.
Palaemon is a hardy and eurytopic organism tolerating a
wide range of salinities down to 1-2 ppt and water of low quality.
As discussed by Newman (1963) and Carlton (1979a), although Palaemon's
geographic distribution within the estuary overlaps with that
of native crangonid shrimp, it is unlikely to substantially compete
with them due to differences in habitat use. In the Delta Palaemon
mainly eats opossum shrimp Neomysis mercedis (Herbold et
al., 1992). Palaemon has been found in the stomachs of
white sturgeon, white catfish and striped bass (Gannsle, 1966;
Thomas, 1967; McKechnie & Fenner, 1971), and is used as sturgeon
bait (Herbold et al., 1992).
Procambarus clarkii (Girard, 1852)
RED SWAMP CRAYFISH
SYNONYMS: Cambarus clarkii Girard, 1852
The red swamp crayfish is native to Louisiana, Texas and other southern states, where it is the main cultivated crayfish due to its rapid growth, reaching a marketable size of 7.5 cm in three months (Herbold et al., 1992). Holmes (1924) reported that it was collected from a stream near Pasadena in the summer of 1924 (Skinner (1962) and BDOC (1994) stating that it was introduced from the Midwest in 1925). Riegel (1959) reported that the crayfish was imported in 1932 by a frog farmer in Lakeside, San Diego County for use as frog food, but that it may have already been present in California before then. Its initial appearance in California probably resulted from an intentional importation for commercial use or as a food resource, followed by an intentional or accidental release.
The red swamp crayfish is now widely distributed throughout the central part of the state and is the only crayfish found south of the Tehachapis (Riegel, 1959). It has been taken regularly in the Delta (Hazel & Kelley, 1966), and in 1995 we found it at Shell Marsh east of Martinez. BDOC (1994) reports that it is fished commercially and recreationally in the Estuary for food and for scientific use, although Kimsey et al. (1982). reported only incidental take of this species for bait and sport.
The red swamp crayfish prefers warmer water than does the signal
crayfish, survives in stagnant water by using atmospheric oxygen,
and tolerates salinities up to 30 ppt. It is frequently found
in rice fields and sloughs with abundant emergent vegetation.
It is regarded as a pest in rice fields and irrigation ditches
because it eats young rice shoots and digs burrows two inches
in diameter and as much as 40 inches deep into levees and banks
(Riegel, 1959; Kimsey et al., 1982; Herbold et al., 1992), and
Skinner (1962, p. 124) described it as "mechanically destructive
to dikes and levees." At Coyote Hills Marsh in Alameda, a
freshwater/brackish wetlands on the eastern shore of south San
Francisco Bay, red swamp crayfish have been shown to reduce the
abundance of sago pondweed, Potamogeton pectinatus and
are preyed upon by raccoon, Procyon lotor. The reduction
or elimination of submersed macrophytes by grazing crayfish may
reduce marsh diversity and secondary production by eliminating
habitat for epiphytic organisms, and on the other hand may benefit
vector control efforts by reducing larval mosquito habitat (Feminella
& Resh, 1989).
Rhithropanopeus harrisii (Gould, 1841)
HARRIS MUD CRAB
Rhithropanopeus is native to the northwest Atlantic from New Brunswick to Florida and from Mississippi to Vera Cruz, Mexico, in upper estuarine areas in fresh and brackish water. It was introduced to Europe, presumably among ship fouling, by 1874, and was collected in the Panama Canal in 1969. The first records of Rhithropanopeus from the Pacific are specimens collected from Lake Merritt, Oakland in 1937. It was subsequently collected from Oregon in Coos Bay in 1950, in Netarts Bay in 1976, and in Yaquina Bay and the Umpqua River in 1978 (Carlton, 1979a, p. 697).
In the Atlantic Rhithropanopeus is commonly found in oyster beds (Ryan, 1956; Wells, 1961; Maurer & Watling, 1973), and it may have been introduced to San Francisco Bay with shipments of the Atlantic oyster Crassostrea virginica, which was still being imported from the Atlantic in small quantities in the 1930s. It could also have been introduced via ship fouling or ballast water.
Though Rhithropanopeus has apparently been absent from Lake Merritt since at least the 1960s, we have found it common in similar habitat among masses of the tubes of the Australian serpulid worm Ficopomatus enigmatica in the Petaluma River at Petaluma, and on the shore under rocks at low tide in Carquinez Strait (associated with the native shorecrab Hemigrapsus oregonensis). It is reported as present to abundant from San Pablo Bay to the Delta, is regularly collected at the Central Valley Project pumps at Tracy in the south Delta (S. Siegfried, pers. comm., 1994), and has been found in the Delta-Mendota Canal (Carlton, 1979a). It has recently been collected in the upper parts of sloughs in the far South Bay, sympatric with juveniles of the recently introduced catadromous mitten crab Eriocheir sinensis (K. Halat, pers. comm., 1995). Rhithropanopeus' planktonic larvae are caught in Suisun Bay and to a much lesser extent in San Pablo Bay, and the abundance of these larvae is inversely correlated with high outflows during the summer (Herbold et al., 1992).
Jones (1940) suggested that Hemigrapsus would be likely
to outcompete Rhithropanopeus where their distributions
overlap in San Francisco Bay, and Jordan (1989) found that the
distribution of Rhithropanopeus is restricted by Hemigrapsus
in Coos Bay, Oregon. In the Delta, Rhithropanopeus is eaten by
white sturgeon, white catfish and striped bass (Stevens, 1966;
Turner, 1966a; Thomas, 1967; McKechnie & Fenner, 1971).
Anisolabis maritima (Gene, 1832)
MARITIME EARWIG
This predaceous maritime earwig is native to the North Atlantic
region and has been reported from Japan, Formosa and New Zealand.
It was first collected in the San Francisco Estuary in 1935, where
it has been found from San Pablo Bay to Carquinez Strait but not
along the ocean coast in this area (Langston, 1974). It was also
reported from Nanaimo in British Columbia (in 1920), and from
Laguna Beach (1921) and Costa Mesa (1944) in southern California,
but there are no subsequent records from these areas (Carlton,
1979a, p. 702). Reports of this insectóotherwise known
only from the seashore, typically near the high-tide levelófrom
shipments of dahlias and crysanthemums arriving in southern California
probably refer to another species. It may have been transported
to the Pacific coast in solid ballast in the late 19th or early
20th century, and remained unrecognized for some years.
Neochetina bruchi Hustache and Neochetina eichhorniae
Warner
In an effort to control water hyacinth, Eichhornia crassipes, the U. S. Department of Agriculture introduced into Florida two weevils from Argentina, Neochetina eichhorniae (in 1972) and N. bruchi (in 1974). Both weevils were subsequently established in Louisiana and Texas, and have been introduced to many other parts of the world (N. eichhorniae to Zambia (1971), Zimbabwe (1971), South Africa (1974), Australia (1975), Fiji (1977), Sudan (1978), Indonesia (1979), Thailand (1979), Egypt (1980), Myanmar (1980), Solomon Islands (1982), India (1983), Malaysia (1983), Vietnam (1985), Papua New Guinea (1985), Sri Lanka (1988) and Honduras (1990); and N. bruchi to Panama (1977), Sudan (1979), India (1984), South Africa (1989), Australia (1990) and Honduras (1990)) (Julien, 1992).
The California Department of Boating and Waterways and the USDA,
responding to a build-up of water hyacinth, released N. bruchi
into the Sacramento-San Joaquin Delta beginning in July 1982,
and N. eichhorniae in 1982 or 1983. Although both weevils
have become established in the Delta, there is no evidence that
they have reduced water hyacinth there (Thomas & Anderson,
1983; L. Thomas, pers. comm., 1994).
Trigonotylus uhleri Reuter
The mirid bug Trigonotylus uhleri is native to the Atlantic coast of North America, where it is an herbivore specialist on cordgrass (Spartina spp.) commonly found on the smooth cordgrass S. alterniflora. It was first collected on the Pacific Coast by Curtis Daehler and Donald Strong in San Francisco Bay in 1993 (Daehler & Strong, 1995).
In San Francisco Bay, where S. alterniflora was introduced from the Atlantic in the early 1970s, Trigonotylus achieves higher densities on S. alterniflora than is typically observed on the Atlantic Coast, exceeding 10 individuals per culm (about 3,000/m2). These high densities, however, appear to have little impact on the plant's vegetative growth, lateral spread, inflorescence or seed production. Trigonotylus is also found on the native Pacific cordgrass S. foliosa (Daehler & Strong, 1995).
Trigonotylus seems likeliest to have been transported to
the Pacific coast with cordgrass plants imported for erosion control
or marsh restoration, possibly with the Spartina alterniflora
introduced to San Francisco Bay, if that stock was imported as
plants rather than seed.
Barentsia benedeni (Foettinger, 1887)
SYNONYMS: Barentsia gracilis of Mariscal, 1965
See Carlton, 1979a for other synonyms.
The distribution of this European entoproct in the northeastern Pacific is poorly known, as it has long been confused with the native Barentsia gracilis. B. benedeni has been recorded from San Francisco Bay since 1929 (as Ascopodaria gracilis, "Barentsia (=Pedicellina)", and Barentsia gracilis), at Lake Merritt, Palo Alto Yacht Harbor and Berkeley Yacht Harbor (Mariscal, 1965; Carlton, 1979a, p. 704). It was also collected in Australia in the 1940s (Wasson & Shepherd, 1995), from the Salton Sea in southern California in 1977 (Jebram & Everitt, 1982), from Coos Bay, Oregon since 1988 (Hewitt, 1993), and in the western Atlantic from Massachusetts in 1977-78 (Jebram & Everitt, 1982).
Barentsia benedeni was probably introduced to San Francisco
Bay in ship fouling, or possibly as fouling on oysters shipped
from Japan, where it has been reported in Matsushima Bay (Toriumi,
1944). Barentsia does not have planktonic larvae and have
not been reported from ballast water (e. g. Carlton & Geller,
1993), although transport of adults on floating debris in ballast
tanks might be possible.
Urnatella gracilis Leidy, 1851
Urnatella gracilis, the world's only freshwater entoproct, is native to North America from the northeastern and midwestern United States west to Texas and Oklahoma. It was first found in Europe in 1939 in Belgium, and later reported from a few sites eastward to western Russia, perhaps derived from a second introduction via the Black Sea (Lukacsovics & Pécsi, 1967). It has also been reported from India (redescribed as Urnatella indica), Uruguay, central Africa, and Japan (Eng, 1977; Emschermann, 1987) and in a Florida canal in 1977 (Hull et al., 1980).
Urnatella was first found west of the Rocky Mountains in 1972-74 in the Delta-Mendota irrigation canal in the San Joaquin Valley (Eng, 1977). The canal runs south from the Delta, and Urnatella colonies were observed locally encrusting the concrete side-lining at 64 km and southward from the Delta. In earth-lined reaches Urnatella was found encrusting the shells of the Asian clam Corbicula fluminea, pebbles and debris, and rarely attached to the Black Sea hydroid Cordylophora caspia. Unattached single entoproct stalks, an asexual dispersal stage, were occasionally found in bottom sediments throughout the concrete-lined reaches. Markmann (1986) indicated that Urnatella was collected in the Delta between 1982 and 1984.
Emschermann (1987) reported that Urnatella produces heavily
cuticularized segments that under disadvantageous conditions,
such as in a low oxygen or low temperature environment, act as
resting buds or hibernacula. The entoproct rarely reproduces sexually,
but relies on asexual production of special propagation branches
which, breaking off, serve as a free-living, creeping and floating
migratory life stage. Since Urnatella frequently colonizes
the shells of freshwater snails and bivalves (Lukacsovics &
Pécsi, 1967; Eng, 1977; Hull et al., 1980) and the surface
of some plants, such as cattails and reeds (Lukacsovics &
Pécsi, 1967; Hull et al., 1980), it was likely transported
to California with aquarium materials or ornamental plants.
Alcyonidium polyoum (Hassall, 1841)
SYNONYMS: Alcyonidium mytili O'Donoghue, 1923
In California Alcyonidium polyoum has been reported from Tomales Bay (Osburn, 1953), from San Francisco Bay on shells of the introduced Atlantic mudsnail Ilyanassa obsoleta (in 1951-52, Filice, 1959), and in Berkeley Yacht Harbor (Banta, 1963). We also observed it at Crown Beach in Alameda (in 1995) and on shells of the introduced Atlantic oyster drill Urosalpinx cinerea in Foster City Lagoon (in 1992).
In the Atlantic A. polyoum has been reported from northern
Labrador and Nova Scotia to Chesapeake Bay, and from Brazil (Osburn,
1944). It has been collected on Ilyanassa shells in Delaware
Bay oyster beds (Maurer & Watling, 1973) and in North Carolina
oyster beds (Wells, 1961). Specimens also referred to A. polyoum
have been recorded from cold boreal waters. In the Pacific Ocean
these records are mainly from Puget Sound northward, including
such locations as the offshore waters near Point Barrow, Alaska.
It seems likely that two species are involved, and we consider
the shallow, estuarine records in San Francisco and Tomales bays
to represent an Atlantic bryozoan. Alcyonidium species
have planktotrophic larvae, which have been found in ballast water
after a 14-day transoceanic voyage (JTC unpublished). Alcyonidium
species, including A. polyoum (as A. mytili), have
also been reported from fouling on ships (WHOI, 1952). Thus this
bryozoan could be either a ballast water introduction, or a late
introduction with oyster shipments or ship fouling.
Anguinella palmata van Beneden, 1845
AMBIGUOUS BRYOZOAN
In 1993-95 we found an arborescent, silt-covered ctenostome bryozoan in San Francisco Bay which was tentatively identified as Anguinella palmata by William Banta. We collected it from underneath floating docks at several locations (Point San Pablo Yacht Harbor and Loch Lomond Yacht Harbor in San Pablo Bay; San Leandro Marina, Mission Rock, Coyote Point and Pete's Harbor in the South Bay), and intertidally on rocks on the east side of Bay Farm Island in the South Bay. A. palmata is an Atlantic species known from England, Netherlands, Belgium, France, from Massachusetts to Florida, Puerto Rico and Brazil, and has been found in salinities ranging from 13 to 32 ppt (Osburn, 1944; Prenant & Bobin, 1956). In 1953 Osburn reported the first collections of A. palmata from the Pacific, made by the Velero III in 1933-42, from Zorritos Light, Peru; Panama City, Panama; Isabel Island, Mexico; and Newport Harbor and Seal Beach, California. It has also been reported from New Zealand (Gordon, 1967).
Anguinella palmata has been reported from ship hulls (WHOI,
1952), and was probably transported from the Atlantic in ship
fouling. As it has lecithotrophic larvae, which spend but a brief
time in the plankton, it is unlikely to have been introduced by
ballast water.
Bowerbankia gracilis Leidy, 1855
CREEPING BRYOZOAN
SYNONYMS: (?) Bowerbankia gracilis of authors (in reference to certain Pacific coast estuarine populations); not (?) of Leidy, 1855 (author of gracilis, not O'Donoghue, 1926 as given in Soule et al., 1975)
(?) Bowerbankia imbricata of authors (in reference to certain
Pacific coast estuarine populations); not (?) of Adams, 1800
We tentatively treat here the cosmopolitan fouling bryozoan Bowerbankia gracilis as introduced. Occurring in the western Atlantic from Greenland to South America (Osburn & Soule, 1953) in salinities down to 10 ppt (Osburn, 1944), to which region it may be native, it has been reported from many other parts of the world including Hawaii, India, England and Saudi Arabia (Soule & Soule, 1977, 1985). A number of subspecies and varieties have been described and these may either represent a single variable species or some number of distinct species. For example, under the varietal names typica, caudata and aggregata, O'Donoghue & O'Donoghue (1923, 1926) reported B. gracilis from a number of British Columbia stations from the intertidal zone to 50 meters. Soule et al. (1980) report B. gracilis as occurring from Puget Sound to Baja California. Records north of central California, however, appear to be restricted to Puget Sound (a single collection of unreported date (Osburn & Soule, 1953) and Coos Bay (since 1970; JTC unpublished; Hewitt, 1993)). Osburn & Soule (1953) report it from collections (likely made in the 1940s) in Tomales Bay and Los Angeles Harbor; it remains abundant in Los Angeles and Monterey Harbors (Soule et al. 1980; Haderlie, 1969). Jebram & Everitt (1982) report a ctenostome as "Bowerbankia cf. gracilis" from the Salton Sea.
Although Light (1941) while reporting on encrusting estuarine
communities in central California did not mention Bowerbankia,
Smith et al. (1954) found it "extremely abundant on pilings"
in the same region (which, based on knowledge of Smith's usual
sampling sites, probably refers to San Francisco Bay), and Banta
(1963) recorded it specifically from San Francisco Bay. Light
and his students may have overlooked this organism, but perhaps
a more likely scenario is its introduction into Tomales Bay with
oyster shipments after the collecting reported by Light in 1941
(or into some other less well examined bay with oysters or in
ship fouling anytime from the 19th century onward), followed by
introduction into San Francisco Bay (again, after the collecting
reported by Light) via coastal shipping or coastwise transport
of fisheries products (e. g. with bait, or oysters shucked at
a bayside restaurant with the shells discarded in the Bay, or
spoiled oysters or crabs (we found Bowerbankia on the shell
of a live crab in Humboldt Bay) dumped in the Bay). Bowerbankia
gracilis is common on oyster beds in the western Atlantic
(Wells, 1961; Maurer & Watling, 1973) and has been reported
from ships' hulls (WHOI, 1952). Introductions of B. gracilis
may continue with fisheries products (Miller, 1969, found a Bowerbankia
sp. on seaweed shipped with lobsters to San Francisco) and conceivably
as small colonies on floating debris in ballast water. Its lecithotrophic
larvae are only briefly planktonic, and thus not likely to be
successfully transported in ballast water.
Bugula "neritina (Linnaeus, 1758)"
This conspicuous red-purple arborescent bryozoan has a broad global distribution in temperate, subtropical and tropical waters, including Japan, Hawaii, Australia, New Zealand, both coasts of Panama, Florida, North Carolina, the Mediterranean, and in the heated effluent from power plants in southern England where it was introduced before 1912 (Okada, 1929; Gordon, 1967; Ryland, 1971; Mook, 1976; Carlton, 1979a; Vail & Wass, 1981). Robertson (1905) and Osburn (1950) reported it as abundant and conspicuous in southern California with a northern limit in Monterey Bay, Carlton (1979a) reported its Pacific coast range as Panama to Monterey Bay, and Ricketts et al. (1985) reported it in fouling from Monterey south. However, its range appears to have recently expanded northward. Kozloff (1983) reported it in San Francisco Bay, stating that it was not native to the region, and we commonly observed it there in 1993 and 1994. It has also been found on the hull of a wooden ship in Humboldt Bay (Carlton & Hodder, 1995), in Coos Bay, Oregon (Hewitt, 1993) and in Friday Harbor, Washington (M. DiMarco-Temkin, pers. comm., 1994).
Bugula neritina has been reported as a common member of fouling communities in harbors and bays, but has also been collected from offshore waters and open coast kelp beds on the Pacific coast. It seems likely that two or more species of red-purple Bugula are present, including both a native warm-water, open coast species and an introduced harbor fouling species.
The origin of this species is unknown, but it was most likely
transported to the northeastern Pacific in hull fouling Bugula
neritina has been frequently collected from ships' hulls (WHOI,
1952; Millard, 1952; Ryland, 1970), and is highly tolerant of
mercury-based anti-fouling compounds (Weiss, 1947). Less likely,
it might have alternatively been introduced with the few shipments
of Atlantic oysters made to southern California in the 19th century
(Carlton, 1979a, p. 97), as it has been reported from oyster beds
in the Atlantic (Wells, 1961). Transport in ballast water is unlikely,
since Bugula neritina, in common with other Bugula
species, has coronate larvae that typically spend less than 10
hours in the plankton before settling (Soule et al., 1980; Woollacott
et al., 1989), though transport as tiny colonies attached to floating
material in ballast tanks, or as colonies attached to the sides
of ballast tanks, might be possible.
Bugula stolonifera Ryland, 1960
SYNONYM: Bugula californica of Pacific coast authors in
reference to certain harbor populations (see below)
The history of this North Atlantic bryozoan remains to be worked out in San Francisco Bay. Soule et al. (1980) reported that "the Bugula californica reported as a fouling organism from ports such as San Francisco Bay and Los Angeles Harbor has recently been recognized as B. stolonifera. Although very similar to B. californica, B. stolonifera is grayish and lacks the distinctive, whorled colony patterns." (Soule & Soule, 1977 (writing in 1975-1976) specifically do not list B. stolonifera for southern California stations.) Okamura (1984) reported B. stolonifera, identified by J. Soule, collected in 1982 from the Berkeley Marina. Bugula californica Robertson, 1905, remains a distinct species, apparently of more open marine conditions (Soule et al., 1980), and we thus take Robertson's (1905) report of B. californica from "Lands End, San Francisco Bay," which is located on the ocean side of San Francisco, to refer to B. californica rather than B. stolonifera.
We tentatively take Soule et al. (1980; writing in 1978) as the first record of B. stolonifera from San Francisco Bay, pending the re-examination of museum collections. A bryozoan reported as B. californica was present in Newport Harbor on dock piles at least by the 1940s (Osburn, 1950), while Reish (1972) reported B. californica to be widespread through Los Angeles-Long Beach Harbors, Alamitos Bay, Marina del Rey, Huntington Harbor, and Newport Bay, based upon collections dating back to 1962. If Bugula stolonifera has not been present an unrecognized in San Francisco Bay for many decades, then it may have first become established in southern California harbors and entered the Bay region in the 1970s via coastal ship traffic.
Bugula stolonifera appears to be native to the northwestern Atlantic and has been introduced to Europe and the Mediterranean (Ryland, 1971), Panama (Soule & Soule, 1977) and Saudi Arabia (Soule & Soule, 1985). Records of Bugula californica in estuarine fouling communities elsewhere in the world (such as Brazil, Hawaii, and Japan (Marcus, 1937; Soule & Soule, 1967; Mawatari, 1956) likely refer to Bugula stolonifera as well. Soule & Soule (1967), in reporting B. californica from the Hawaiian Islands, noted it was "common as a fouling organism on dock pilings and boat hulls (and) it could presumably be spread by boats or floating logs." Bugula californica in the Galapagos Islands may represent a mixture of both the native marine species and B. stolonifera.
We regard B. stolonifera as a probable ship fouling introduction.
As discussed under B. "neritina," Bugulas
are unlikely candidates for introduction in ballast water.
Conopeum tenuissimum (Canu, 1908)
SYNONYMS: probably include Conopeum commensale of Filice,
1959 and of Aldrich, 1961 (north Bay estuarine stations)
This very common western North Atlantic bryozoan occurs in fouling communities, on oyster shells, eelgrass, and many other estuarine substrates from Delaware Bay to the Gulf of Mexico (Dudley, 1973). It was first described as a Holocene subfossil from Argentina (Dudley, 1973) and has also been recorded from West Africa (Cook, 1968) and Sydney, Australia (Vail & Wass, 1981). On the Pacific coast Conopeum tenuissimum has been identified by Patricia Cook from San Francisco Bay (collected since 1951-52; Carlton, 1979a,b) and from Coos Bay, Oregon (collected since 1970; JTC, unpublished). Light's (1941) record of "Membranipora" as a summer invader of Lake Merritt, Oakland, could refer to either or both of C. tenuissimum and the cryptogenic species C. reticulum, as could the U. S. Navy's (1951) report of "Electra sp." on fouling panels at Mare Island in 1944-47 and at Port Chicago in 1945-47.
We collected a Conopeum that we tentatively identify as tenuissimum on docks in the brackish northern part of San Francisco Bay in 1993-1994, where it was particularly conspicuous overgrowing masses of the introduced hydroid Garveia franciscana, and in scattered, small colonies on docks throughout the northern, central and southern parts of the Bay after the wet spring of 1995.
Conopeum tenuissimum has planktotrophic larvae and thus
might have been introduced in ballast water. Alternatively it
could have been introduced in ship fouling or with Atlantic oysters
(with which it occurs; Maurer & Watling, 1973), perhaps as
early as the 19th century.
Cryptosula pallasiana (Moll, 1803)
This Atlantic bryozoan has been reported in the eastern Atlantic from Norway and Great Britain to Morocco and in the Mediterranean and Black Seas (Osburn, 1952; Ryland, 1971, 1974), in the western Atlantic from Nova Scotia to North Carolina (Osburn, 1952) and Florida (Winston, 1982), and has been introduced to Japan (Mawatari, 1963), New Zealand (Gordon, 1967) and Australia (Ryland, 1971; Vail & Wass, 1981). Osburn (1952) noted that it was not recorded by early Pacific coast bryozoan workers (except for a single questionable 1925 record from Homer, Alaska). Between 1943 and 1972 it was reported from various southern California bays, from offshore southern California waters to 35 meters depth, and from Mexican waters. It was collected from Monterey Bay in 1952, Vancouver Island, British Columbia in 1970, Bodega Harbor in 1975 (Carlton, 1979a, p. 720) and Coos Bay, Oregon in 1988 (Hewitt, 1993). The U. S. Navy (1951) reported a Cryptosula sp. (presumably pallasiana) from Hunters Point Shipyard in San Francisco Bay in 1944-47, Banta (1963) reported C. pallasiana from the Berkeley Yacht Harbor in 1963 (believing it to be the first central California record), and we observed small colonies on shells and floating docks at a few scattered sites in San Francisco Bay in 1994-95.
Cryptosula was likely introduced to the eastern Pacific
either as hull fouling or with shipments of Atlantic oysters,
with which it occurs on the Atlantic coast (Wells, 1961). It has
lecithotrophic larvae that spend a very short time in the plankton,
and thus is a poor candidate for interoceanic transport by ballast
water.
Schizoporella unicornis (Johnston, 1847)
SYNONYMS: Schizopodrella unicornis
This conspicuous, orange-colored, western Pacific encrusting bryozoan was not reported on the eastern Pacific coast by early bryozoan workers, as noted by Osburn (1952). It has been reported in various embayments and shore locations in Washington state since 1927, in California since 1938, in British Columbia since 1966 (Carlton, 1979a, p. 723), and in Coos Bay, Oregon since 1986 (JTC, unpublished). S. unicornis has also been reported from Baja California and the Galapagos, and from offshore sites in southern California, but as discussed by Carlton (1979a), these and some other southern California records may be properly referred to the Atlantic species S. errata, or to a third Schizoporella species.
In San Francisco Bay Schizoporella unicornis was recorded
from the Berkeley Yacht Harbor in 1963 (Banta, 1963), and we collected
it from various locations in the Bay in 1970 and 1993-95. Though
we never found it abundant, Kozloff (1983) described it as the
most common encrusting bryozoan in the Bay. It is often found
encrusting on shells and has been frequently reported as fouling
on ship hulls (WHOI, 1952), and thus may have been introduced
to the northeastern Pacific either with shipments of Japanese
oysters (Crassostrea gigas)or as hull fouling. Like
many other bryozoans, it has lecithotrophic larvae with a brief
planktonic phase, and is unlikely to have been carried across
the Pacific in ballast water.
Victorella pavida Kent, 1870
This "cosmopolitan" bryozoan has been reported from many, widely-dispersed sites and from the bottoms of vessels. Reviewing its global distribution, Carlton (1979a) suggested that it was native to the Indian Ocean and introduced via hull fouling to Europe (first reported in the late 1860s), eastern North America (by 1920), Japan (by 1943) and eastern South America (by 1947). A 1955 record from the Salton Sea has now been recognized by Jebram & Everitt (1982) as representing a distinct species, Victorella pseudoarachnida.
It was collected in Lake Merritt in San Francisco Bay in 1967,
though relatively inconspicuous mats of Victorella could
have been present for many years before they were noticed. Thus
this introduction could have resulted from the importation of
Japanese oysters (in the 1930s), from the importation of Atlantic
oysters (from the 1870s to the 1930s), or from transport as hull
fouling (it has been reported from the bottoms of boats; Osburn,
1944). Transport in ballast water is unlikely, as Victorella's
lecithotrophic larvae are only briefly planktonic.
Watersipora "subtorquata (d'Orbigny, 1852)"
Since the 1960s two species of Watersipora have appeared in California where none were previously known. These species are distinguished from each other by the shape of the proximal border of the aperture, with the border curving into the aperture in W. arcuata (=nigra) and curving outward to form a sinus in W. "subtorquata." The identification of the latter species remains uncertain (the one or more species with a sinusoid aperture have been variously referred to W. subtorquata, subovoidea, cucullata, atrofusca, aterrima and edmundsoni) due to the variability in the characters used to distinguish sinusoid species and the unstable taxonomy of the genus (Gordon (1989), for example, referred to it as "a taxonomic 'can of worms'").
W. arcuata was collected in southern California embayments from San Diego to Santa Monica beginning in 1964 (although the first collection is reported in the literature as 1967; W. Banta, pers. comm., 1994). W."subtorquata" was first collected in southern California in 1963 (although the first clear report of its collection in the literature is 1989; W. Banta pers. comm., 1994), in Drakes Estero in 1984 (J. Goddard, pers. comm., 1995) and in Coos Bay, Oregon in 1990 (C. Hewitt, pers. comm., 1990) (where, however, we did not find it in 1995). We found W. "subtorquata" in San Francisco Bay in 1992, and in Bodega Harbor, Tomales Bay, Half Moon Bay, Moss Landing Harbor and Monterey Harbor in 1993-95. In San Francisco Bay it was common as flat circular colonies on docks and rocks in the South and Central bays and the southern part of San Pablo Bay, and growing in 10 cm thick "reefs" on docks near the mouth of San Francisco Bay in 1993 and 1994. After an unusually wet spring, we found only dead or dying colonies in San Francisco Bay in 1995.
Watersipora specimens with a sinusoid aperture, belonging
to one or more species, have been reported from many parts of
the world. The native region of W. "subtorquata"
is thus unknown, although its distribution and spread suggests
the northwest Pacific as the likeliest origin, with populations
introduced (if these are the same species) to American Samoa,
Hawaii, the Galapagos Islands, western Mexico, Australia, New
Zealand, the Carribean, Brazil, the Mediterranean, the Red and
Arabian seas and the Atlantic coast of France. Watersipora species
have coronate larvae which remain in the plankton for less than
a day before settling (Mawatari, 1952; Wisely, 1958), and thus
could not have been transported long distances as larvae in currents
or in ballast water. Transport as fouling on ship hulls seems
most likely, as Watersipora has been frequently found both
in fouling and on ship bottoms (WHOI, 1952; Ryland, 1970), and
is highly tolerant of copper-based anti-fouling compounds (Weiss,
1947; WHOI, 1952; Allen, 1953; Ryland, 1970).
Zoobotryon verticillatum (Delle Chiaje, 1828)
SYNONYMS: Zoobotryon pellucidum
The origin of this subtropical ctenostome bryozoan is unknown. Alice Robertson (1905) reported it in Japan, Hawaii and in abundance in Madras Harbor, India, and noted that it occurred in abundance in San Diego Bay in the summer of 1905, where, "in water of 10 or 12 feet deep, it grew in luxuriant masses of a green tint, the whole resembling clumps of freshly cut hay" (Robertson, 1921). Such large colonial masses (to 1 m x 2 m) can still be found in San Diego and Mission bays, colonized by anemones and shading out and killing eelgrass (A. Sewell, pers. comm., 1995). Osburn (1940; cited in Osburn, 1953) described it as circumtropical, and added records from the Mediterranean, Bermuda, Florida, Puerto Rico, the Gulf of Mexico and Brazil. Soule et al. (1980) report its northeastern Pacific ranges as extending from San Diego to the Gulf of California and Central America, and "in recent years" in harbors north to Los Angeles. It has also been collected in New Zealand (Gordon, 1967) and Australia (Vail & Wass, 1981).
Zoobotryon was collected in Redwood Creek in South San
Francisco Bay in 1993, where it was abundant and producing active
larvae (K. Wasson, pers. comm.). It is a common hull fouling organism
in warm waters (WHOI, 1952; Ryland, 1970), which was its likely
mechanism of introduction to California.
Ascidia sp.
This introduced tunicate of unknown origin has been collected off and on since 1983 in harbors from San Diego to Los Angeles (G. Lambert, pers. comm., 1995), and in 1993-94 we found it (identified by G. Lambert), sometimes very abundant in fouling on floating docks, from Richmond to San Leandro on the east shore and from Redwood Creek to Pier 39 on the west shore of San Francisco Bay. We know of only one earlier record of an Ascidia species in San Francisco Bay, which was collected at Tiburon and possibly in the Berkeley Marina in 1981 (B. Okamura, pers. comm., 1995). The specimens, no longer extant, were identified at the time as the native species A. ceratodes.
Ascidia species have been reported from ship fouling (Stubbings,
1961) which may have been the transport mechanism for this species.
Alternatively, it may have arrived via ballast water, since some
solitary ascidians have planktonic stages (from fertilized egg
through tadpole) that last two weeks or more (as discussed below
under Ciona intestinalis). In San Francisco Bay we sometimes
found the amphipod Leucothoe sp., here considered to be
introduced, living within the body cavity of this Ascidia.
Botryllus schlosseri (Pallas, 1774)
Botryllus aurantius Oka, 1927 (=Botrylloides violaceus)
Botryllus sp. (large zooid) (=Botrylloides sp.)
We consider at least three species of botryllid ascidians to be introduced into San Francisco Bay. All three are locally common to abundant members of Bay fouling communities, sometimes forming extensive gelatinous masses. The genus- and species-level systematics of the common, harbor-dwelling, fouling botryllids are matters of considerable complexity (Carlton, 1979a; Monniot & Monniot, 1987; Monniot, 1988) and the species-level identification of all three of the species treated here remains uncertain or unknown. Most American literature refers the common fouling species to two genera, Botryllus and Botrylloides. Monniot & Monniot (1987) and Monniot (1988) have, however, discussed the purported distinctions between these two genera and offer compelling reasons why Botrylloides should be synonymized under Botryllus, an approach we follow here.
A common botryllid of San Francisco Bay with star-shaped or oval clusters of zooids we tentatively refer to as Botryllus schlosseri, a common North Atlantic species which Van Name (1945) regarded as native to Europe and introduced to the western Atlantic in ship fouling. This species has up to about 20 functional zooids arranged in stellate clusters around a central, common exhalant opening. Morphologically, it is virtually identical to the B. schlosseri of Long Island Sound (JTC pers. obs.; C. Hewitt, pers. comm., 1992).
A second botryllid found in San Francisco Bay, also with star-shaped or oval clusters of zooids, keys out to Botryllus tuberatus Ritter & Forsyth, 1917 (S. Cohen, pers. comm., 1994). Van Name (1945) reported this species, described from Santa Barbara, to be confined to southern California. Abbott & Newberry (1980) reported its occurrence from Bodega Bay to San Diego and in Japan, in the Philippines, on the Asian mainland, and on several Pacific islands. We consider this botryllid, at least in central California, to be cryptogenic.
Yet another botryllid, also very common in San Francisco Bay, has dozens of small zooids arranged in meandering (serpentine) chains and appears identical to Coos Bay material that Hewitt (1993) referred to the Japanese native Botrylloides violaceus Oka, 1927. Boyd et al. (1990) also identified Monterey Bay material as Botrylloides violaceus. Monniot (1988, p. 169) has noted that the name "violaceus" for a botryllid is preoccupied at least twice before Oka's usage, and that the proper name for this species is Botryllus aurantius. This species is illustrated in Morris et al. (1980), figure 12.30, based upon a slide taken by JTC ("J. Carlson") at Nahcotta, Willapa Bay, Washington.
Finally, we collected another botryllid with chain zooids in San Francisco Bay in 1993 and 1994, but with each zooid typically twice the size of those in B. aurantius. This appears to be a fourth species (S. Cohen, pers. comm., 1993). It is illustrated in Kozloff (1983; plate 29, as Botrylloides) based upon material from San Francisco Bay.
The failure of Van Name (1945) to record any botryllid sea squirt north of southern California, and its absence from all faunal accounts of the marine invertebrate biota of the Pacific coast from Monterey Bay north until the mid-1940s, suggests that these now extraordinarily abundant sea squirts have been introduced. Botryllus schlosseri was first recorded in San Francisco Bay from fouling panels at the Mare Island and Hunters Point naval bases in 1944-1947 (US Navy, 1951), although it evidently remained sufficiently rare or localized in the Bay to escape the attention of Smith et al. (1954). Botryllus aurantius was present in San Francisco Bay by at least 1973 (JTC, pers. obs.). Botryllus sp. ("large zooid") was photographed at the Berkeley Marina by Eugene Kozloff in the late 1970s or early 1980s (Kozloff, 1983, plate 29; E. Kozloff, pers. comm., 1994).
Botryllus species have frequently been reported from ship fouling (WHOI, 1952). Botryllus schlosseri was introduced to the Bay either with Atlantic oysters or on ship fouling. Botryllus aurantius may have been introduced with Japanese oysters or on ship fouling (although the latter would not have been a likely mechanism from Japan until after World War II, further suggesting a post-1940s arrival if with ships). Botryllus sp. may also have entered with Japanese oysters or ship-fouling. No similar large-zooid botryllid is known from the American Atlantic coast.
The distribution of all three of these species remains to be worked out on the Pacific coast. Tunicates similar to Botryllus schlosseri are known from at least Monterey Bay to British Columbia (Boyd et al. 1990; Carlton, 1979a; Hewitt, 1993; JTC, pers. obs.). Tunicates similar to Botryllus aurantius are known from Monterey Bay to British Columbia (Boyd et al., 1990; Carlton, 1979a; JTC, pers. obs.) and may now be present in southern California as well (Carlton, 1979a). The large-zooid Botryllus is at present known only from San Francisco Bay and Pillar Point Harbor in Half Moon Bay, San Mateo County.
Ciona intestinalis (Linnaeus, 1767)
SEA VASE
Ciona intestinalis is one of the most widely distributed ascidians in the world, recorded from the tropics to the subarctic. It was first described from Europe and appears to be native to one or both sides of the North Atlantic Ocean. It was reported in the northeastern Pacific at San Diego in 1897, followed decades later by collections in San Francisco Bay in 1932, Newport Bay in 1934, several other southern California bays from the 1950s to the 1970s, and Monterey Harbor in 1974 (Carlton, 1979a, p. 732). There are intermittent records from Vancouver Island, British Columbia in 1908-09, the 1930s (Carlton, 1979a) and in recent years (G. Lambert, pers. comm., 1995). As discussed by Carlton (1979a), there are no records of C. intestinalis from Oregon, and the few Washington and Alaska records are doubtful.
Ciona intestinalis is a common fouler of ships (WHOI, 1952; Stubbings (1961) provides a photograph of a ship in drydock whose hull is completely covered by C. intestinalis), which was probably the initial means of transport to the Pacific coast. Later introductions could have occurred via ballast water: although the ascidian larval phase, known as a tadpole, typically lasts only a few hours, some solitary ascidians including Ciona intestinalis have total planktonic phases (from release of gametes through settlement of tadpole) that can last two weeks or more. Carlton & Geller (1993) found ascidian tadpole larvae in the ballast water of five Japanese wood chip carriers that had completed transpacific voyages of 13 to 16 days, some of which were reared to Ciona sp. (JTC, unpublished). Carlton & Geller (1993) also found metamorphosed ascidians settled on floating wood chips in their ballast water samples.
In San Francisco Bay we have found the amphipod Leucothoe
sp., here considered to be introduced, living within the body
cavity of Ciona.
Ciona savignyi Herdman, 1882
In our survey of San Francisco Bay fouling in 1993-94 we found
both Ciona savignyi (identified by G. Lambert) and C.
intestinalis, the former distinguished from the latter by
the presence of flecks of white or yellow pigment in the body
wall and the absence of any red pigment at the end of the vas
deferens. Like Ciona intestinalis, C. savignyi was
likely transported to San Francisco Bay as ship fouling or in
ballast water. It has been collected from Long Beach and other
southern California marinas by C. Lambert since 1986, when it
already was abundant, and is now found from San Diego to Santa
Barbara. It is probably native to Japan (G. Lambert, pers. comm.,
1995).
Molgula manhattensis (DeKay, 1843)
This tunicate occurs on both sides of the North Atlantic Ocean, from Maine to Louisiana (Van Name, 1945) and from northern Norway to Portugal (Millar, 1966). Van Name (1945) reported it as the commonest solitary tunicate on the coast between Massachusetts and Chesapeake Bay. It was first recorded in the Pacific from Tomales Bay in 1949, was "widespread in San Francisco Bay in the 1950s," and collected in Coos Bay, Oregon in 1974, and in Bodega Bay (Abbott & Newberry, 1980). As noted by Carlton (1979a), there is also a questionable record from San Felipe in the Gulf of Mexico. It has also been introduced to Europe from the White Sea to the Adriatic Sea, northwestern Africa, Japan and Australia (Abbott & Newberry, 1980).
In San Francisco Bay, Molgula has been collected from the South Bay, along the eastern shore of the Central Bay, in San Pablo Bay and upstream to Martinez and Grizzly Bay, at concentrations of up to 100-2,400/square meter (Hopkins, 1968; Markmann, 1986). Ganssle (1966) reported it (as M. verrucifera) in 1963-64 as "so abundant in San Pablo Bay bottom tows that it was impossible to haul the trawl aboard by hand." It is apparently the most low-salinity-tolerant tunicate in the Bay: it ranges further upstream than the others and was virtually the only tunicate we collected in the Bay in the summer of 1995 following an unusually wet spring. It is also reputed to be highly tolerant of municipal and industrial pollution (Van Name, 1945; Carlton, 1979a; Abbott & Newberry, 1980).
Molgula could have been transported to central California
in ship fouling (from which it has been frequently reported; WHOI,
1952), with oyster shipments (Wells (1961) and Maurer & Watling
(1973) reported Molgula manhattensis from Atlantic oyster
beds, and we have often found it attached to shells dredged from
the bottom of San Francisco Bay; eastern oysters (Crassotrea
virginica) were being planted in both Tomales and San Francisco
bays in the 1940s), or, as discussed above under Ciona intestinalis,
in ballast water.
Styela clava Herdman, 1881
SYNONYMS: Styela barnharti
Styela clava is native to the western Pacific from the Sea of Okhotsk south to Shanghai, and though present in California since at least the 1930s was not recognized as the Asian species until the 1970s. It was collected at Newport Bay in 1932-33, in Elkhorn Slough (a single small specimen) in 1935, in San Francisco Bay in 1949, in Mission Bay in 1959, in Monterey Harbor in 1961, in several bays from San Diego to Morro Bay in the early 1970s, in Coos Bay, Oregon in 1993-94 (R. Emlet, A. Moran, pers. comm.), and in 1994-95 at a marina north of Nanaimo, British Columbia, but not at other sites on the eastern shore of Vancouver Island (G. Lambert, pers. comm., 1995). It has also been introduced to northwestern Europe, northeastern United States and Australia (Abbott & Newberry, 1980).
Styela clava is a common fouling organism in harbors and may have been transported to the Pacific coast as ship fouling. However, since it has also been reported from fouling associations in Japanese oyster farms (Carlton, 1979a) and Japanese oysters (Crassostrea gigas) were planted in Elkhorn Slough from 1929-1934 (Bonnot, 1935b), it could have crossed the ocean with oyster shipments and been transported to Newport Bay with coastal shipping. As noted above under Ciona intestinalis, it could also have been introduced in ballast water.
Styela clava is harvested and eaten in southern Korea,
where it is called "mideuduck." In Japan it has been
blamed for an asthmatic condition in oyster shuckers, apparently
caused by an allergenic reaction when Styela-fouled oysters
are hammered open in poorly-ventilated work areas. (Abbott &
Newberry, 1980).
Acanthogobius flavimanus (Temminck & Schlegel, 1845)
[GOBIIDAE]
YELLOWFIN GOBY, MAHAZE
The yellowfin goby is native to Japan, South Korea and China where it ranges from marine into fresh water near sea level (Brittan et al., 1963; Haaker, 1979). It is reportedly catadromous in Japan, moving downstream onto saline mudflats to spawn (Herbold & Moyle 1989).
The first yellowfin goby in California was collected in Jan. 1963 in a midwater trawl in the San Joaquin River off Prisoners Point, Venice Island. The fish measured 155 mm total length, and was estimated to be entering its second year (Brittan et al., 1963). Brittan et al. (1963) suggested that the goby was transported across the Pacific in the fouled seawater system of a ship, and Haaker (1979) suggested the possibility of transport as eggs laid on fouling organisms on ships' hulls. Eschmeyer et al. (1983) proposed transport in ballast water or with live seed oysters (presumably as eggs). However, except for occasional experimental plants, Japanese oysters have not been planted in San Francisco Bay since the 1930s (Carlton, 1979a).
The goby was widespread throughout the Bay and Delta area by 1966 (Brittan et al., 1970) and is now well established in central and southern California (Eschmeyer et al., 1983). Common throughout the Bay and Delta, it has been collected from: lagoons around the Bay such as Foster City Lagoon, Berkeley Aquatic Park and Lake Merritt, and the salt ponds at Alviso; the Delta north to the Sacramento Ship Channel almost to the Port of Sacramento, and south to the Tracy Pumping Plant and the Stockton Deepwater Channel; the Delta-Mendota Canal at Newman, and the San Luis Reservoir in Merced County; and Contra Loma Reservoir in Contra Costa County (Brittan et al., 1970; McGinnis, 1984; ANC & JTC, pers. obs.). It was reported from Elkhorn Slough (Kukowski, 1972) and Tomales Bay and Estero Americano (Miller & Lea, 1976), and one specimen was collected from Bolinas Lagoon (Brittan et al., 1970). McGinnis (1984) reported that it was expanding its range in central coastal California.
In southern California the yellowfin goby was photographed in Los Angeles Harbor on Sept. 22, 1977 and collected from Long Beach Harbor on Mar. 29, 1978. It has also been collected from Upper Newport Bay and the San Gabriel River (Haaker, 1979), and south as far as San Diego and perhaps into Mexico (Courtenay et al., 1986). The largest specimen reported in California, with a total length of 234 mm, was taken from Berkeley Aquatic Park (Brittan et al., 1970). The goby has also been introduced to Sydney Harbor, Australia (Miller & Lea, 1976).
The goby is considered a delicacy in Japan (Eschmeyer et al.,
1983), but in the Bay Area it is known to be used only for bait,
primarily for striped bass. It supports a commercial trap fishery,
and individual anglers catch it by hook-and-line.
Alosa sapidissima (Wilson, 1811) [CLUPEIDAE]
AMERICAN SHAD, ATLANTIC SHAD
SYNONYMS: Clupea sapidissima
Shad are native to the Atlantic coast from Labrador to Florida (Page & Burr, 1991). They were the first fish successfully introduced into California. In June 1871, about 10,000 Hudson River shad fry, which had been carried across the country in four 8-gallon milk cans by Seth Green of the California Fish Commission, were planted in the Sacramento River at Tehama (Lampman, 1946). A second shipment was lost in June 1873 when a railroad bridge over Nebraska's Elkhorn River collapsed and the aquarium car was destroyed. A third shipment of 35,000 fry was successfully planted on July 1873. The U. S. Fish Commission made several other shipments from 1876 to 1881, with all the fry, totaling 829,000, planted in the Sacramento River at Tehama (Skinner, 1962; Stevens, 1972; Nidever, 1916, and Shebley, 1917, report the total as 619,000). A few mature shad were taken from San Francisco Bay by 1873, and shad were found in the Columbia River by 1876. (Nidever, 1916; Shebley, 1917). The population spread rapidly to other estuaries from Baja California to Alaska and as far away as Kamchatka, through a combination of ocean migration and intentional transplants (Herbold et al., 1992).
Several researchers have suggested that shad and striped bass did well in the Delta watershed in the late 1800s because their drifting eggs were not smothered by sediment from gold mining operations, as presumably were the sinking or attached eggs of native fish; and because they spawned in the main river channels while the native salmonids spawned in smaller tributary streams that were more extensively disrupted by mining activities (Herbold et al., 1992; Blount, 1994). In any event by 1874 shad were numerous enough to support a small commercial harvest, and by 1880 the "catch had to be curtailed to keep from glutting the market" (Skinner, 1962). Between 1900 and 1945 the catch was frequently over a million pounds, peaking at 5.7 million pounds in 1917 (Skinner, 1962; Herbold & Moyle, 1989). By 1953, however, Roedel described the shad as a minor commercial species taken with gill and trammel nets with Pittsburg accounting for most of the landings, which totaled about 0.4-1.3 million pounds annually during the 1950s (Skinner, 1962). It is unclear, however, whether the reduced catch was due to a declining stock or a weak market. Most of the sport fishing at that time was done with dipnets, and was referred to as the "bump net" fishery. The commercial fishery was eliminated in 1957 when the California legislature banned gill-net fishing within the Golden Gate to avoid competition with sportfishing.
In the early decades of the fishery virtually all of the shad were sold in local fresh markets. Then for a while after 1912 most of the fish were salted and exported to China (Nidever, 1916). By the 1950s most of the meat was again sold fresh, though the main value of the fishery was in the roe, which was salted, canned or sold fresh (Roedel, 1953).
Today, spawning runs are found on the Sacramento, Feather, Yuba, American, Mokelumne, Stanislaus and San Joaquin rivers in the Delta watershed, and in the Russian, Eel and Klamath rivers in northern California. There are also shad in Millerton Lake in Fresno County, San Luis Reservoir in Merced County, and in other waters of the Central Valley irrigation system (McGinnis, 1984). Stevens (1972) reported "crude" estimates of over 750,000 shad running on the Sacramento River based on trap data, and between 2 and 4 million fish based on past commercial catch records. Herbold et al. (1992) reported estimates of 3.04 million fish in 1976 and 2.79 million in 1977 on the Sacramento River, with populations probably 2-3 times as large early in the century. Emmett et al. (1991) estimated the combined run in all Delta tributaries at 0.7-4.0 million shad per year.
Studies have shown adult shad to be wide-ranging travelers, with some individuals caught 3,000 km from the tagging site (Emmett et al., 1991), but little is known of their life in the Pacific Ocean. The males usually mature in three years and the females in four. The mature fish migrate upstream between February and June, with the peak migration occurring in March or April. Before the construction of the Red Bluff Dam in 1967, some shad traveled more than 300 miles up the Sacramento River (Nidever, 1916; Smith & Kato, 1979). Most spawning takes place between April and June, with temperatures generally between 14° and 24°C, although spawn survival is poor at the higher temperatures. On the Pacific coast most adults die after spawning, which may be related to high water temperatures (Stevens, 1972; Moyle, 1976a; Emmett et al., 1991).
Moyle (1976a) reports that spawning females release 30,000-300,000 eggs (on the Atlantic coast, shad are reported as spawning 116,000 to 4,680,000 eggs (Skinner, 1962)). The eggs can tolerate 7.5-15 ppt salinity depending on temperature, with optimal temperatures of 16-27°C., and hatch in 3-6 days (Emmett et al., 1991). Juveniles are found in abundance in the Delta in late summer and fall, with most moving downstream into brackish water by the winter (Skinner, 1962; Moyle, 1976a).
Young shad are reported to feed on zooplankton, primarily cladocerans and copepods, with adults in the Delta feeding on Neomysis mercedis, along with cladocerans, copepods and amphipods, and an occasional clam or larval fish. The adults cease feeding once they enter the main rivers (Stevens, 1972; Moyle, 1976a). The stomachs of coastal shad were found to contain anchovies and euphausids (Skinner, 1962). Juvenile shad are prey for salmonids, striped bass, other fish, birds and harbor seals (Emmett et al., 1991).
Curtis (1942) stated that "no detrimental effects are reported
for this fish...It seems to be possible to point to this species
as the one case which has caused no complaint from any quarter.
It has apparently found an ecological niche which was not only
completely unoccupied but also large enough to accommodate an
enormous population." Emmett et al. (1991) concluded that
the introduction of shad "does not appear to have displaced
natives, but competition may occur."
Ameiurus catus (Linnaeus, 1758) [ICTALURIDAE]
WHITE CATFISH, SCHUYLKILL CAT, FORKED-TAIL CATFISH, COMMON CATFISH
SYNONYMS: Ictalurus catus
White catfish are native to coastal streams from New York to Mississippi (Page & Burr, 1991). In 1874 Livingston Stone of the U. S. Fish Commission planted 54 (or 56) large white catfish from the Raritan River, New Jersey (along with 18 unidentified catfish from the Elkhorn River in Nebraska) in the San Joaquin River near Stockton (Smith, 1896; Shebley, 1917). In 1875, the California Fish Commission reported that these fish had grown rapidly and spawned, and predicted that they would be numerous enough to support a commercial fishery by the following year. By 1877 the Commissioners reported that the descendants "already furnish an important addition to the fish food supply of the city of Sacramento" and had 8,400 of them distributed to water bodies in 13 counties. In 1879, the Commissioners reported that white catfish had increased to the millions and furnished "an immense supply of food," and they had 39,000 of them distributed to 22 counties (Smith, 1896). By 1900 the fishery was large enough to ship catfish to Mississippi (Cohen, 1993). The commercial fishery was abolished in 1953 when the catfish population appeared to be overfished (Miller, 1966a; Borgeson & McCammon, 1967).
The white catfish occurs in San Diego County and possibly other parts of southern California, and in Clear Lake, and is common in warm water lakes and slow moving areas of large rivers in the Central Valley (Curtis, 1949; McGinnis, 1984). It is said to be the most popular warmwater sportfish in California (Herbold & Moyle 1989), with the angling effort in the Delta in 1962-1963 estimated at almost 450,000 angler days (Miller, 1966a). It is the most abundant species of catfish in the Delta, accounting for 97% of 26,000 catfish collected in the Delta in 1963-1964. Young white catfish were taken mainly in channels in the southern and eastern Delta; adults were most abundant in dead-end sloughs, flooded islands, and the San Joaquin River below Stockton (Turner, 1966a). The white catfish also occurs downstream to Suisun Bay in salinities of 8 ppt (Ganssle, 1966; Herbold & Moyle 1989).
White catfish collected from Clear Lake in 1943 had eaten hitch,
sculpin, bluegill, tule perch, black crappie, frogs, insects,
clams, and the remains of carp and coot (Miller, 1966a). The stomachs
of white catfish collected in 1953-1954 from the Delta contained
Corophium, American shad, plant and animal debris, unidentified
fish, insects, clams, the crayfish Pacifastacus, and Neomysis
(Borgeson & McCammon, 1967). The stomachs of catfish collected
in 1963-1964 from the Delta contained several introduced fish
and invertebrates (threadfin shad, American shad, striped bass,
bluegill, Corbicula fluminea, Rithropanopeus harrisii)
and other interesting food items (terrestrial slugs, earthworms,
small birds and mammals, a lizard, a pair of coot feet) (Turner,
1966a). Curtis (1942) described the white catfish and the brown
bullhead as "scavengers and to some extent predators upon
the eggs and young of many other fish." He and Smith (1896)
noted that some believed them responsible for the decline in Sacramento
perch (which others have blamed on introduced striped bass, black
bass or sunfish), and that they inhibit trout populations in high
mountain waters. BDOC (1994) noted that white catfish can destroy
the spawning sites of native fish by preying on eggs, larvae and
juveniles.
Ameiurus melas (Rafinesque, 1820) [ICTALURIDAE]
BLACK BULLHEAD
Synonyms: Ictalurus melas
Black bullhead originally ranged from southern Saskatchewan and
Montana to the upper tributaries of the St. Lawrence River and
Hudson Bay, and south to Texas, northern Mexico and Alabama (Page
& Burr, 1991). They were probably introduced to California
along with several other species of catfish in 1874 (Miller, 1966c;
Moyle, 1976b). They are present in most major rivers and in some
low and middle elevation reservoirs in California, often in shallow
and silty water, including the Colorado, Kern and Kings rivers
(Curtis, 1949; Miller, 1966c; McGinnis, 1984), and are reported
as common in the Delta (Herbold & Moyle, 1989). In 1963-1964
only 100 out of 26,000 catfish (0.4%) collected in the Delta were
black bullhead, with most of them taken from the quiet waters
of dead-end sloughs in the eastern and southwestern Delta (Turner,
1966a); one was collected downstream in Honker Bay (Ganssle, 1966).
Black bullhead are exceptionally tolerant of high water temperatures,
low oxygen and high carbon dioxide levels. They eat insects, crustaceans,
worms, mollusks, fish eggs, fish and plants (Miller, 1966c; McGinnis,
1984).
Ameiurus natalis (Lesueur, 1819) [ICTALURIDAE]
YELLOW BULLHEAD
Synonyms: Ictalurus natalis
Yellow bullhead originally ranged from North Dakota to the St. Lawrence River drainages and south to eastern Oklahoma, Texas and northern Mexico (Page & Burr, 1991). Neale (1915) and Moyle (1976b) reported them introduced into California in 1874, although Miller (1966d) reported them introduced to the Colorado river "before 1942" but absent elsewhere in California.
They are now reported as common in the Colorado River and rare
in warm, clear, low elevation waters elsewhere in California and
in the Delta (McGinnis, 1984; Herbold & Moyle 1989). The yellow
bullhead is basically a stream dweller, and feeds on fish and
crayfish more than do other bullheads (McGinnis, 1984).
Ameirus nebulosus (Lesueur, 1819) [ICTALURIDAE]
BROWN BULLHEAD, COMMON BULLHEAD, HORNED POUT, HORNPOUT, SQUARE-TAIL
CATFISH, BULLHEAD CATFISH
Synonyms: Ictalurus nebulosus
Brown bullhead originally ranged from southern Saskatchewan, the Great lakes, Hudson Bay and Nova Scotia south to Louisiana and Florida (Page & Burr, 1991), and have been introduced widely in western North America (Emig, 1966e). In 1874 Livingston Stone of the U. S. Fish Commission planted 70 brown bullhead from Lake Champlain, Vermont in ponds and sloughs near Sacramento (Smith, 1896; Shebley, 1917). In 1875 the California Fish Commissioners reported that these fish had become so abundant that the population could not be exhausted by fishing, and they had nearly a thousand of them caught and transplanted to other waters (Smith, 1896). Within a few years they had spread throughout the Delta (Emig, 1966e).
In 1963-1964, only 89 out of 26,000 catfish (0.3%) collected from the Delta were brown bullhead, with most of them taken from the quiet waters of dead-end sloughs in the southwestern and eastern Delta (Turner, 1966a); one was collected downstream in Grizzly Bay (Ganssle, 1966). Today brown bullhead are found in warm water habitats throughout California (Emig, 1966e; McGinnis, 1984), and are reported as common in the Delta (Herbold & Moyle 1989).
Pat O'Brien of CDFG reports that 2 to 3 high elevation lakes in
California are taken over each year by illegally planted brown
bullhead and golden shiner. Curtis (1942) described this catfish
and the white catfish as "scavengers and to some extent predators
upon the eggs and young of many other fish." He noted that
some believed them responsible for the decline in Sacramento perch
(which others have blamed on introduced striped bass, black bass
or sunfish), and that they inhibit trout populations in high mountain
waters.
Carassius auratus (Linnaeus, 1758) [CYPRINIDAE]
GOLDFISH
The goldfish, native to China, was the first exotic fish to be introduced into North America, some time in the late 1600s. It has been collected in the wild from every state except Alaska, and is clearly established in 27 states and 2 Canadian provinces (Courtenay et al., 1986). It was introduced to California waters some time after 1900, probably as a released pet (Moyle, 1976b; McGinnis, 1984). Goldfish may be found in any low or medium elevation habitat in California, and some small lakes, such as Lake Temescal, Alameda County, have been completely overrun by goldfish (McGinnis, 1984). Goldfish are common in the Delta (Herbold & Moyle 1989), where they made up 420 of 12,400 cyprinids (3%) collected in 1963-1964. These were mainly taken in Indian Slough and at Mossdale on the San Joaquin River (Turner, 1966c), but they have been occasionally caught downstream to Honker Bay (Ganssle, 1966). Most of the goldfish in the Delta migrate upriver to fresher water to breed (Herbold & Moyle, 1989).
Goldfish grow to 40 cm, and females may lay up to 15,000 eggs
per year. They primarily feed on plankton and bottom organic debris,
and thus compete for food with fry of other species (McGinnis,
1984).
Cyprinus carpio Linnaeus, 1758 [CYPRINIDAE]
COMMON CARP
Carp, native to Eurasia, were first introduced into North America in the Hudson River in 1831 (Courtenay et al., 1986). In 1872 Julius Poppé imported 5 carp from Holstein, Germany and, stocking them in his pond in Sonoma County, "did a thriving business for a number of years, selling their progeny for purposes of propagation." In 1877 the California Fish Commission traded trout eggs for 88 young carp from the Japanese government, and began its own carp rearing program. In 1879 the U. S. Fish Commission shipped 298 carp to California, planting 60 in Sutterville Lake and the rest in a private pond in Alameda County to be "at the disposal of the State Commission" (Smith, 1896). These fish may have come from a carp rearing program in Washington, D. C. which, beginning with 338 carp from Germany in 1877 and accompanied by a national ad campaign, supplied carp to government agencies throughout the country (see McGinnis, 1984, for a description of "carp fever"). In 1882 the U. S. Fish Commission began delivering carp to private applicants, and in 1883 the California Fish Commission purchased 600 German carp from J. V. Shebley, a fish-culturalist in Nevada County, and planted them in the Sacramento River near Sacramento (Shebley, 1917; McGinnis, 1984; Herbold et al., 1992).
By the early 20th century, carp were reported from "nearly all public and private waters of the state" (Shebley, 1917). Today they are present in most freshwater habitats in California other than the Klamath River drainage (McGinnis, 1984), and are abundant in the Delta (Herbold & Moyle 1989) where they are found down into brackish water in Suisun Bay, being tolerant of salinities up to 4.5 ppt (eggs) or 6 ppt (young fish) (Ganssle, 1966; Burns, 1966b). Of 12,400 cyprinids collected in the Delta in 1963-1964, 84 percent were carp (Turner, 1966c). Most of the Delta carp migrate upriver to fresher water to breed (Herbold & Moyle, 1989). A large female may lay over 2,000,000 eggs per year. The largest carp reported from California weighed 26.3 kg (McGinnis, 1984).
Carp feed by "grubbing" in bottom sediments in shallow water, which digs up the bottom, destroys aquatic plants, and muddies the water, rendering potentially productive areas unsuitable for use as spawning or nursery areas by other fish species (McGinnis, 1984). Smith (1896, citing Jordan and Gilbert, 1894) reported that the carp's destruction of water celery Vallisneria might have reduced the population of canvasback and other ducks that feed on it. Shebley (1917) reported that carp "probably have been the principal cause of destruction of the California [Sacramento] perch, by eating the eggs and digging up the nests" (as Jordan & Gilbert (1894, cited in Smith, 1896) similarly reported from Clear Lake). Shebley believed that carp were the main food of black and striped bass, and that this outweighed the destruction of native perch. Burns (1966b) however, found carp to be of little forage value because they grow large too rapidly.
Smith (1896) reports that both muskellunge and sea lions were introduced into Lake Merced, San Francisco in order to eliminate carp. Shebley (1917) says of the introduction of carp to California that "at the time these plants were made the carp was one of the most popular of fishes; they were recommended as valuable food fish that would thrive in all of the warmer lakes, ponds and streams of California. Much has been said for and a great deal more against the introduction of carp into California...In time, as other species become more scarce, the carp will probably become one of the state's most valuable food fishes..." However by 1942 Curtis reported that carp "had become the most unpopular fish ever brought into California. It stands as Public Enemy No. 1 on the fisherman's books" for preying on the spawn of other fish, muddying the water and destroying plants. BDOC (1994) reported that considerable effort is expended on controlling carp in some waters and that their spread should be prevented.
Carp have supported small commercial fisheries in Clear Lake,
Lake Co. and in San Luis Reservoir, Merced Co. (McGinnis, 1984),
with statewide landings in the 1960s of about 300,000 pounds per
year valued at $15,000 (Davis, 1963; Burns, 1966b).
Dorosoma petenense (Günther, 1867) [CLUPEIDAE]
THREADFIN SHAD, MISSISSIPPI THREADFIN SHAD
SYNONYMS: Signalosa petenensis atchafaylae
Threadfin shad are native to the Gulf coast from Florida to Guatemala, north to Indiana and Illinois (Page & Burr, 1991). The California Department of Fish and Game planted 314 threadfin shad from Tennessee into four ponds in San Diego in 1953 (Kimsey, 1954). In 1954 and 1955, 1,020 of their progeny were planted in Lake Havasu on the Colorado River, and by the end of 1955 "appeared to be in every habitable part of the Colorado River from Davis Dam to the Mexican border, and in adjacent irrigation ditches, canals, settling basins and the Salton Sea" (Shapovalov et al., 1959). In 1959 threadfin shad were introduced into Central Valley reservoirs as a forage fish for largemouth bass, and spread downstream to the Delta by 1961 (Burns, 1966a; Turner, 1966d; Moyle, 1976b; McGinnis, 1984; Herbold et al., 1992)
Though mainly found in fresh water, threadfin shad are occasionally found in the sea off California and Oregon. They have been taken in Long Beach Harbor, San Francisco Bay, Drake's Estero and Humboldt Bay, and they grew well but did not spawn in the Salton Sea (Burns, 1966a; Miller & Lea, 1972; Eschmeyer & Herald, 1983). They are present in most lower and middle elevation freshwater habitats in California, including nearly all warm water reservoirs, and are abundant throughout the Delta (McGinnis, 1984; Herbold & Moyle 1989; Herbold et al., 1992). They have been caught at every Department of Fish and Game sampling station in the Delta, with few were taken in the western Delta (Turner, 1966d). They were the most abundant species of fish caught at stations east of Chipps Island in the Department of Fish and Game's Fall Midwater Trawl Survey for 1967-1988, and were usually found east of Sherman Island except during high outflow (Herbold et al., 1992).
Threadfin shad are most abundant in September and least abundant in January, so that heavy mortality must occur during the winter months. Young Corbicula, less then 1 mm in length, are common in stomachs in the spring (Turner, 1966d).
Burns (1966a) and McGinnis (1984) reported threadfin shad as an important forage fish for striped bass, but Moyle (1976) found them to be a "relatively minor component of striped bass diet." According to Turner (1966d), its "importance as a forage fish in the Delta may be limited because it is abundant only in restricted areas of quiet water." McConnell & Gerdes (1961) found that threadfin shad failed to provide adequate forage for largemouth bass and black crappie, possibly because of rapid growth by shad after a short spawning period, and that they may compete with the bass and crappie for cladocerans. Burns (1966a) reported threadfin shad as a major food of salmonids in lake Shasta and white catfish in Pine Flat Reservoir.
McGinnis (1984) suggested, based on its feeding habits and its
abundance in inshore zones, that threadfin shad compete for food
with the fry of striped bass and other game fish in the San Joaquin
River and in reservoirs. Turner argued that such competition was
limited, because in the summer and fall young striped bass are
in the western Delta eating Neomysis and Corophium
while threadfin shad are in the rest of the Delta eating copepods
and cladocerans. "Before the threadfin shad was introduced
into the Central Valley of California, Kimsey (1958) expressed
concern over the possibility that threadfin shad and small striped
bass would compete for food in the Delta. I do not believe that
competition between the two species is severe...Relatively few
young bass of this age inhabit the areas in the Delta where threadfin
shad have become abundant" (Turner, 1966d). Von Geldern &
Mitchil (1975, cited in Moyle, 1976b) reported that in many reservoirs
threadfin shad reduced the populations of many game fish, including
largemouth bass, through competition.
Gambusia affinis (Baird & Girard, 1853) [POECILIIDAE]
WESTERN MOSQUITOFISH
Mosquitofish are native to coastal drainages from New Jersey to Mexico, and to the Mississippi River basin north to Indiana and Illinois (Page & Burr, 1991). They were introduced to California in 1922 either from the southeastern United States (according to Moyle, 1976b) or from the southern Midwest (according to McGinnis, 1984) to control mosquitoes. They are now found in nearly every low and middle elevation fresh and brackish water habitat, and may be the most widely distributed and numerous freshwater fish species in the state (McGinnis, 1984). We (JTC) collected it in Lake Merritt in 1964-65, and it is today common in sloughs around the Bay and a common anadromous or resident fish in the Delta (Herbold & Moyle, 1989).
Mosquito fish are tolerant of what are normally considered unfavorable
water conditions, including high pesticide levels. Females produce
up to 300 live young per birth (McGinnis, 1984). Mosquitofish
compete with fry that occupy shallow shore edge environments,
and reportedly prey on California red-legged frogs (Anon., 1993).
They also eat adult pupfish (Cyprinodon sp.), and may have
contributed to the decline of a number of endemic pupfish in southern
California (Moyle, 1976b; McGinnis, 1984; BDOC, 1994).
Ictalurus furcatus (Lesueur, 1840) [ICTALURIDAE]
BLUE CATFISH
Blue catfish are native to coastal drainages from Alabama to Mexico, the Mississippi River basin north to southern South Dakota and western Pennsylvania, and the Rio Grande drainage (Page & Burr, 1991). In 1969, 1,758 blue catfish were flown from Stuttgart, Arkansas to San Diego County and planted in Lake Jennings on an "experimental basis" (Richardson et al., 1970), and later planted in a few other lakes in San Diego County (Taylor, 1980). Blue catfish were known to feed on the introduced clam Corbicula fluminea which was "abundant and a nuisance in many southern California waters but is virtually unutilized by present game fish," and, as the largest American catfish, they were expected to "enhance our fisheries by providing another trophy sized fish" (Richardson et al., 1970).
In 1978 a 4-pound blue catfish was caught in the San Joaquin River
near Mossdale, the possible source of the specimen being one of
18 fish breeders in the Central Valley licensed to raise blue
catfish (Taylor, 1980). Herbold & Moyle (1989) report that
blue catfish first appeared in the Delta in 1979, and that young-of-the-year
were found in Clifton Court Forebay in 1986, but that they remain
rare in the Delta.
Ictalurus punctatus (Rafinesque, 1818) [ICTALURIDAE]
CHANNEL CATFISH, SPOTTED CAT
Channel catfish originally ranged from the Gulf States and northern Mexico northward to Hudson Bay, the Great Lakes and Manitoba (Page & Burr, 1991). It is unclear just when the channel catfish was first introduced or became established in California. Shebley (1917) reports it introduced in 1874, and Smith (1896) reports that in that year Livingston Stone introduced some catfish, which could have been channel catfish, from Nebraska's Elkhorn River into the San Joaquin River near Stockton. Curtis (1949) states that this catfish was introduced to the Sacramento River system in 1891, but unnoticed for many years. Smith (1896) says that 250 yearlings each were planted in the Feather River (tributary to the Sacramento) and Lake Cuyamaca (in San Diego County) in 1891, and that 10 fish were planted in the Balsa Chico (Bolsa Chica?) River in 1895. Moyle (1976b) listed it as successfully introduced around 1925. Herbold & Moyle (1989) say that it became established only after several attempts to introduce it, and was first recorded from the Delta in the 1940s. Miller (1966b) reports that channel catfish were planted in the Colorado River at an unknown date and have been taken from there since 1920; and that the first authenticated capture in the Central Valley was in 1942.
In 1963-64 only 571 out of 26,000 catfish (2%) collected from the Delta were channel catfish, with most taken in swifter water in channels upstream from the central Delta (Turner, 1966a). They are now found in warm, low elevation rivers and lakes in California, but in some places will not spawn and must be maintained by hatchery stocking (McGinnis, 1984). They are common in the Delta, especially in the channels of the Sacramento River (Herbold & Moyle, 1989). BDOC (1994) noted that channel catfish can destroy the spawning sites of native fish by preying on eggs, larvae and juveniles.
Channel catfish live up to 39 years, and grow up to 1 meter in
length and 20 kg weight. A single female may lay up to 70,000
eggs. They are the only warm water food fish that is reared commercially
in the state, with farms in the Central Valley and elsewhere (McGinnis,
1984).
Lepomis cyanellus Rafinesque, 1819 [CENTRARCHIDAE]
GREEN SUNFISH
Green sunfish originally ranged on the Gulf coast from Florida to northern Mexico north to Ontario to Montana, and have been introduced to much of the United States (Page & Burr, 1991). In 1891 a few unidentified sunfish from Quincy, Illinois were accidentally introduced with other fish into Lake Cuyamaca near San Diego, and green sunfish were taken from that lake by 1895. Another 36 sunfish from Illinois, possibly including green sunfish, were planted in Elsinore Lake and the Balsa Chico (Bolsa Chica?) River in 1895 (Smith, 1895; Shebley, 1917; Curtis, 1949).
Today they are present in most low and middle elevation freshwater habitats in California, except in the Klamath River drainage, and are reported as common and widely distributed in the Delta (McKechnie & Tharratt, 1966; McGinnis, 1984; Herbold & Moyle, 1989). However, in 1963-64, only 15 of 11,750 centrarchids collected in the Delta (0.1%) were green sunfish (Turner, 1966b).
Green sunfish are tolerant of high temperatures, low oxygen and high alkalinity, and are territorially aggressive (McGinnis, 1984). They often hybridize with bluegill, producing sterile crosses (Curtis, 1949).
Predation by green sunfish nearly eliminated the California roach,
Hesperoleucus symmetricus, from the upper San Joaquin,
Fresno and Chowchilla rivers (Moyle, 1976b). Along with bluegills,
the green sunfish competes with another California endemic, the
Sacramento perch (Archoplites interruptus). In some areas
the introduced sunfish exclude the native perch from feeding sites,
and may have been contributed to the perch's extermination from
its native waters in the Delta (McGinnis, 1984). Predation by
green sunfish may have also contributed to declines in red-legged
and yellow-legged frogs (BDOC, 1994).
Lepomis gulosus (Cuvier, 1829) [CENTRARCHIDAE]
WARMOUTH
SYNONYMS: Chaenobryttus gulosus
Warmouth are native to coastal drainages from Virginia to Texas, the Mississippi River basin north to Pennsylvania, the Great Lakes and Montana, and the Rio Grande upstream to New Mexico (Page & Burr, 1991), and have been widely introduced elsewhere in the West (Hubbell, 1966). In 1891 the U. S. Fish Commission planted 400 yearling warmouth from the fish station in Quincy, Illinois into Lake Cuyamaca in San Diego County, and 100 yearlings into the Feather River near Gridley, in Butte County. In 1895 another 12 warmouth were delivered to the Sisson hatchery, but died before spawning (Smith, 1895; Shebley, 1917; Curtis, 1949). They were first recorded in the Delta after 1921 (Herbold & Moyle, 1989).
Warmouth are present in the Colorado River and present though rarely abundant in many parts of the Central Valley and Delta, usually in warm waters with little gradient, soft bottom, and abundant cover (Hubbell, 1966; McGinnis, 1984). In the Delta they are largely restricted to dead-end sloughs of the eastern Delta (Herbold & Moyle, 1989). Only 240 of 11,750 centrarchids collected in the Delta in 1963-64 (2%) were warmouth (Turner, 1966b).
Warmouth hybridize with bluegill, pumpkinseed and green sunfish.
They are of limited importance as a gamefish in California (Hubbell,
1966).
Lepomis macrochirus Rafinesque, 1819 [CENTRARCHIDAE]
BLUEGILL, BLUE BREAM
Bluegill are native to drainages from Virginia to northern Mexico, the Mississippi River basin north to Quebec, the Great Lakes and Montana, and the Rio Grande upstream to New Mexico (Page & Burr, 1991). They may have first been introduced to California along with green sunfish in 1891 (Smith, 1895; Shebley, 1917), but the first unequivocal reports date from 1908 when the U. S. Fish Commission shipped bluegill from Meredosia, Illinois to California (Curtis, 1949). These were planted in Honey Lake in Lassen County, various lakes in Placer County, Clear Lake in Lake County, Buena Vista Lake in Kern County, Russells Lake in Ventura County, and the Feather, Sacramento, San Joaquin, Kings and Kern rivers, including the San Joaquin River near Stockton (Vogelsand, 1931; Moyle, 1976b). Bluegill today are widely distributed in warm freshwater habitats and are the most abundant sunfish in California (McGinnis, 1984; Herbold & Moyle, 1989). They are common in the Delta, where they accounted for 26 percent of 11,750 centrarchids collected in 1963-64 (Turner, 1966b), and have been collected downstream in San Pablo Bay in the winter (Ganssle, 1966).
Bluegill have been known to spawn as yearlings, and females produce 2,000 to 50,000 eggs per spawning. In many areas, overpopulation has produced populations of stunted fish (Emig, 1966c; McGinnis, 1984).
The elimination of the Sacramento perch from its native range
in the Delta has sometimes been attributed to competition for
food and breeding sites by the more aggressive bluegill (Moyle,
1976b; McGinnis, 1984; BDOC, 1994), but competition from green
sunfish and predation by striped bass and largemouth bass have
also been cited as contributing factors. Bluegill eat bass eggs
(McGinnis, 1984), and may have contributed to declines in red-legged
and yellow-legged frogs (Anon., 1993; BDOC, 1994).
Lepomis microlophus (Günther, 1859) [CENTRARCHIDAE]
REDEAR SUNFISH
Redear sunfish are native to the southeastern United States, ranging from the Carolinas and Florida to Missouri and Texas, and north in the Mississippi River basin to southern Indiana and Illinois (Page & Burr, 1991). They were first introduced into California in 1948 or 1949 (Emig, 1966d; Moyle, 1976b). In 1954, 3,960 redear fingerlings from the federal hatchery in Dexter, New Mexico were planted in ponds in southern California, and in the fall of 1956 some of the southern California fish were sent to ponds in the San Joaquin Valley and the Central Valleys Hatchery. The progeny from these fish were then distributed to other water bodies in the state (Shapovalov et al., 1959). Herbold & Moyle (1989) report that redear sunfish were first introduced or captured in the Delta after 1949.
Today redear are present in warm, freshwater habitats of southern and central California (McGinnis, 1984), including a few streams in the San Joaquin River drainage (Brown & Moyle, 1993). They are uncommon in the Delta, where they are mainly found in the channels of the Sacramento River (Herbold & Moyle, 1989). None of the 11,750 centrarchids collected in the Delta in 1963-1964 were redear sunfish (Turner, 1966b).
The redear is a deep-water bottom feeder, and is less prolific
than the bluegill, producing only about 2,000 eggs per spawning
(McGinnis, 1984).
Lucania parva (Baird, 1855) [CYPRINODONTIDAE]
SYNONYMS: Cyprinodon parvus
Lucania venusta
Lucania affinis
see Hubbs & Miller (1965) for a detailed discussion of synonymy
RAINWATER KILLIFISH
The rainwater killifish is native to Atlantic coastal regions from Massachusetts to northeastern Mexico, and the Rio Grande drainage. It mainly inhabits protected salt and brackish waters, penetrating into fresher waters in the southern part of its range, and up the Rio Grande into the highly mineralized lower portion of the Pecos River in Texas and New Mexico. It was first collected west of this region in San Francisco Bay at Aquatic Park, Berkeley "not later than the spring of 1958," followed by collections at Richmond and in Corte Madera Creek in Marin County (1958), Lake Merritt, Oakland (1961) and Palo Alto Yacht Harbor (1962). It has also been introduced into Yaquina Bay, Oregon (first collected in 1958), Timpie Springs (1959) and Blue Lake (1961) in northwestern Utah, and Irvine Lake in southern California (1963) (Hubbs & Miller, 1965).
Hubbs & Miller (1965) provide evidence indicating that the killifish was probably introduced to Utah and southern California with shipments of gamefish (bluegill, largemouth bass, black crappie or bullhead) from fishery stations on the Pecos River. They suggest that it was transported to San Francisco and Yaquina bays as eggs in shipments of eastern oyster (which continued into the 1940s), or possibly in ballast water.
However, the nearly simultaneous discovery of this fish in five separate water bodies in the West suggests that a single transport mechanism was at work. Hubbs & Miller rejected the possibility of accidental transport with New Mexico gamefish planted in the San Francisco and Yaquina bay areas because they could find no records of such plantings. For example, they quote from a letter (Dec. 17, 1959) from Leo Shapovalov of the California Department of Fish and Game that he had "not been able to locate any definite information on shipments of fish into California from the U. S. Fish and Wildlife Service hatchery at Dexter, New Mexico, in relation to the appearance of Lucania in the San Francisco Bay area." However Shapovalov et al. (1959) reported that redear sunfish fingerlings from the Dexter hatchery were planted in southern California ponds in 1954, that the redear sunfish from these ponds were then planted in San Joaquin Valley ponds and brought to the Central Valleys Hatchery (in the San Francisco Bay watershed) in 1956, and that between 1956 and 1959 redear sunfish from this hatchery were planted into "a number of waters" in California. Given the apparent importance of the Dexter hatchery in the 1950s as a source of gamefish stock for western states, and the frequent shipments of gamefish to and between hatcheries, private ponds and public waters (with many of these transactions apparently never recorded), it seems likely that transport with gamefish was responsible for all five introductions of killifish.
Hubbs & Miller (1965) discuss morphometric and meristic evidence
to support their contention that the Utah and southern California
killifish populations originated from New Mexico while the San
Francisco Bay and Yaquina Bay populations originated from the
Atlantic coast, but the correlations they provide are weak at
best, and are as readily explained by ecophenotypic variation
(e. g. fish inhabiting interior waters versus fish inhabiting
tidal waters). We predict that molecular genetic analysis would
show all five introduced populations to be more closely related
to New Mexico than Atlantic coast stocks.
Menidia beryllina (Cope, 1866) [ATHERINIDAE]
INLAND SILVERSIDE, MISSISSIPPI SILVERSIDE
Synonyms: Menidia audens
The inland silverside is native to coastal drainages from Massachusetts to Texas, the Mississippi River and major tributaries to southern Illinois and eastern Oklahoma, and the Rio Grande in Texas and southeastern New Mexico (Page & Burr, 1991). In the fall of 1967, the California Department of Fish and Game and the Lake County Mosquito Abatement District planted about 9,000 young-of-the-year silver sides from Oklahoma into Upper and Lower Blue Lakes and Clear Lake in Lake County, California, to control gnats and midges and to reduce nuisance blooms of green algae, although the silverside's ability to control either gnats or algae had not been demonstrated (Moyle, 1976b). The stocking into Clear Lake was apparently also done without the permission of the California Fish and Game Commission or the "official endorsement" of the California Fish and Game (Cook & Moore, 1970; McGinnis, 1984). The silverside population exploded in Clear Lake, such that silversides were the most abundant species taken in seine hauls by the fall of 1968 (one year after the introduction of less than 3,000 fish), with up to 2,500 silversides in a single haul (Cook & Moore, 1970). Silversides became the dominant inshore fish in the lake and, according to McGinnis (1984), provided "the final competitive blow for the extinction of the native Clear Lake splittail."
Inland silversides from Clear Lake were introduced into three ponds in Santa Clara County in 1968 and two lakes in Alameda County in 1969 and 1970, and unauthorized transplants, possibly occurring when these fish were used as bait, were subsequently made to other water bodies in these counties (Moyle et al., 1974). Silversides were collected in the San Joaquin River near Manteca in 1971, and became the dominant inshore species there by 1976. By 1980 it was one of the most numerous fish in the Delta system. Its current distribution includes Clear Lake, Cache Creek, Putah Creeks, throughout the Delta downstream to Antioch, and in the tributary rivers and associated reservoirs of the San Joaquin Valley, and it continues to spread (Meinz & Mecum, 1977; McGinnis, 1984).
Inland silversides tolerate a wide range of water conditions, including high temperatures, low oxygen and moderate organic pollution. Females may spawn up to 15,000 eggs per year. Inland silversides feed on zooplankton and small, bottom-dwelling invertebrates in the inshore zone, and thus may not be very effective at gnat and midge control (McGinnis, 1984).
Inland silversides may compete with striped bass in the Delta.
McGinnis (1984) found that in the middle San Joaquin River Neomysis
mercedis is the preferred food of both inland silversides
and striped bass. Silversides may also be a significant predator
of the larvae and eggs of the endangered Delta smelt (BDOC, 1994;
Moyle, pers. comm.). Li et al. (1976) discuss data suggesting
that silversides compete with and caused a decline in the growth
rate of black and white crappie in Clear Lake.
Micropterus dolomieu Lacepéde, 1802 [CENTRARCHIDAE]
SMALLMOUTH BASS, SMALLMOUTH BLACK BASS
Synonyms: Micropterus dolomieui
The smallmouth bass is native to the Hudson Bay, Great Lakes and Mississippi River drainages from southern Quebec to North Dakota, south to northern Alabama and Oklahoma (Page & Burr, 1991). In 1874 Livingston Stone planted 73 full-grown smallmouth bass from Lake Champlain, Vermont, in Napa Creek, and 12 small bass from the Saint Joseph River, Michigan in Alameda Creek. Bass apparently reproduced in both creeks, but the Napa Creek population was fished out by 1878 while the Alameda Creek population grew large enough to stock other streams. Sometime before 1879, Seth Green imported a shipment of black bass, either smallmouth or largemouth, for the Sportsmen's Club of San Francisco and planted them in Lake Temescal in Oakland. In 1879 Livingston Stone planted another 22 full-grown smallmouth bass in Crystal Springs Reservoir in San Mateo County. These increased rapidly and their progeny were planted around the state, with much of the distribution during this period done by private parties and never recorded. In 1887 black bass were reported in the Russian River (apparently stocked by private parties) and by 1894 anglers were illegally harvesting bass from the river with seine hauls and dynamite. From 1889 to 1895 state authorities engaged in a major redistribution of black bass in the state, taking many of them from the San Andreas Reservoir in San Mateo County and the Russian River (where 9,350 were collected in 1894 and 25,600 fry in 1895) and planting them in waters from San Diego County to Butte County, including the American River and the San Joaquin River in Fresno County. At this time black bass were also reported from the Sacramento River at Colusa (Smith, 1895; Shebley, 1917).
Curtis (1949) reported smallmouth bass in Putah Creek and the Russian, Feather, American, Tulomne, Stanislaus, Merced, San Joaquin, Kings and Kern rivers, with 1,890,000 black bass (both smallmouth and largemouth) caught by anglers in 1948. Smallmouth bass are now present in many rivers and lower and mid-elevation lakes in California (McGinnis, 1984), though uncommon in the Delta where they are largely restricted to dead-end sloughs (Herbold & Moyle 1989). None of the 11,750 centrarchids collected in the Delta in 1963-64 were smallmouth bass (Turner, 1966b).
Brown & Moyle (1993) report that a decline in native hardhead
(Mylopharodon conocephalus) in streams of the San Joaquin
River drainage was associated with an expansion of smallmouth
bass.
Micropterus salmoides (Lacepéde, 1802) [CENTRARCHIDAE]
LARGEMOUTH BASS, LARGEMOUTH BLACK BASS
SYNONYMS: Huro salmoides
Largemouth bass are said to be "the most popular warm-water game fish in North America" (McGinnis, 1984). They are native to the Hudson Bay, Great Lakes and Mississippi River drainages from southern Quebec to Montana, south to Louisiana, and coastal drainages from North Carolina to northern Mexico (Page & Burr, 1991). Although a pre-1879 private stocking of "black bass" in Lake Temescal in Oakland may have involved either largemouth or smallmouth bass, and largemouth bass were planted in Washington state in 1890, the first unequivocal planting of largemouth bass into California occurred in 1891, when the U. S. Fish Commission planted 620 yearlings in the Feather River near Gridley and 2,000 yearlings in Lake Cuyamaca in San Diego County. In 1895 the California Fish Commission took delivery of 2,500 fry which they raised in the Sisson Hatchery and distributed the progeny throughout the state. As noted above under smallmouth bass, there was also considerable redistribution of black bass around the state at this time (Smith, 1895; Shebley, 1917).
Curtis (1949) reported largemouth bass to be common throughout the Sacramento-San Joaquin river system and in southern California, with 1,890,000 black bass (both smallmouth and largemouth) caught by anglers in 1948. Largemouth are reported as common in the Delta, especially in dead-end sloughs (Herbold & Moyle, 1989), although only 34 of 11,750 centrarchids collected in the Delta in 1963-64 (0.3%) were largemouth bass (Turner, 1966b).
In the Delta, predation by largemouth bass and striped bass may have been a key factor in the global extinction of the thicktail chub (Gila crassicauda) and in the elimination of the Sacramento perch (Archoplites interruptus) from its native range in the Delta (Moyle, pers. comm., 1993), though competition from introduced sunfish is also said to be a cause of the perch's decline (McGinnis, 1984). Predation by largemouth bass may also have contributed to the decline of native red-legged and yellow-legged frogs (BDOC, 1994). In eastern California, predation by largemouth bass was probably a major cause of the near extinction of the Owens pupfish, Cyprinodon radiosus (Moyle, 1976; Wilcove et al., 1992). Curtis (1942) reported that trout declines in some waters are caused by black bass. It is interesting to note that even as they made the initial plantings, fishery agents were aware of the bass' potential to reduce native fish populations. As Smith (1896) reported, "State fish commissioners have refrained from depositing fry or yearling bass in waters already stocked with salmon or trout, but have restricted the distribution to lakes, reservoirs, ponds, and rivers in which the predaceous bass could do no damage. It seems only a question of time, however, when the bass will naturally find their way into and become abundant in all those rivers in which they have not already been planted."
Largemouth bass have also been introduced to Europe and Africa
(Emig, 1966a).
Morone saxatilis (Walbaum, 1792) [PERCICTHYIDAE]
STRIPED BASS, STRIPER, ROCK BASS
SYNONYMS: Roccus saxatilis, Roccus lineatus
The striped bass is native to the Atlantic coast from the St. Lawrence River to northern Florida, and the Gulf coast from western Florida to Louisiana (Robins & Ray, 1986). In 1879 Livingston Stone planted about 135 fish (from a shipment that started as 132 fish, 1.5 to 5 inches long, plus 30 medium-sized fish) from the Navesink River, New Jersey in Carquinez Strait at Martinez. In 1882, a little over 300 fish (from a shipment that started as 450 fish, 5 to 9 inches long) from the Shrewsbury River, New Jersey were planted in Carquinez Strait at Army Point, Benicia. By 1889, hundreds were being sold in the San Francisco markets (Shebley, 1917). Several workers have theorized that conditions in the late 1800s "probably favored striped bass and American shad reproduction, because their semi-buoyant eggs would not be smothered by silt from gold mining operations" (Herbold et al., 1992), unlike the eggs of many native fish that are laid in the bottom gravel or attached to submerged vegetation or other substrate.
Striped bass are present today in the Sacramento-San Joaquin river system, in San Antonio Reservoir, in Lake Mendocino and in the lower Colorado River (McGinnis, 1984). Unsuccessful attempts were also made to establish striped bass in the Salton Sea (Roedel, 1953). Land-locked populations exist in Millerton Reservoir in Fresno County (a self-sustaining population) and San Luis Reservoir (restocked continuously by means of water imported from the Delta, which entrains young bass). Striped bass were propagated in hatcheries by the California Department of Fish and Game and annually released to the Delta from 1982 to 1992, when stocking was curtailed due to concern over predation on the endangered winter-run chinook salmon (BDOC, 1994). An estimated 80 million fry were entrained by State Water Project pumps each year, and 165 million fry a year by the cooling water intakes for the PG&E power plants in Antioch and Pittsburg. The striped bass population dropped from an estimated 4 million fish in 1960, to 2 million in 1970, to 1 million in 1980 (McGinnis, 1984). Herbold et al. (1992) reported the population in the Estuary at 1,480,000 to 1,880,000 prior to 1976, and 520,000 to 1,160,000 after 1977.
Striped bass were the most common fish collected in trawls of Suisun Marsh sloughs in 1979-86 (Brown, 1987). They were reported as abundant in the Delta (Herbold & Moyle, 1989), and common to abundant in San Francisco Bay (Emmett at al., 1991). Striped bass were also reported as common in Tomales Bay, and in Coos Bay, the Umpqua River and the Siuslaw River in Oregon. They have been reported north to British Columbia and south into Mexico, but populations in the southern bays are not self-sustaining (Emmett at al., 1991). Striped bass from the San Francisco Bay watershed have been captured from central Oregon to southern California, but most travel no further than 40 km from the Golden Gate (Herbold et al., 1992).
Mean fecundity for striped bass has been reported at 243,000 eggs (for 4-year-olds) to 1,427,000 eggs (for 8-year-olds and older). A 5-pound fish spawns up to 25,000 eggs, a 12-pound fish up to 1,250,000 eggs, and a 75-pound fish up to 10,000,000 eggs (CDFG 1987; Emmett at al., 1991). Herbold et al. (1992) reported that "females commonly broadcast from 500,000 to 4.5 million eggs (Hassler 1988), although estimates range from 11,000 (Moyle 1976) to a high of 5.3 million (Hollis 1967; Hardy 1978; Wang 1986)."
Striped bass eggs are found from fresh water to salinities of 11 ppt (with optimal salinities between 1.5 and 3.0 ppt) and tolerate temperatures of 12-24°C (with an optimum of 18°C). Larvae occur in both freshwater and oligohaline water. Juveniles and adults are found in all parts of the estuary. Most males mature in their 2nd or 3rd year, females in their 4th or 5th year. Maximum reported age is over 30 years.
Striped bass fry are pelagic carnivores feeding on small invertebrates. Juveniles and adults are epibenthic and pelagic carnivores, the juveniles feeding on the young of small fish and larger invertebrates, while the adults are primarily piscivorous (McGinnis, 1984; Emmett at al., 1991).
The commercial catch in 1899, 2 decades after introduction, was 560 tons and usually exceeded 450 tons up to 1915. Commercial fishing in the Estuary was banned in 1935 to avoid competition with the sport fishery. Although there is no longer a commercial fishery, "each year thousands of kilograms of illegal striped bass are believed to make their way to restaurants and fish markets in the greater San Francisco Bay area. Some of these come from massive nighttime netting operations in the lower Delta area. Small time operators, however, simply use standard sport fishing techniques to catch far more than the legal limit and then proceed directly to some local buyer" (McGinnis, 1984).
Striped bass is the principal sport fish caught in San Francisco Bay, and the economically most important fish in the Delta. The sport catch ranged from 107,000 to 403,000 fish in 1975-78 (Emmett at al., 1991). In 1980 California anglers took about 1 million bass, spending about $7 million in the process (McGinnis, 1984). "The subsidiary industries surrounding striped bass fishing (boats, marinas, and paraphernalia) are estimated to bring $45 million into the local economies" (Herbold et al., 1992).
Striped bass were the most numerous predator at three sampled
locations in the Delta (Pickard et al., 1982). Moyle has suggested
that striped bass and largemouth bass preyed on and contributed
to the global extinction of thicktail chub (Gila crassicauda),
and the elimination of Sacramento perch (Archoplites interruptus)
from its native waters in the Delta (Moyle, pers. comm., 1993),
though competition with introduced sunfish has also been raised
as a factor in the decline of the perch (McGinnis, 1984). Striped
bass have been reported as a major predator of salmon fingerlings
in the Delta (USBR, 1983), though chinook salmon formed only a
minor component of the stomach contents of subadult and adult
striped bass collected in the Delta in 1963-64 (Stevens, 1966).
BDOC (1994) noted that few young salmon are eaten by striped bass
in the Estuary (except at salmon stocking sites and Clifton Court
Forebay), but sometimes form a substantial part of the diet of
striped bass upstream in the Sacramento River, and concluded that
striped bass predation reduces salmon abundance by an unquantified
amount.
Notemigonus crysoleucas (Mitchill, 1814) [CYPRINIDAE]
GOLDEN SHINER
The golden shiner is native to coastal drainages from Nova Scotia to Texas, and the Hudson Bay, Great Lakes and Mississippi River drainages west to Alberta and Oklahoma, and "widely introduced (via bait buckets) elsewhere in U. S." (Page & Burr, 1991). It was imported into southern California in 1891, and was widespread in the Sacramento-San Joaquin River system by 1964 (Kimsey & Fisk, 1964), probably distributed as bait releases by anglers (Herbold & Moyle 1989). In 1963-64, 212 of 12,400 cyprinids (2%) collected in the Delta were golden shiner, mainly taken in dead-end sloughs (Turner, 1966c). They are reported as widely established in California (Moyle, 1976b; McGinnis, 1984) and common in the Delta (Herbold & Moyle, 1989).
The golden shiner is one of three legal freshwater bait fishes in California (the others, also nonnative fish, are red shiner and fathead minnow), supporting a "rather lucrative small industry" of bait fish propagation and leading to its wide distribution in the state. It is a popular bait for striped bass (McGinnis, 1984).
Golden shiner reportedly compete with both native cyprinids and
the fry of some gamefish (McKechnie, 1966b; McGinnis, 1984). Trout
production in some lakes has been reduced by competition between
trout parr and golden shiner (McGinnis, 1984). Pat O'Brien of
the California Department of Fish and Game reports that 2 to 3
high elevation lakes in California are taken over each year by
illegally planted brown bullhead and golden shiner.
Percina macrolepida Stevenson, 1971 [PERCIDAE]
BIGSCALE LOGPERCH
SYNONYMS: Percina caprodes
The native range of the bigscale logperch runs from the Sabine
River in Louisiana to the Red River in Oklahoma, the Rio Grande
drainage in Texas and New Mexico, and Mexico (Page & Burr,
1991). It was accidentally introduced from Texas in 1953 in an
airplane shipment of largemouth bass and bluegill that was planted
in Miller, Blackwelder and Polk lakes at Beale Air Force Base,
Yuba County, by the U. S. Fish and Wildlife Service. The lakes
are in the Yuba River drainage, a tributary of the Sacramento,
and regularly overflow (Shapovalov et al., 1959; Moyle, 1976b;
McGinnis, 1984). By 1972-73 the logperch was established in the
lower Sacramento River and the Delta (Moyle et al., 1974), and
are now widespread throughout the Sacramento-San Joaquin river
system (Moyle, 1976b; McGinnis, 1984) and common in the Delta
(Herbold & Moyle, 1989). They are also abundant in Lake Del
Valle in Alameda County, probably pumped in from the Delta via
the State Water Project pumps and the South Bay Aqueduct (Moyle
et al., 1974).
Pimephales promelas Rafinesque, 1820 [CYPRINIDAE]
FATHEAD MINNOW
The native range of the fathead minnow runs from Quebec to the Northwest Territories and south to Alabama, Texas and New Mexico (Page & Burr, 1991). The first record of it in California is from a bait tank near the Colorado River in 1950. In 1953, 40,000 were imported by a fish breeder in Turlock. The California Department of Fish and Game purchased 1,000 of these fish, spawned them at the Central Valleys Hatchery, and planted the progeny in various water bodies as forage fish (Shapovalov et al., 1959). The fathead minnow is one of California's three legal freshwater bait fish, and it has been further spread through the state as bait releases by anglers (McGinnis, 1984; Herbold & Moyle, 1989). Herbold & Moyle (1989) report it first appearing in the Delta in the 1950s, where it is now occasionally collected and common only in localized patches, generally in small creeks.
The fathead minnow is tolerant of high temperatures, low oxygen
and organic pollution (McGinnis, 1984). It has the potential to
compete with the ecologically-similar native, the California roach
Hesperoleucus symmetricus, whose distinct forms may actually
be separate species (Moyle, 1976b). McGinnis (1984) warned that
its "ability to establish populations readily in pools of
intermittent streams and backwater areas in California poses a
serious threat to several native cyprinids adapted to such habitats."
Pomoxis annularis Rafinesque, 1818 [CENTRARCHIDAE]
WHITE CRAPPIE
Pomoxis nigromaculatus (Lesueur, 1829) [CENTRARCHIDAE]
SYNONYMS: Pomoxis sparoides
BLACK CRAPPIE, CALICO BASS, STRAWBERRY BASS
The black crappie is native to the eastern United States from Virginia to Texas and north through the Mississippi River basin to the Great Lakes. The white crappie's native range runs from the Gulf coast between Alabama and Texas north through the Mississippi River basin to the Great Lakes and Hudson Bay (Goodson, 1966a; Page & Burr, 1991). The history of the introduction and spread of these fish in California is uncertain because there were numerous attempted introductions, both successful and unsuccessful, and because some authors failed to distinguish (or confused) the two fish.
The first recorded introduction of these fish on the Pacific coast was near Seattle, Washington in 1890. In 1891, 285 yearling black and white crappie from the U. S. Fish Commission station at Quincy, Illinois were planted in Lake Cuyamaca near San Diego. Vogelsang (1931) and Goodson (1966a) state that this introduction was unsuccessful. In 1895 a second shipment, of 50,000 fry, was sent to the Sisson Hatchery, but none survived (Smith, 1895; Shebley, 1917; Curtis, 1949). Goodson (1966a) states that another unsuccessful attempt was made in 1901 (citing Vogelsang (1931) who, however, makes no reference to a 1901 attempt). In 1908, crappie from the Illinois station were planted in Honey Lake in Lassen County, Vera Lake in Nevada County, Clear Lake in Lake County, in sloughs and tributaries of the Feather, Sacramento, San Joaquin, Kings and Kern rivers (including the San Joaquin River near Stockton in the Delta), and possibly at other sites in southern California (Shebley, 1917; Vogelsang, 1931; Goodson, 1966a). Of this effort, Vogelsang (1931) implies that both species of crappie were introduced (Vogelsang introduces his paper as an account of "the first successful introduction of the crappie, calico bass [=respectively, the white crappie and the black crappie; Smith (1896) and Shebley (1917) use the same nomenclature], blue gill and green sunfishes and the yellow perch" into California, although in the rest of the paper he only refers to "crappie"), Shebley (1917) states only that the white crappie was introduced, and Goodson (1966a) argues that probably only the black crappie was introduced, since white crappie were not reported north of the Tehachapi Mountains until 1951.
Goodson (1966a) reports the introduction of 16 crappie from an unknown source into a pond in San Diego County in 1917, and the subsequent stocking of nine San Diego County reservoirs from that pond. Since only white crappie have since been reported from these reservoirs, he argues that the original plant of 16 fish were all white crappie, and that all white crappie in California are descended from those 16 fish. Curtis (1949) reported the white crappie surviving only in the San Diego area and the Colorado River drainage, and the black crappie widespread in the state. Nearly 3 million crappie were caught in the state in 1948, mainly in southern California. In 1951 white crappie from one of the San Diego reservoirs were planted in a reservoir in Colusa County, and subsequent plants were made in other California waters (Goodson, 1966a).
Moyle (1976b), more-or-less consistent with Goodson, lists the black crappie as introduced in 1908 (citing Vogelsang, 1931) and the white crappie as introduced, from Illinois, in 1917 (citing Curtis, 1949, who, however, describes both species as introduced in 1891). Herbold & Moyle (1989) list the "year of introduction or first capture" in the Delta as 1908 for the black crappie and 1951 for the white crappie. We relied on Moyle's dates for our analysis.
Black crappie are today present in low and middle elevation reservoirs
and slow streams (McGinnis, 1984). They are common in the Delta,
accounting for 71% of the 11,750 centrarchids collected in the
Delta in 1963-1964 (Turner, 1966b), and have on occasion been
collected downstream to Martinez (Gannsle, 1966). McGinnis (1984)
reported the white crappie's distribution as throughout southern
California and in Clear Lake. It is apparently uncommon in the
Delta, with only one white crappie out of 11,750 centrarchids
collected there in 1963-1964 (Turner, 1966b). A large crappie
can produce more than 200,000 eggs per spawning (McGinnis, 1984).
In a study of their feeding habits in the Delta, black crappie
mainly ate threadfin shad and striped bass, along with small numbers
of chinook salmon, Delta smelt and other fish (Turner, 1966b).
Curtis (1949) reported that crappie compete with bass for food.
Tridentiger bifasciatus Steindachner [GOBIIDAE]
SHIMOFURI GOBY
It was discovered in 1994 that the introduced gobies in California called chameleon gobies consisted of two different species. The shimofuri goby, native to Japan and China, is adapted to fresher water than the chameleon goby and was first recorded in 1985 from Suisun Bay, having probably arrived in ballast water. By 1989 it was the most abundant fish in Suisun Bay, and by 1990 the most abundant larval fish in the upper Estuary. By 1990 it had also been transported 513 km south via the California Aqueduct to Pyramid Reservoir, and thence into Piru Creek by 1992 (Matern & Fleming, in prep.).
Experiments indicate that if the shimofuri goby disperses to coastal
waters harboring the endangered tidewater goby Eucyclogobius
newberryi, it could have a substantial impact by preying on
juvenile tidewater gobies, competing for food, and disturbing
mating activities (Swenson & Matern, 1995).
Tridentiger trigonocephalus (Gill, 1859) [GOBIIDAE]
CHAMELEON GOBY, TRIDENT GOBY, SHIMAHAZE
The chameleon goby is native to marine and brackish waters of Japan, China and Siberia (Eschmeyer et al., 1983). One specimen (70.4 mm standard length) was collected from Los Angeles Harbor in June 1960, with others were collected there in 1977 (Haaker, 1979). It was collected from the Redwood City docks in southern San Francisco Bay in 1962 (Matern & Fleming, in prep.)
Various workers have suggested that the goby could have been transported across the Pacific in ballast water, in ships' seawater systems, as eggs laid on fouling organisms on ships' hulls, or (for transport to San Francisco Bay) as eggs laid on imported Japanese oysters (Hubbs & Miller, 1965; Haaker, 1979). However, except for occasional experimental plants, Japanese oysters have not been planted in San Francisco Bay since the 1930s, and have never been planted in Los Angeles Harbor (Carlton, 1979a)
The chameleon goby has also become established in Sydney Harbor,
Australia (Haaker, 1979).
Rana catesbeiana
AMERICAN BULLFROG
The bullfrog is native to North America east of Colorado and New Mexico, and has become established in most western states, Hawaii, Mexico, Cuba, Japan and Italy (Stebbins, 1966). The bullfrog appears to have been independently introduced to California several times between 1910 and 1920. Bullfrogs were reported, but not confirmed, from Little Lake, Inyo County in 1918, and from ponds on the Stanford University campus in 1920. In July, 1922, adult and tadpole bullfrogs were collected from Sonoma Creek near El Verano, Sonoma County. These frogs were believed to be the descendants of 132 frogs purchased from New Orleans and 12 frogs purchased from a San Francisco frog merchant in 1914 and 1915 and planted in a nearby reservoir. Bullfrogs were also collected from Mockingbird Lake, Riverside County in 1922 and then from other lakes and streams in the area, possibly derived from a stock of Illinois and Louisiana bullfrogs kept by the physiology instructor at the Loma Linda College of Medical Evangelists since at least 1914 (Storer, 1922; George, 1927). Moyle (1979) reports that in 1929 bullfrogs were collected from the Kings River and planted in the San Joaquin River near Friant, and were introduced tno pons at the San Joaquin Experimental Range in Madera County in 1934.
The bullfrog was well established in the San Joaquin Valley by
1930, and is now common in many parts of California, including
the Delta (Moyle, 1973; Herbold & Moyle, 1989). Although several
authors have reported that reductions in populations of the California
red-legged frog Rana aurora, and possibly of the foothill
yellow-legged frog Rana boylii, may be due to predation
by or competition from bullfrogs (Moyle, 1973; Herbold & Moyle,
1989; Anon., 1993; BDOC, 1994), other factors (including overharvesting
of red-legged frog prior to the introduction of bullfrog, habitat
changes, and predation by introduced fish) make it difficult to
assess the bullfrog's true impact (Harvey et al., 1992).
Pseudemys scripta
POND SLIDER, RED-EARED SLIDER
Pond sliders are native to the eastern United States south to
Panama (Stebbins, 1966). They were presumably introduced to California
as released or escaped pets and are common in the Delta and elsewhere
in California (Herbold & Moyle, 1989; Harvey et al., 1992,
p. 180). The frequency with which they are encountered, our (ANC)
observations of a female laying eggs and of live, hatched young
in a nest at San Pablo Reservoir in Alameda County in July 1994,
and reports of reproducing populations at sites surrounding the
Estuary (in Putah Creek in Solano County, Walnut Creek and Jewel
Lake in Contra Costa County, Boronda Lake in Santa Clara County
and Stow lake in San Francisco County; Harvey et al., 1992), suggest
that they are almost certainly established in the Delta as well.
Although reportedly banned in the early 1970s (Harvey et al.),
we (ANC) have recently seen live sliders for sale in Asian markets
in San Francisco.
Ondatra zibethicus
MUSKRAT
The muskrat, native to the eastern United States, is common in the Delta and other parts of California in riparian woodland, freshwater and brackish marsh, and aquatic habitats (Josselyn, 1983; Herbold & Moyle, 1989, Harvey et al., 1992). Muskrat can damage banks and levees with their burrowing.
Skinner (1962, p. 161) reported that over the previous twenty
years muskrat had "risen to the status of the most important
fur bearer in the state, in terms of number of animals and total
value of the raw furs...Originally introduced into the northeastern
counties, they have moved down the Sacramento and into the San
Joaquin system since 1943." He reports trap data for the
state beginning in 1921-22, and for the San Francisco Bay Area
starting in 1939-40, with the number trapped annually in the Bay
Area rising from less than 100 until 1950 to between 6,000 and
9,500 in 1951-56. Herbold & Moyle (1989, citing a 1962 report)
reported about 11,000 trapped annually in the Delta.
Numerous species of marine plants and animals occur in the San Francisco Estuary whose status as introduced or native organisms remains unknown. These taxa are known as cryptogenic species (Carlton, 1995). We list here examples of 123 such taxa (Table 2). Many additional unidentified or taxonomically unresolved marine protists and smaller invertebrates exist in the Bay's estuarine margins as well and are not treated here. These include, in particular, roundworms (nematodes), flatworms (turbellarians), rotifers, harpacticoid copepods, and many species of planktonic and benthic ciliate protozoans. These unidentified taxa (representing at least an additional 25 distinct morphological entities), including members of groups also commonly occurring on oyster shells and in ballast water, are often found abundantly amidst communities dominated by species recognized as introduced. Most of the species listed in Table 2 represent one or more of the following categories:
1) Species frequently reported from fouling communities or planktonic assemblages in many cool- to warm-temperate harbors and ports around the world and which represent taxa easily transported with oysters, in ship fouling, in solid ship ballast, in ballast water, or by other means.
2) Species whose estuarine populations may represent a different species from populations occurring on outer, high-energy, full marine coasts that bear the same name.
3) Species believed to have appeared relatively recently in the Estuary.
4) Species symbiotic with known introduced species.
The taxonomy and distribution of the taxa listed as cryptogenic usually remain sufficiently unresolved as to prevent a clear resolution of their endemic versus exotic status without further data. In some cases, a species name is available; in other cases, only generic assignments are possible but enough evidence is at hand to question whether the taxon can automatically be considered native. In a number of cases (e. g. diatoms and other phytoplankters; hydroids) we have chosen examples of genera within which one or more (and sometimes many) species have been reported from the Estuary that represent cosmopolitan taxa potentially transported by human dispersal vectors and whose aboriginal history in the Eastern Pacific has not yet been worked out.
It is worth noting that cosmopolitan species represent one of three biogeographic categories: (1) a single species with truly broad and/or disjunct distributions achieved by natural means, (2) a single species spread by human-mediated transport, or (3) multiple species described as a single species. Combinations of these categories may complicate this trichotomy. Thus, one or more species may be spread globally by a mixture of natural and human-mediated mechanisms, creating a complex intermingling of pure and hybrid populations which are then described as a single cosmopolitan species.
The importance of recognizing cryptogenic species in elucidating potentially profound changes to the environment is discussed in Chapter 6. As noted there, no introduced diatoms, dinoflagellates, or other phytoplankters (such as chlorophyceaens, chrysophyceaens, cryptophyceaens, or cyanophyceaens) have been recognized from the Bay, despite a reported flora that includes many cosmopolitan taxa.
Prominent cryptogenic guilds in the Bay include phytoplankton
(25 percent), annelid worms (19 percent), protozoans (15 percent),
and cnidarians and crustaceans (about 10 percent each).
Table 2. Cryptogenic Species in the San Francisco
Estuary
Names of genera listed without species indicate at
least one cryptogenic species. Names of genera followed by "spp."
indicate at least two cryptogenic species.
[+] indicates San Francisco Bay populations, distinguished
from open coast populations bearing the same name
MICROALGAE
Bacillariophyceae (Diatoms)
Achnanthes
Asterionella
Aulacoseira (= Melosira) spp. (including A. distans var. lirata and A. granulata)
Biddulphia spp.
Chaetoceros spp.
Coscinodiscus spp.
Cyclotella spp. (including C. caspia)
Navicula spp.
Nitzschia
Pleurosigma
Rhizosolenia
Skeletonema (including S. costatum [+])
Thalassiosira (including T. decipiens)
Thalassiothrix
Dinophyceae (Dinoflagellates)
Dinophysis
Gonyaulax spp.
Gymnodinium
Protoperidinium spp.
Chlorophyceae
Monoraphidium
Scenedesmus
Cryptophyceae (Microflagellates)
Chroomonas minuta
Cryptomonas
Cyanophyceae (Blue-Green Algae)
Anabaena
Oscillatoria
Table 2. Cryptogenic Species - continued
MACROALGAE (Seaweeds)
Chlorophyta (Green Algae)
Cladophora
Enteromorpha "intestinalis" [+]
Enteromorpha spp.
Ulothrix
Ulva "lactuca" [+]
Rhodophyta (Red Algae)
Gigartina sp.
Gracilaria verrucosa
Grateloupia doryphora
VASCULAR PLANTS
Dicotyledones
Myriophyllum sibiricum
Polygonum amphibium
PROTOZOANS (examples only)
Epizoic or endozoic ciliates
Acineta sp. (on the introduced gribble isopod Limnoria)
Ancistrumina kofoidi (in the introduced clam Petricolaria)
Ciliate A (in the introduced shipworm Teredo navalis)
Ciliate B (in the introduced shipworm Teredo navalis)
Ciliate S1 (on the introduced isopod Sphaeroma quoyanum)
Ciliate S2 (on the introduced isopod Sphaeroma quoyanum)
Cochliophilus depressus (in the introduced snail Ovatella)
Cochliophilus minor (in the introduced snail Ovatella)
Epistylis sp. (on the introduced gribble isopod Limnoria)
Opercularia sp. (on the introduced gribble isopod Limnoria)
Vorticella spp. (on the introduced gribble isopod Limnoria)
Fouling ciliates
Suctorian sp. A
Vorticella sp.
Zoothamnium spp.
Free-living Benthic/Fouling ciliates
Spirorhynchus verrucosus
Planktonic holotrich ciliates
Mesodinium rubrum
Foraminifera
Ammobaculites exiguus
Milammina fusca
Table 2. Cryptogenic Species - continued
INVERTEBRATES
Porifera
Scypha sp.
Rotifera
Synchaeta bicornis
Cnidaria
Hydrozoa (examples only)
Bougainvillia ramosa
Campanularia
Clytia
Cryptolaria pulchella
Gonothyraea
Plumularia
Sarsia spp.
Sertularella
Sertularia
Syncoryne eximia
Anthozoa
Nematostella vectensis
Metridium senile [+]
Platyhelminthes
Trematoda
Austrobilharzia variglandis
Turbellaria
Childia groenlandica
Nemertea
Lineus ruber
Annelida
Oligochaeta
Aulodrilus limnobius
Bothrioneurum vejdovskyanum
Limnodrilus hoffmeisteri
Limnodrilus udekemianus
Polychaeta
Capitella spp.
Cirratulidae, unidentified species ("Tharyx parvus" of Bay authors)
Ctenodrilus "serratus"
Eteone californica/Eteone longa complex [+]
Euchone limnicola
Exogone "lourei"
Fabricia sp.
Glycera dibranchiata [+]
Glycinde sp.
Harmothoe imbricata [+]
Nereis virens [+]
Table 2. Cryptogenic Species - continued
Polychaeta - continued
Ophryotrocha puerilis
Polydora socialis
Prionospio pinnata [+]
Pygospio elegans [+]
Spiophanes "bombyx" [+]
Spirorbidae, unidentified species
Typosyllis sp.
Arthropoda: Crustacea
Copepoda
Eurytemora affinis
Notodelphyoid species (commensal in the introduced seasquirt Molgula)
Cumacea
Cumella vulgaris [+], in part: estuarine populations
Tanaidacea
Leptochelia dubia
Amphipoda
Caprella "equilibra" [+]
Caprella "penantis" [+]
Grandifoxus grandis ( = Paraphoxus milleri of San Francisco Bay authors)
Hyale sp.
Ischyroceridae, unidentified species
Listriella sp.
Photis sp.
Synchelidium sp.
Arthropoda: Insecta
Prokelisia marginata (on
the introduced cordgrass Spartina alterniflora)
Bryozoa
Alcyonidium parasiticum
Aspidelectra sp. (?)
Conopeum reticulum
Electra crustulenta [+], in part: estuarine populations
Membranipora sp. (?)
Smittoidea sp.
Chordata: Tunicata
Botryllus "tuberatus" [+]
Didemnum sp.
In all, we documented 212 species of introduced organisms in the Estuary. The numbers of species per taxonomic group are presented in Figures 2 and 3 at lower and higher levels of aggregation. Invertebrates are the most common major group of introduced species, accounting for nearly 70% of the total, followed by vertebrates and plants with respectively about 15 and 12 percent of the total. The most abundant invertebrates were the arthropods (36% of invertebrates) followed by molluscs (20%), annelids (14%) and cnidarians (12%). Nearly all the vertebrates were fish, and most of the plants were vascular plants, which were about evenly split between monocots and dicots.
These numbers are generally in accord with our expectations prior
to this study, based upon our knowledge of the Estuary's biota
and consideration of other regional reviews of introduced marine
and aquatic species, with the exception of the number of species
of vascular plants, which we had anticipated would be higher.
This result is in part due to our application of relatively more
restrictive criteria for the inclusion of marsh-edge plants, as
discussed in Chapter 2.

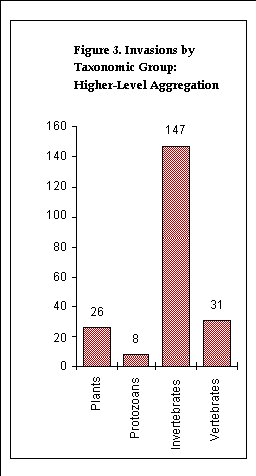
For example, a study of introduced species in the Great Lakes
using less restrictive criteria produced a list of 139 introduced
species of which 59 species (42%)were vascular plants (Mills et
al., 1993), and a similar study of the Hudson River produced a
list of 154 introduced species with 97 (63%) vascular plants (Mills
et al., 1995). As suggested in the "Methods" section,
adding the plants in Appendix 1 (essentially terrestrial plants
that have been reported in or at the edge of the tidal waters
of the Estuary) to the list of organisms in Table 1 produces a
list of introduced species that can more reasonably be compared
to the Great Lakes and Hudson River lists. This expanded list
for the Estuary contains 240 introduced species of which 49 (20%)
are vascular plants. These three and one other study are compared
in Appendix 5.
The numbers of species per native region are presented in Figure 4. Species were treated as either marine or continental species, as shown in Table 3, for assignment to appropriate regions. No introduced species were identified from the marine regions of the Eastern South Atlantic, the Western South Atlantic or the Eastern North Pacific, or from the continental region of Australia/New Zealand, so these regions do not appear in Figure 4.
The Estuary's marine introductions are dominated by species from
the Western North Atlantic (accounting for 41% of all marine introductions),
the Western North Pacific (33%) and the Eastern North Atlantic
(15%). The Western North Atlantic provided mainly mollusks, arthropods
and annelids, the Western North Pacific predominantly arthropods,
followed by annelids, and the Eastern North Atlantic provided
a few species from each of several groups. The Estuary's continental
introductions are dominated by species from North America (54%
of continental introductions; mainly fish) and Eurasia (29%, mainly
plants).
Table 3. Treatment of Introduced Species as Marine
or Continental, for Analysis by Native Region
PLANTS
Seaweeds marine
Vascular Plants
Spartina spp. marine
all other vascular plants continental
PROTOZOANS marine
INVERTEBRATES
Annelida
Oligochaeta
Branchiura sowerbyi continental
Limnodrilus monothecus marine
Paranais frici marine
Potamothrix bavaricus continental
Tubificoides spp. marine
Varichaetadrilus angustipenis continental
Polychaeta
Manayunkia speciosa continental
all other polychaetes marine
Mollusca
Cipangopaludina chinensis malleata continental
Melanoides tuberculata continental
Corbicula fluminea continental
all other molluscs marine
Arthropoda: Crustacea
crayfish continental
all other crustaceans marine
Arthropoda: Insecta
Anisolabis maritima marine
Neochetina spp. continental
Trigonotylus uhleri marine
Entoprocta
Barentsia benedeni marine
Urnatella gracilis continental
all other invertebrates
marine
VERTEBRATES
Fish
gobies marine
Alosa sapidissima marine
Morone saxatilis marine
all other fish continental
all other vertebrates
continental
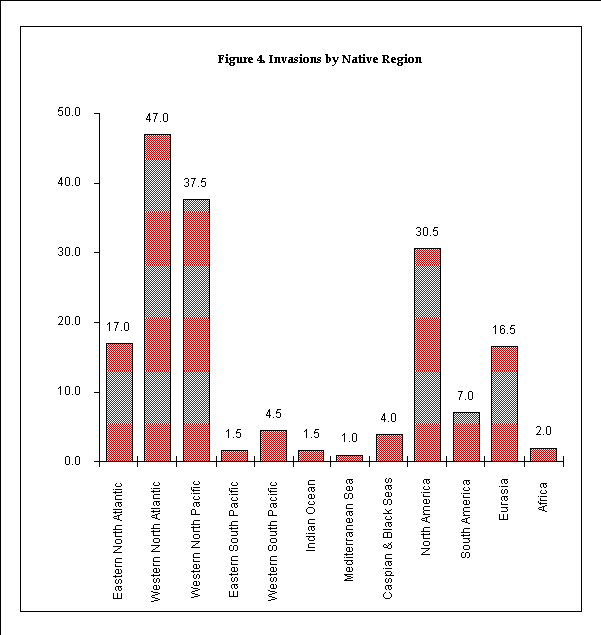
Analyses of the timing of introductions, done with the intent to distinguish pulses or patterns of invasions, are fraught with difficulties. In the San Francisco Estuary, as everywhere, larger and more conspicuous species (such as certain crabs, fish, and mollusks) tend to be noticed relatively soon after their arrival, while smaller and more cryptic organisms may be present but remain unnoticed for scores of years until the arrival of an appropriately specialized biologist. For example, the Bay's mud-dwelling worms received little attention until Olga Hartman began sampling in the Bay in the 1930s, and thus some of the polychaetes derived from the Atlantic might well have been introduced (with Atlantic oysters) as early as the 1870s. The biases introduced by taxonomist-dependent records of arrival are not limited to the earlier part of this century. With enough effort from appropriate taxonomic experts, many species of tiny introduced organismsósuch as protozoans, nematodes, flatworms and so forthócould certainly be collected today and identified from San Francisco Bay for the first time, although they may have been in the Estuary for 100 or more years.
Given these challenges, we have, as noted in Chapter 2, excluded from our tabulations of the temporal patterns of introductions both those species whose only available dates of first record are the first written accounts, and those species for which the date of first record seems a clear artifact of the arrival or participation of an interested taxonomist (e. g. Olga Hartman in the 1930s (polychaetes), Eugene Kozloff in the 1940s (symbiotic protozoans), Willard Hartman in the 1950s (sponges), and Ralph Brinkhurst in the 1960s (oligochaetes)), or an artifact of an especially focused sampling effort (e. g. the Albatross survey of 1912-23, and our survey of Bay fouling communities in 1993-95), or simply the fortuitous discovery of a species in a restricted habitat or locality (such as Transorchestia enigmatica, known only from the shore of Lake Merritt, and Littorina saxatilis, known only from ten meters of cobbly beach in the Emeryville Marina), and whose inclusion would provide a misleading view of the invasion history of the Estuary. These species are marked with an asterisk (*) in Table 1.
The dates of first record were tabulated in five time periods (four 30-year periods and one 26-year period) beginning in 1850. Tabulations of the dates of first record in the Estuary are shown in Figure 5, and of the dates of first record in the northeastern Pacific region in Figure 6. The results show a clear trend toward more first records in more recent periods. Over 40% of the first records of introductions in the Estuary date from 1970 or later, and over 63% from 1940 or later. Since the first records for the northeastern Pacific are inclusive of the records for the Estuary, they necessarily average somewhat earlier; nevertheless, 51% still date from 1940 or later. Some of these results should be interpreted with caution. The dates of arrival must of course precede the dates of first record, by an unknown but possibly significant average period. And although we have excluded records that would cause a specific and obvious temporal bias, there might exist a general bias toward increasing numbers of first records, which could be caused by such changes as an increase in sampling effort, by the development of improved techniques for sampling and sorting, by a general increase in taxonomic knowledge, by an increased availability and improvement of keys and other identification tools, or by other changes.
On the other hand, several factors in the analysis create a bias toward a lower number of first records in the most recent period relative to earlier periods.
· The length of the most recent period is a little under 26 years long, compared to 30 years for the earlier periods. Extrapolating to 30 years at the same rate of production of first records as has prevailed in the period so far would add another 9 species to the recent period's tally for the Estuary, and 7 species to the tally for the northeastern Pacific.
· While a substantial number of first records were excluded (for the reasons discussed above) from the third, fourth and fifth periods, virtually none were excluded from the first two periods.
· Some organisms collected in the most recent period but
excluded from the list of introductions because of inadequate
evidence to determine whether they are established (see Table
8) will probably, with the passage of time, be recognized as established.
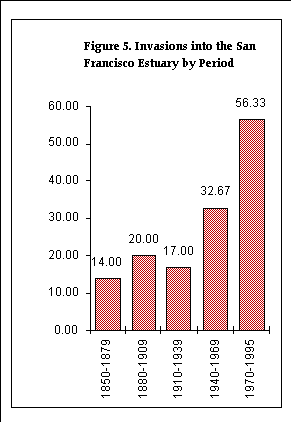
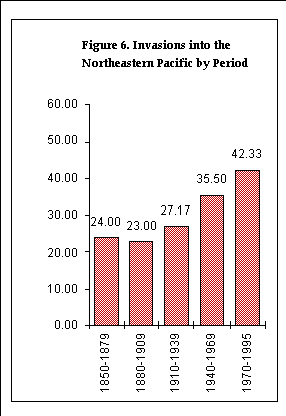
· With the passage of time, the taxonomic problems that bar
the listing of some species will be resolved. There appear to
be a substantial number of species that were only recently recorded
from the Estuary that fall into this category.
Taking these factors into account, it appears that the data signal
a substantial pulse of invasions detected in the Estuary since
1970. The overall rate of introductions to the Estuary (212 species
between 1850 and 1995) averages one new species established every
36 weeks. In the period since 1970, the dates of first record
indicate a rate of one new species every 24 weeks (even after
excluding one-third of the 212 documented introductions from the
analysis, for reasons discussed above).
Carlton (1994) presented a tabular overview of global dispersal mechanisms by human agencies in five broad categories: (1) Vessels; (2) Aquaculture, Fisheries, and Aquarium Industries; (3) Other Commercial, Government, and Private Activities; (4) Scientific Research; and (5) Canals. These have been reviewed in detail by Carlton (1979a, 1979b, 1985, 1987, 1992a) and by Carlton et al. (1995). Our data indicate that all of these mechanisms except for canals have served to transport non-native species to the San Francisco Bay area. Within these categories, twelve mechanisms (Table 1) and their approximate time of initiation relative to human-mediated invasions of the San Francisco Estuary are summarize here (a thirteenth mechanism, "gradual spread," accounts for the arrival of a number of species, including muskrats, purple loosestrife, and watercress, all in the 20th century, that spread either naturally, by human activities, or both, from eastern to western North America).
We focus here primarily on those mechanisms that serve to transport
new species to the northeastern Pacific, rather than on intraregional
vectors. The latter may include, for example, the intentional
movement of fish between watersheds by members of the public with
the intent of establishing new populations for sport fisheries
or pest control (such as the mosquitofish Gambusia); the
accidental movement of invertebrates in river gravels dredged
for use as aggregate for concrete (such as the Asian clam Corbicula),
and the spreading of organisms by dredging activity (such as the
cordgrass Spartina alterniflora). No studies are available
on the scale or role of these within-system vectors. We note later
that such work would be of great value in terms of both understanding
dispersal potential and dispersal histories and in establishing
management policies.
The transport of marine organisms to San Francisco Bay by ships has been theoretically possible since the 16th century, when ships either traveling along the coast and passing by the entrance to the Bay, or making landfall on the shores of the gulf outside the Bay, could have released organisms that made their way into the Bay. Thus, for example, Carlton & Hodder (1995) have shown that vessels passing the California coast in the 1570s could have released larvae-laden hydroid polyps that could have drifted into the Bay. The first ship known to actually enter the Bay was the San Carlos, on August 5, 1775 (Galvin, 1971). By the turn of the 18th century a number of ships from the Atlantic and Pacific oceans had entered the Bay (Kemble, 1957). After 1849, international shipping to the Bay picked up dramatically due to a combination of the California Gold Rush, the increased export of lumber, grain, minerals, furs, hides, and other products from the rapidly developing industries of central California, and increased colonization and industrialization in general. Kemble (1957) reviews the general maritime history of the Bay area.
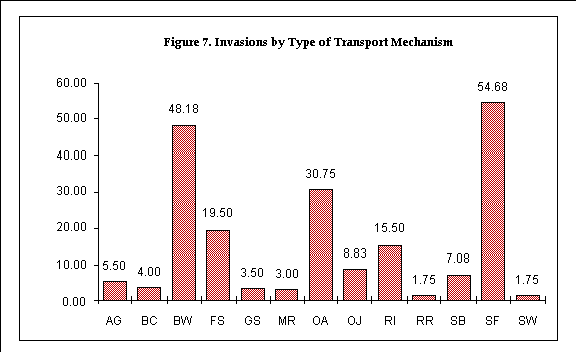
Little is known of the modern role of ship fouling in transporting marine animals and plants into San Francisco Bay, although there is evidence that this mechanism could assume an increasingly higher profile due to the decreasing use (for environmental reasons) globally of effective antifouling paints (such as those including tributyltins (TBTs)) (A. Taylor, BHP Inc., Australia, pers. comm., 1995).
The earliest clear records of ship fouling-mediated introductions
(though not recognized as such at the time) are the collections
of several North Atlantic fouling organisms in San Francisco Bay
between 1853 and 1860: the barnacle Balanus improvisus
(1853), the hydroid Tubularia crocea (1859) and the hydroid
Sarsia tubulosa (1860) (Table 1). Approximately 26 percent
of Bay invasions (55 species) have arrived by ship fouling and
boring (Figure 7).
No history of the release of ships' solid ballast into the Bay Area is available. It presumably parallels the general history of shipping into the Bay, but source regions for rock and sand ballast, amounts released, and so forth remain to be investigated.
That rock and sand ballast may have played an early role is suggested
by the appearance of the South African shore plant brass buttons
(Cotula coronopifolia) and the Atlantic marsh snail Ovatella
myosotis in the Bay in the 1870s (Table 1). Another example
of such activity was the release of ballast derived from Chilean
port regions (such as Iquique and Valparaiso) into the Oakland
Estuary up until about the 1920s, a transport vector that may
have led to the introduction of the southern hemisphere beach
hopper Transorchestia into nearby Lake Merritt. About 3
percent of Bay invasions (7 species) are linked to this mechanism
(Figure 7). It is probable that this is an underestimate, and
that with further studies more species (especially among non-crustacean
arthropods, such as coastal insects and spiders) will be found
to have been ballast-transported, similar to the studies of Lindroth
(1957) on North Atlantic beetles.
Ballast water may have been released into San Francisco Bay as early as the 1880s-1890s, but, as with solid ballast, the early history of ballast water in the Estuary remains to be studied. Of particular interest would be data on the timing of increased pulses of ballast water release into the Estuary. Modern ballast patterns for selected ports within San Francisco Bay have been investigated by Carlton et al. (1995). In the Ports of Oakland and San Francisco alone there were more than 2,000 arrivals of bulk cargo vessels and petroleum product tankers in 1991. "Acknowledged" ballast water released from those vessels in these two ports exceeded 130,000 metric tons (approximately 34,000,000 gallons) of water. "Unacknowledged" ballast water (water that is on board but not recorded because the vessel is classified as being "in cargo" rather than "in ballast") arriving in these two ports is estimated at approximately an additional 130,000 metric tons (34,000,000 gallons) (Carlton et al., 1995). Thus, more than 68 million gallons of ballast water per year are released by bulkers and tankers alone in the Central Bay area. Additional ports in the Bay system receiving large volumes of water include Sacramento and Stockton.
In 1991 the Ports of Oakland and San Francisco primarily received shipping from other North Pacific ports. Shipping from Asia accounted for 26 percent of ship arrivals in San Francisco and 48 percent in Oakland. Ships (and thus water) also arrived from Central Pacific and South Pacific ports and, to a smaller extent, from the Atlantic and Indian oceans (Carlton et al., 1995).
While some species may have been brought to the Estuary in the
first half of the 20th century by ballast water (Table 1), the
first reasonably unambiguous signal of the role of ballast water
was the arrival of two Asian species, the shrimp Palaemon macrodactylus
(first collected in 1957) and the Japanese goby Tridentiger
trigonocephalus (first collected in 1962). The arrival of
both may have been associated with increased transpacific shipping
related to the Korean War. Twenty-three percent (48 species) of
the Estuary's nonindigenous species are now linked to ballast
water transport, with a greatly increasing number of these apparently
having arrived since the 1960s (Figure 5). The pulse of recent
ballast invaders into the Estuary is particularly evident in the
discovery, since the 1970s, of 15 species of small Asian crustaceans
(copepods, one cumacean, one isopod, 3 mysids, and 2 amphipods),
and, since the 1980s, of two Asian clams (Potamocorbula
and Theora), one Japanese fish (Tridentiger bifasciatus),
and a New Zealand carnivorous sea slug (Philine). The appearance
of the Chinese mitten crab Eriocheir sinensis in the Bay
may also be linked to ballast water (but see mechanism 11, below).
The first Atlantic oysters were planted in San Francisco Bay in 1869, the year of the completion of the Transcontinental Railroad. Early shipments were largely from New York and New Jersey and occasionally from Chesapeake Bay. The industry grew and flourished in the 1890s, tapering off sharply after 1900 (for reasons variously cited as increases in pollution and changes in the Bay's hydrology and flushing dynamics; see Carlton, 1979a). The last oyster seed shipments occurred about 1910, and adult oysters continued to be received for holding in the Bay until the 1930s. Barrett (1963) and Carlton (1979a) review the history of Atlantic oystering in the Bay in detail.
The first Japanese oysters were planted out in the Bay in 1932, with plantings continuing until 1939. Occasional plantings for "experimental" purposes were started in the 1950s. Carlton (1979a) reviews this brief and little-known history.
The "signal" of Atlantic oystering in terms of invasions occurred early, with the appearance of the common Atlantic soft-shelled clam Mya arenaria in the Bay by 1874 (it was, oddly enough, not recognized as such, and described as a new species!). The Atlantic marsh snail Ovatella may have also arrived with oysters, if not with ship's ballast, at this time. Coincident, however, with the greatly increased pulse of plantings in the 1890s of Atlantic oysters was the appearance in the Bay of a variety of well-known East Coast clams and snails, including the oyster drill Urosalpinx (1890), the tiny gem clam Gemma (1893), the marsh mussel Arcuatula (=Ischadium) demissa (1894), two species of slipper limpets Crepidula convexa and plana (1898, 1901) and the mudsnail Ilyanassa (1907). Similarly, the Atlantic shell-boring sponge Cliona (1891) and the common Atlantic pileworm Nereis succinea (1896) had been recorded by this time. Thirty species representing about 15% of the introduced biota are now recognized as originating from Atlantic oystering activity.
In concert with the much lower level of Japanese oystering in
the Bay, only a few species in the Bay are recognized as having
arrived with this industry. After the pulse of 1930 plantings,
the Japanese mussel Musculista (1946) and the Japanese
clam Venerupis philippinarum (=Tapes japonica) (1946)
were collected in the Bay. The immediate role of Japanese oystering
in transporting other species is not as clear, as many candidate
taxa may also have entered the Bay by ship fouling or other means
(Table 1). The Japanese brown seaweed Sargassum muticum,
while apparently introduced to the Pacific coast by Japanese oystering,
may have entered the Bay as drift seaweed from elsewhere on the
coast or, even more likely, as fouling on coastal ship traffic.
The Japanese parasitic copepod Mytilicola may similarly
have been transported into the Bay in mussels in ship fouling
from more northern stations. About 4 percent of the Bay's invasions
are linked to Japanese oystering (Figure 5).
We review the early attempts to move Eastern fish West, facilitated
by the completion of the Transcontinental Railroad, in Chapter
3. American shad, white catfish, several species of bullhead,
and striped bass were all successfully transported, released,
and established in the Bay commencing in the 1870s. Intentional
fish stocking by government agencies of freshwater and estuarine
fish into California and the Bay region has continued to varying
degrees throughout the 20th century (see discussions in Chapter
3). Nineteen species (9 percent) of the exotic biota owe their
origins to this mechanism.
Plantings either for marsh restoration or possibly for erosion control were involved in the introduction of four species of the cordgrass Spartina in the Bay in the 1960s and 1970s. One was planted in Washington state, and then transplanted from there to San Francisco Bay; another was likely introduced to Washington in solid ballast, and later independently introduced to the Bay from the Atlantic coast for marsh restoration; the third was introduced to Humboldt Bay in solid ballast, then transplanted to San Francisco Bay; the fourth, first reported in the Bay in 1968, presumably arrived with an undocumented restoration or erosion control project (Chapter 3).
As we based our analysis on the mechanisms that brought to the
northeastern Pacific the stocks of organisms introduced to the
Estuary, we counted three of these cordgrasses as introduced via
marsh restoration or erosion control (1.4% of the exotic biota),
and one via solid ballast.
Accidental releases of plants, fish, and invertebrates through
stocking and planting programs began to be detected in the 1950s
in the Bay region, although these may have occurred much earlier.
Thus the rainwater killifish Lucania parva appeared in
1958 on the Bay's margins, apparently having been released accidentally
with shipments of other fish in more eastern localities. The green
sunfish and bigscale logperch, as well as the curly-leaf pondweed,
are additional accidental releases. Less than 3 percent of the
Estuary's invaders come under this category.
Miller (1969) first described this mechanism (focusing on lobster
packing) as an active vector for transporting northwestern Atlantic
marine organisms to San Francisco Bay. As discussed in Chapter
3 (under the periwinkle Littorina saxatilis), this mechanism
continues vigorously today. Large quantities of Atlantic bait
worms, and with them as packing material Atlantic rocky shore
seaweeds (mainly Ascophyllum nodosum), are air-shipped
weekly to sport-fishing supply stores in the Bay Area. Investigations
in progress (Lau, 1995; Cohen, Lau & Carlton, in prep.) reveal
that these seaweeds support large numbers of living Atlantic coast
invertebrates, including mollusks, worms, crustaceans, and insects,
which are routinely released into the Bay by anglers. The apparently
recent appearance of the Atlantic red alga Callithamnion
in the Bay, the establishment of a population of the Atlantic
periwinkle Littorina saxatilis, and perhaps even the appearance
of the Atlantic green crab Carcinus maenas may be linked
to this active and unregulated flow of New England rocky shore
organisms to the Bay. To date, less than one percent of the Estuary's
invaders are clearly linked to this mechanism, but the occasional
appearance of other species not yet known to be established (such
as the Atlantic periwinkle Littorina littorea; Table 8)
and the continual release of living seaweeds in the Bay which
could themselves become established (for example, Ascophyllum
nodosum has now gained a foothold in the Hood Canal, Puget
Sound; L. Goff, pers. comm., 1992), predictably herald the imminent
establishment of yet additional Atlantic species.
Invertebrates and fish released for biocontrol in the Bay region
have been few, although the release of muskellunge and sea lions
in San Francisco's Lake Merced to control introduced carp is a
noteworthy incident in the history of human attempts at biocontrol
(Chapter 2). Two South American weevils (Neochetina spp.)
were released in the 1980s for water hyacinth control; these became
established but appear to have had little impact on these weeds
(Chapter 3). An early introduction (1922) to the state was the
mosquitofish Gambusia affinis which arrived on Bay shores
at least by the 1960s if not much earlier. The inland silversides
Menidia beryllina, brought to the state for gnat and midge
control in 1967, soon entered (1971) Bay waters. These four species
represent about two percent of the Estuary's exotic biota.
Under this mechanism we include non-government releases to establish
food resources (the snail Cipangopaludina, the clam Corbicula,
the crayfish Procambarus clarkii, carp, bullfrog, and perhaps
the Chinese mitten crab Eriocheir sinensis and the pond
slider turtle); releases or escapes from residential ponds and
aquariums (plants (and oligochaete worms with them), possibly
the snail Melanoides, goldfish, carp, and the turtle);
escapes from commercial breeding or rearing ponds (crayfish, carp,
bullfrog) and discards of market goods (the snail Cipangopaludina
again). Fifteen species representing 7 percent of the introduced
biota have been linked to this mechanism according to our data.
With the possible exception of carp, water hyacinth and Cipangopaludina,
these have all been 20th century activities.
Scientific research efforts have resulted in relatively few introductions
to the Estuary. The bullfrog and the virile crayfish both owe
their establishment, at least in part, to releases from educational
and research institutions in the last half of this century. The
green crab Carcinus maenas, as noted below, may be a further
and more recent example of this vector. Less than one percent
of the Estuaries nonindigenous biota has arrived via this mechanism.
The complexities and challenges in analyzing and properly weighting
these many transport vectors, in terms of both developing an historical
perspective and establishing effective management options, is
illustrated by the many species in Table 1 for which multiple
transport vectors can be assigned. The recent appearance of the
Atlantic green crab Carcinus maenas in San Francisco Bay
is a superb illustration of the analytical and managerial hurdles
involved. The green crab could have arrived by at least four different
mechanisms (Cohen et al., 1995), whose relative likelihood is
difficult to estimate. As discussed in Chapter 3, it may have
arrived in ballast water from any of several different source
regions (Atlantic America, Australia, Europe or South Africa,
with the first two perhaps more likely based on shipping patterns);
via seaweed released from the bait worm industry; via active release
from a school or research aquarium; or via a ship's sea chest
or seawater pipe system. Clearly, the control of future invasions
hinges on a clearer and more detailed resolution of which mechanism
served to introduce Carcinus to the Bay. Recent collections
in the Estuary of the Atlantic amphipod Gammarus daiberi
(1983), the Atlantic worm Marenzelleria viridis (1991)
and the Atlantic snail Littorina saxatilis (1993) may point
to the Atlantic as the source region for Carcinus (1989/1990),
and may further suggest the modern resurgence of an active Northwest
Atlantic to San Francisco Bay transport corridor.
Nonindigenous aquatic animals and plants have had a profound impact on the Estuary's ecosystem. No habitatówith the possible exception of the deep floor of the Central Bayóremains uninvaded by exotic species, and in some habitats it is difficult to find any natives. The depth and extent of biological invasions now recognized for the Estuary is greater than for any other aquatic ecosystem in North America, a phenomenon which apparently results from a combination of factors, including: 150 years of intense human commercial activity involving both the frequent disturbance and alteration of the ecosystem and the importation of nonindigenous organisms (Nichols et al. 1986), the prior geological and ecological history of the Bay, and the amount of research into biological invasions in this system. Despite the intensity of research effort our understanding of the ecological and biological consequences of the estuary's nonindigenous biota, in terms of both the individual and the collective impacts of many species, remains strikingly limited.
A brief survey of the estuary reveals the scale of dominance by the nonindigenous biota. At the Bay's mouth, under the shadow of the Golden Gate Bridge, orange-red clumps of the Indo-Pacific bryozoan Watersipora, 30 centimeters across and 20 centimeters deep, covers the dock sides. To the north, in San Pablo and Suisun bays, the Chinese clam Potamocorbula forms thick beds in the mud while Japanese gobies and Korean shrimp swim overhead. In a brackish river a few kilometers distant large, coral-like masses formed from the calcareous tubes of an Australian serpulid worm harbor an abundant population of the Atlantic shore crab Rhithropanopeus. Upstream in the Delta a Eurasian freshwater hydroid forms thick colonies on ropes and marina floats. Swimming nearby may be any of several warmwater gamefish native to eastern North America, including six species of catfish, four species of sunfish and four species of bass.
Along the eastern and southern Bay shores, great masses of Atlantic and Asian seasquirts comprise the dominant fouling biota along with dense populations of bay mussels, represented in San Francisco Bay by both the native Mytilus trossulus and the Mediterranean Mytilus galloprovincialis. On the fringes of the Bay, dense beds of the New England ribbed mussel bind the upper intertidal sediments and lower marsh fringes, clonal colonies of the Atlantic cordgrass Spartina alterniflora encroach upon the mudflats, and a New Zealand burrowing isopod inexorably bores into the clay and mud banks of the Bay's shore. Moving in seasonal migrations over the mudflats, vast herds of the Atlantic mudsnail Ilyanassa rework the uppermost layers of sediment above the subsurface beds of the Atlantic softshell clam and the Japanese littleneck clam.
With seasonal changes, with dramatic interannual variation in the amount of freshwater runoff or saltwater intrusion, with the discharge of point-source or diffuse pollutants, and with many other variables, these associations of introduced species may shift significantly, but the overall aspect remains the same: the dominant members of many of the Bay and Delta aquatic communities are organisms that were not present 150 years ago.
Considered here are the ecological and biological impacts that
have been caused by the introduction of nonindigenous animals
and plants into the marine, brackish, and freshwater environments
of the Bay and Delta region. We review examples of communities
in which introduced species are the dominant members, both in
terms of diversity and biomass, consider trophic changes in the
Bay as a result of invasions, and then consider additional community-level
and habitat changes that have occurred. We conclude with prospects
for future invasions.
In some regions of the Estuary, 100% of the common species
are introduced.
As Carlton (1975, 1979a, 1979b), Nichols & Thompson (1985a,b) and Nichols & Pamatmat (1988) have noted, the shallow-water benthos of San Francisco Bay is dominated by nonindigenous speciesóindeed, Nichols & Thompson (1985b) have used the phrase, "introduced mudflat community" in reference to South San Francisco Bay. Nichols and Pamatmat (1988), in describing the Bay's soft-bottom benthic communities, state that:
"The principal contributors to biomass throughout much of
the bay are the mollusks Tapes [now Venerupis] philippinarum,
Musculista senhousia, Macoma balthica
[now petalum], Mya arenaria, Gemma
gemma, and Ilyanassa obsoleta. In addition,
the large tube-dwelling polychaete Asychis [now Sabaco]
elongata is a major contributor to total biomass in the
muddy subtidal areas of South Bay...[Since 1987] the Asian bivalve,
Potamocorbula amurensis...has become the dominant
macroinvertebrate throughout the northern portions of the bay
and is found in South Bay sloughs as well."
Each of these species is introduced to San Francisco Bay, arriving in the following approximate sequence:
Time of First Observation (O)
or Hypothesized Arrival (H)
Introduced with Atlantic Oysters
Atlantic soft-shell clam Mya early 1870s (O)
Atlantic tellinid clam Macoma 1870s-1890s (H)
Atlantic gem clam Gemma before 1893 (O)
Atlantic mudsnail Ilyanassa before 1907 (O)
Atlantic bamboo worm Sabaco after 1912 (H)
Introduced with Japanese Oysters
Japanese mussel Musculista before 1946 (O)
Japanese clam Venerupis before 1946 (O)
Introduced with Ballast Water
Chinese clam Potamocorbula before 1986 (O)
Although these nonindigenous species dominated the intertidal and subtidal mudflat communities, many other species of mollusks, crustaceans, polychaetes, and other invertebrates were added to the Bay's soft-bottom communities during these periods as well (Table 1). Each new addition or set of additions presumably altered the previously-existing community, in ways that may have prevented or facilitated the invasion of the next introduced species. While these "successional" concepts of the roles of inhibition or facilitation by preceding invaders are not well developed in invasion ecology, the assembly of these communities over a relatively long period of time, from different source regions (and thus of species that did not coevolve), may prove to be key factors in understanding the structure of invaded communities, and of which species do and do not invade.
A review of several faunal studies around the Bay conducted between the 1940s and 1970s (Carlton, 1979a; Table 4, herein) demonstrates the importance of introduced species in intertidal epifaunal (on the surface), intertidal infaunal (under the surface) and fouling communities. In locations ranging from freshwater sites in the Delta through estuarine sites in the northern bays, the Central Bay and the South Bay, introduced species account for the majority of the species diversity at most sites. On South Bay mudflats, Vassallo (1969) found that the infaunal communities could be characterized in terms of introduced species: the upper intertidal was essentially a "Macoma balthica community," whereas the lower intertidal was an "Ampelisca abdita community." At some sites, 100% of the common to abundant species were found to be introduced. We discuss later in this section the question of the replacement or displacement of a native biota by these introduced species.
Thus, extensive communities in the Bay are structured around introduced species: the abundant filter feeders, the abundant herbivores, the abundant detritivores, and the abundant carnivores are not native. With few exceptions, the introduced versus native status of the abundant primary producers (phytoplankton and algae) is not known, and thus the extent to which the entire food chain is constructed of invasions is not yet known. However, few, if any, of the estuarine phytoplankton or algae are clearly native. These communities are further composed of species originating from different regions of the worldóspecies that evolved in the presence of other species (that did not arrive with them in San Francisco Bay) and that evolved under different environmental regimes. The extent to which these introduced species, artificially placed together in a novel environment, are undergoing coadaptation, in terms of predator-prey relationships or competitive interactions, remains unknown.
The predominance of nonnative species in the Bay's communities
suggest that a vast amount of energy, in terms of dissolved organic
and inorganic compounds, and in terms of primary and secondary
production, now pass through and are utilized by the nonindigenous
biota of the Bay. We explore some of these trophic changes below,
as well as the role of competition, habitat alterations, and the
regional or global extirpation of native species.
Table 4. Associations of Introduced Species in
the San Francisco Estuary.
The number and percentage of introduced species (excluding
cryptogenic species) in selected communities.
Reference
Location Number of Introduced Species [date of collections]
DELTA & SUISUN BAY
Antioch and Bradford 6 out of 7 (= 86%) epibenthic/fouling species are Aldrich, 1961
introduced.
Sacramento River, 3 out of 5 (=60%) dominant benthic species are Siegfried et al.,
Decker Is. to Chipps Is. introduced. 1980 [1976]
Delta to Grizzly Bay 2 out of 4 (=50%) dominant benthic species are Markmann, 1986
introduced. [1975-81]
Suisun Bay 4 out of 7 (=57%) common benthic species
are Nichols & introduced. Thompson, 1985a
Grizzly Bay to Old River 2 out of 5 (=40%) dominant benthic species are Herbold & Moyle
introduced. 1989 [1983-84]
Delta 26 out of 52 (=50%) fish present, and 25 of 36 Herbold & Moyle,
(=69%) fish resident, in the Delta are introduced.
1989
Delta: Old River, Frank's 6 out of 22 (=27%) benthic invertebrate species Hymanson et al.,
Tract and Sherman Lake are introduced. 1984 [1980-90]
Sacramento River at 10 out of 17 (=59%) benthic invertebrate species Hymanson et al.,
Sherman Island are introduced. 1984 [1980-90]
Grizzly Bay 16 out of 19 (=84%) benthic invertebrate species Hymanson et al.,
are introduced. 1984 [1980-90]
SAN PABLO BAY
San Pablo Bay east 8 out of 13 (= 62%) epifaunal species, and 16 out Filice, 1959
to the Delta of 17 (= 94%) infaunal species are introduced.*
Carquinez Strait 7 out of 7 (=100%) of common benthic species are Markmann, 1986
introduced. [1975-81]
San Pablo Bay shallows 9 out of 9 (=100%) common benthic species are Nichols & introduced. Thompson, 1985a
CENTRAL BAY
Oakland Estuary All 4 species (= 100%) dominant in the fouling Graham & Gay,
fauna are introduced.* 1945 [1940-42]
Lake Merritt 31 out of 35 (= 88%) epifaunal species, and 6 out Carlton, 1979a
of 8 (= 75%) infaunal species are introduced.* [1962-72]
Table 4. Associations of Introduced Species -
continued
Location Number of Introduced Species Reference
[date of collections]
SOUTH BAY
Hayward 4 out of 5 (= 80%) upper intertidal infaunal species Vassallo, 1969
are introduced. The infauna is numerically
dominated by the introduced clam Macoma petalum;
the epifauna is numerically dominated by the
introduced mudsnail Ilyanassa obsoleta.
7 out of 9 (= 77%) lower intertidal infaunal species are
introduced. The community is numerically dominated
by the introduced amphipod Ampelisca abdita.
Palo Alto 14 out of 14 (=100%) species of mudflat infauna Nichols, 1977
and epifauna are introduced.
South Bay channels 10 out of 10 (=100%) common benthic species in the Nichols & channels, and 6 out of 6 (=100%) dominant benthic Thompson (1985a)
species in the shallows are introduced.
* For these calculations, all mussels reported as
Mytilus edulis were assumed to be native.
In the 1990s, introduced and cryptogenic species dominate
the Estuary's food webs.
We consider here trophic alterations to the Bay's ecosystem by
introduced species utilizing different feeding levels and strategies:
the phytoplankton, the zooplankton, water column consumers (filter
feeders), epibenthic and shallow-infaunal grazers and deposit
feeders, and carnivores.
Although various mechanisms have transported and continue to transport large numbers of nonindigenous phytoplankton to the San Francisco Bay and Delta (today mainly via ballast water, but in the past including settled diatoms transported with oysters and freshwater phytoplankton in the water used to transport game fish), and researchers have identified introduced diatoms and dinoflagellates in other areas of the world (in Australia: Hallegraeff, 1993; Hallegraeff and Bolch, 1992; in Europe: Boalch, 1994; in the Great Lakes: Mills et al., 1993), none of the phytoplankton in the estuary have yet been reported as introduced species. We consider at least 31 species of phytoplankton to be cryptogenic (Table 2), which is probably only a small fraction of the total number of planktonic, benthic, and epibiotic species that have been introduced to the Bay and Delta system.
The diatoms Cyclotella caspia, Coscinodiscus spp., Aulacoseira (=Melosira) spp., Aulacoseira (=Melosira) distans variety lirata, Skeletonema costatum and Thalassiosira decipiens and the microflagellate Chroomonas minuta are dominant and important members of the phytoplankton in San Francisco Bay (Cloern et al.,1985). All are broadly distributed globally and are cryptogenic species in San Francisco Bay. The diatom Aulacoseira granulata (=Melosira granulata, Round et al., 1990) has recently come to dominate phytoplankton blooms in the San Joaquin River (Herbold & Moyle, 1989). In Suisun Bay, the diatom Thalassiosira decipiens alternates between dominating the water column or the benthos, apparently depending upon the degree of water column mixing (Cloern et al., 1985; Nichols and Pamatmat, 1988). Both Aulacoseira granulata and Thalassiosira decipiens are cosmopolitan species (e.g., Cholnoky, 1968) and may well be introductions in the Bay system.
While these taxa are also often reported from open-ocean systems, including upwellings, the possibility remains that these brackish water and freshwater diatoms represent estuarine genotypes transported by oysters and ships around the world, and may be distinct from the oceanic genotypes transported by ocean currents. A similar example has been provided by Greenberg (1995), who found that the estuarine populations of the jellyfish Aurelia aurita in San Francisco Bay are closely related to those from Japan (and thus probable ship-borne introductions as attached fouling scyphistomae or planktonic ephyrae), and less similar genetically to coastal populations from Monterey Bay.
Thus, it remains possible that many of the estuary's major phytoplankton
species, accounting for the bulk of the estuary's primary production,
are in fact introduced. Resolution of these cryptogenic diatoms
as native or exotic would significantly improve our understanding
of the origin and structure of the Bay and Delta's food webs;
and is essential to developing a correct interpretation of their
biology and their patterns of distribution and abundance in terms
of, on the one hand, adaptation to and co-evolution with the estuary's
physical conditions and other biota, or on the other, opportunistic
establishment and exploitation of available resources.
The planktonic secondary producers are represented by a diverse zooplankton community in San Francisco Bay. Many copepod species in San Francisco Bay are considered widespread if not cosmopolitan, and thus those susceptible to human transport mechanisms should be considered cryptogenic species. Notable in this regard, for example, are the abundant estuarine copepod Eurytemora affinis and the estuarine rotifer Synchaeta bicornis, which often characterize the zooplankton communities of the Sacramento-San Joaquin Delta (Orsi & Mecum, 1986) and whose biogeographic status remains unresolved. Eurytemora affinis in particular has been suspected of being an introduced species (Orsi, 1995). Similarly, some microplankton in the Bay are candidate cryptogenic species: the cosmopolitan estuarine ciliate Mesodinium rubrum, for example, caused red tides in South San Francisco Bay in spring 1993 (Cloern et al., 1994).
While the diverse meroplanktonic larvae of the large numbers of introduced benthic invertebrates and fish must play a role in water column dynamics, no studies appear to be available on this aspect of zooplankton trophic dynamics for the Bay. Mills and Sommer (1995) have noted that the introduced hydromedusae Maeotias inexspectata and Blackfordia virginica in San Francisco Bay estuarine tributaries fed almost exclusively on barnacle larvae, copepods, and the larvae of the introduced crab Rhithropanopeus. Whether these jellyfish decrease the abundance of their prey in an ecologically significant manner remains to be determined. Maeotias and Blackfordia are two of a large number of new invasive zooplanktonic organisms that have been recorded from the estuary since the 1970s, including another hydromedusan (Cladonema uchidai), the Japanese stock of the moon jelly Aurelia aurita, eight species of Asian copepods, three species of mysids and the demersal (vertically migrating) Japanese cumacean Nippoleucon (=Hemileucon) hinumensis.
The role of this new guild of often abundant Asian copepods and mysids in the upper estuary is of particular interest. Complicating both speculations and interpretations, however, are the number and interrelationships of the potential factors that control copepod abundance. Changing densities and distributions of copepods may be correlated with fluctuations in environmental parameters (such as salinity, temperature and chlorophyll concentration), predator abundance (including carnivorous zooplankton, fish and benthic filter-feeders (such as the Asian clam Potamocorbula) capable of zooplanktivory), selective predation on different copepod species, competition between copepod species (the intensity of which may be moderated by food availability), and declines in the overall abundance of zooplankton (reducing interspecific competition and making more food available).
Orsi et al. (1983) speculated that competition between the Chinese copepod Sinocalanus doerri and the "native" copepod Eurytemora affinis (considered here to be cryptogenic) was not likely because they preferred different salinity regimes; rather, competition and/or predation between Sinocalanus and the presumably native freshwater copepods Cyclops and Diaptomus appeared to be more likely. Herbold et al. (1992) noted that the introduction of Sinocalanus and Pseudodiaptomus forbesii was followed by a decline in Eurytemora and almost complete elimination of Diaptomus, implying potential interactions between these new invaders and the previous copepod residents. Meng and Orsi (1991) further found in laboratory experiments that the larvae of striped bass (itself an introduced species) selected Cyclops and Eurytemora over Sinocalanus (perhaps because of differences in copepod swimming and escape behavior). Thus, the possibility arises that the striped bass larvae's preferred prey is being replaced by an introduced, and less preferred, prey.
A further complication, however, arises when the role of the newly
introduced clam Potamocorbula is considered, which involves
both the consumption of phytoplankton, thereby removing a significant
portion of the potential food resource for water-column zooplankton,
and the consumption of the zooplankton themselves. Thus, as reviewed
below, Kimmerer et al. (1994) show that the decline in Eurytemora
was likely due to consumption by Potamocorbula, rather
than by interspecific copepod competition. Indeed, Potamocorbula
consumes Eurytemora and not Pseudodiaptomus (Kimmerer,
1991), further reducing the preferred copepod resource of striped
bass larvae.
Introduced clams can filter the entire volume of the South
Bay and Suisun Bay at least once a day.
A large number of nonindigenous suspension-feeding organisms are now filtering the waters of the estuary. In the intertidal and sublittoral soft-bottom sediments these include the introduced bivalves Macoma petalum (="balthica"), Venerupis, Mya, Potamocorbula, Theora, Petricolaria, Gemma, Arcuatula, Musculista and Corbicula, most of which are abundant to extremely abundant in the estuary. Introduced, suspension-feeding polychaete worms, especially spionids, and suspension-feeding tubicolous gammarid amphipods may occur by the thousands per square meter at and near the sediment surface. Intertidal and subtidal hard substrates are often thickly-coated, sometimes several organisms deep, with dense populations of introduced macrofilterers (including the seasquirts Molgula, Styela clava, Botryllus spp., Ciona spp. and Ascidiaósee Whitlatch et al., 1995, regarding the complex roles of Styela clava and Botrylloides diegensis, both introduced into Long Island Sound, in regulating community dynamics) and introduced microfilterers (including bryozoans and sponges). Introduced carnivorous suspension feeders, such as hydroids and sea anemones, can also be abundant: dense populations of the Indian Ocean hydroid Bimeria franciscana occur on floats in brackish tributaries, while the exotic sea anemone Diadumene franciscana is sometimes found in dense clonal clusters on marina floats on the southwestern shore of the Bay. Both doubtless have an impact on adjacent plankton communities. In some parts of the estuary the Mediterranean mussel Mytilus galloprovincialis and two introduced barnacles, Balanus improvisus and Balanus amphitrite, are exceedingly abundant filter-feeders on all hard substrates.
We consider in detail below the role of the benthic filter-feeding bivalve guild in regulating phytoplankton production in San Francisco Bay. The holistic role of the entire nonindigenous filter-feeding guildóclams, mussels, bryozoans, barnacles, amphipods, seasquirts, spionids, serpulids, sponges, hydroids, and sea anemonesóin altering and controlling the trophic dynamics of the Bay-Delta system remains unknown. The potential role of just one species, the Atlantic ribbed horsemussel Arcuatula demissa, provides insight into the potentially profound impact of introduced filter feeders on the estuary's ecosystems. Studying the energy flow in these mussels in a Georgia marsh, Kuenzler (1961) reported that,
"The mussels... have a definite effect upon the water over the marsh, daily removing one-third of the particulate phosphorus from suspension. They regenerate a small part of this into phosphate, and reject the remainder in pseudofeces and feces which drop to the mud surface. It appears, therefore, that the mussel population may be very important in the phosphate cycle as a depositional agent, furnishing raw materials to deposit-feeders which regenerate the phosphorus."
The potential tantalizing role of Arcuatula in the economy
of Bay marshes as a biogeochemical agent remains to be investigated.
In two fundamental papers, Cloern (1982) and Officer et al. (1982) demonstrated that the primary mechanism controlling phytoplankton biomass during summer and fall in South San Francisco Bay is "grazing" (filter feeding) by benthic organisms, in particular the introduced Atlantic gem clam Gemma gemma and the introduced Japanese bivalves Musculista (as Musculus) senhousia and Venerupis philippinarum (as Tapes japonica). [footnote on page 209]
Cloern (1982) calculated that "suspension-feeding bivalves
are sufficiently abundant to filter a volume equivalent to the
volume of South Bay at least once daily" (emphasis
added). This remarkable process must have a significant impact
on the standing phytoplankton stock in the South Bay; and with
nearly the entire primary production of the South Bay potentially
passing through the guts of introduced clams, this may have fundamentally
altered the energy available for native biota.
Nichols (1985) extended this model of benthic control of water column production to the northern Bay. He noted that during the central California drought of 1976-1977, several species typically more common west of Carquinez Strait invaded and became abundant in Suisun Bay (including four introduced Atlantic species: the clam Mya arenaria (which Nichols noted was introduced), the amphipods Corophium acherusicum and Ampelisca abdita, and the spionid polychaete Streblospio benedicti. In addition, a resident species, the tellinid clam Macoma balthica (now Macoma petalum, see Chapter 3), increased in abundance; this species too is introduced. With the arrival of these species and the increase in Macoma, total community abundance peaked at 153,000/m2 at one site in 1976 and 20,000/m2 at one site in 1977. During these two years, the usual summer diatom bloom failed to appear (Cloern et al. 1983). Nichols (1985) proposed that this guild of estuarine invaders led to increase benthic "grazing" (filter feeding), particularly by the clam Mya, but also by the other species (Nichols noted, for example, that the worm Streblospio switches from deposit feeding to suspension feeding at higher phytoplankton concentrations). Indeed, Nichols estimated that Mya alone "could have filtered all of the particles (including the diatoms) from the water column on the order of once per day" (emphasis added).
Cloern et al. (1983) noted that the presumably native phytoplanktivorous
mysid (opossum) shrimp Neomysis mercedis suffered
a "near-complete collapse" in the Suisun estuary in
1977, which they describe in part as a potential result of food
limitation. In turn, 1977 was a year of record low abundance of
juvenile striped bass in the north Bay; larval bass rely heavily
on the mysid Neomysis (Cloern et al. 1983). Both collapses
may have been "a direct consequence of low phytoplankton
biomass" (Nichols, 1985), which, if Nichols is correct in
linking the decline of the phytoplankton standing stock to a rise
in benthic bivalve grazing, provides a direct and remarkable example
of the potential impact of an introduced species on the Bay's
food web. Thus:
Populations of the Atlantic Clam Mya arenaria
>>Significantly Reduces Phytoplankton Standing Stock
>>Leads to a Decline in Zooplankton (e. g. Mysids)
>>Leads to a Decline in Fish (e. g. Juvenile Striped Bass)
At about the same time (1985) that Nichols first proposed that introduced clams could be controlling primary productivity in Suisun Bay, a ship inbound from China was deballasting into Suisun Bay a species of clam that would vastly overshadow the trophic impact of the existing guild of benthic phytoplanktivores. In October 1986 three specimens of Potamocorbula amurensis, a species previously known only from Asian waters, were collected in Suisun Bay. By the following summer, Potamocorbula was the most abundant benthic macro-organism in Suisun bay, achieving average densities of over 2,000/m2, and peak densities at some sites of over 10,000/m2. Potamocorbula has since spread and become the dominant subtidal clam in San Pablo Bay and South Bay as well.
What has been the impact of adding Potamocorbula to the Bay's ecosystem? Alpine and Cloern (1992) calculated that the mean annual primary production in Suisun Bay during the years of lower benthic clam density (<2,000 clams/m2) was 106 grams of carbon/m2, compared to an estimated mean annual production of only 39 grams/m2 when clams were dense (>2,000 clams/m2; these clams were mainly Potamocorbula, but included some Mya, whose densities declined sharply after the arrival of PotamocorbulaóNichols et al., 1990). Thus, since the proliferation and spread of Potamocorbula in 1987, the summer phytoplankton biomass maximum in the northern estuary (the diatom bloom) has disappeared, presumably because of feeding by this new invader. Thus since 1987, the invasion of the Bay by Potamocorbula has added a striking and persistent "top down" level of control to biological productivity in the estuary.
Werner and Hollibaugh (1993) may have recently provided the answer to one of the puzzles associated with the radical alteration of the estuary by Potamocorbula: if the phytoplankton bloom has been eliminated by Potamocorbula's filter feeding, then what are those billions of clams now eating? (Cohen, 1990). Werner and Hollibaugh showed that Potamocorbula consumes bacteria as well as phytoplankton. Though it consumes bacteria at lower efficiency than diatoms, Potamocorbula assimilates both with high efficiency. At present densities in northern San Francisco Bay, Potamocorbula is capable of filtering the entire water column over the deep channels more than once per day and over the shallows almost 13 times per day, a rate of filtration which exceeds the phytoplankton's specific growth rate and approaches or exceeds the bacterioplankton's specific growth rate.
Kimmerer et al. (1994) have now provided evidence that Potamocorbula
substantially reduces zooplanktonic copepod populations in the
North Bay by direct predation. Thus, Potamocorbula operates
at multiple levels in the food chain: not only does it reduce
phytoplankton (which would indirectly lead to reductions in zooplankton),
but it also directly consumes zooplankton. It will be both critical
to our understanding of the trophic dynamics of the estuary and
inordinately challenging to sort out the complex and changing
interrelationships of (a) these two levels of Potamocorbula's
interaction with the food chain, (b) competition between Potamocorbula
and other introduced and native benthic filter feeders, (c) the
roles of additional first and second order consumers introduced
to the zooplankton (copepods and mysids) in reducing phytoplankton
stocks, (d) the role of interspecific competition between and
among introduced and native copepods and mysids, (e) selective
predation by higher order consumers, many of them introduced fish
species, on the zooplankton, and (f) competition between and among
both introduced and native higher order consumers. Invasions by
new species of phytoplankton, zooplankton, and benthic filter
feeders in the Bayóinvasions that can be predicted with
some degree of confidence (Chapter X)ówill add further
complexities to this framework.
Benthic non-filter feeding invaders in San Francisco Bay include a number of carnivores and omnivores (considered below) as well as epibenthic and shallow infaunal grazers on surface sediments. The latter include a number of species of introduced polychaetes (such as the extremely abundant maldanid worm Sabaco) which act as selective or non-selective deposit feeders, interfacial bivalves such as Macoma petalum, which uses its siphons to graze on the mud surface but can also suspension feed, grazing peracarid crustaceans (including many introduced species of amphipods, isopods, tanaids, cumaceans and mysids), and the Atlantic mudsnail Ilyanassa obsoleta.
The recent discovery of the deposit-feeding Atlantic spionid Marenzelleria viridis in San Francisco Bay is of particular interest. Marenzelleria was transported by ballast water to western Europe in the 1980s and has since become one of the most common macrobenthic species in the North and Baltic Seas (Essink and Kleef, 1993; Bastrop et al., 1995). Preliminary studies reveal a variety of species interactions, in particular a significant positive relationship between increasing densities of Marenzelleria and increasing densities of Corophium, although the mechanism of this interaction is not known (Essink and Kleef, 1993).
As with the guild of filter feeders, the overall picture of the
impact of introduced grazers and deposit feeders in the San Francisco
Bay and Delta is not known. Based upon Atlantic studies, however,
it can be predicted that the mudsnail Ilyanassa is playing
a significantóif not criticalórole in altering the
diversity, abundance, size distribution, and recruitment of many
species on intertidal mudflats of San Francisco Bay. Millions
of migrating mudsnails sweep large areas of mudflat clear of epibenthic
diatoms (JTC, pers. obs., Barnstable Harbor, MA), and Ilyanassa
has further been shown to be an opportunistic omnivore, consuming
spionid worms and littorinid snail egg cases (Brenchley &
Carlton, 1983).
"... the arrival and establishment of the green crab signals another potentially exceptional level of ecosystem change in San Francisco Bay..."
óCohen et al. (1995)
".... Carcinus maenas will significantly alter community structure, ecological interactions, and evolutionary processes in embayments of western North America"
óGrosholz & Ruiz (1995)
Introduced carnivorous and omnivorous crabs, snails, fish and terrestrial mammals undoubtedly have broad impacts throughout the San Francisco Bay and Delta ecosystem. Smaller introduced carnivores are now present (and often abundant) throughout the Bay. These include on soft sediments the recently introduced clam-eating slug Philine auriformis from New Zealand; on rocks and pilings the Atlantic barnacle-eating oyster drill Urosalpinx cinerea; and in hydroid masses on floats and navigation buoys the large Japanese isopod Synidotea laevidorsalis. We consider (here and in Section 5 below) three categories of carnivorous invaders in the estuary: the European green crab Carcinus maenas, introduced anadromous and warmwater gamefish, and introduced mammals.
The potential and observed roles of Carcinus maenas, first collected in California in 1989-1990 in the Estero Americano and in San Francisco Bay, have been addressed at length by Cohen et al. (1995) and by Grosholz & Ruiz (1995), the essence of whose findings have been quoted above. Cohen et al. (1995) noted that Carcinus consumes "an enormous variety of prey items," including organisms from five plant and protist phyla and 14 animal phyla. They predict that Carcinus will prey on many of the previously introduced species in San Francisco Bayóboth epifaunal and infaunal taxaówith the clam Potamocorbula being a potential major prey item. Carcinus' habitat range includes marshes, rocky substrates and fouling communities, and the European and New England literature indicates broad and striking potential for this crab to become an important carnivore in these systems (Cohen et al., 1995). Grosholz & Ruiz (1995) report that Carcinus has already "significantly reduced densities" of the most abundant near-surface dwellers in Bodega Harbor, 75 km to the north of San Francisco. These taxa included the native bivalves Transennella spp., the cumacean Cumella vulgaris and the amphipod Corophium sp. In laboratory experiments, Carcinus captured and consumed Dungeness crab (Cancer magister) up to its own size.
The twenty-eight species of introduced anadromous, freshwater
or euryhaline fish in the estuary include many important carnivores
now found throughout the upper estuary. In particular, carp, mosquitofish,
catfish, green sunfish, bluegill, inland silverside, largemouth
and smallmouth bass, and striped bass have been found to be among
the most significant predators throughout the brackish and freshwater
reaches of the Delta. Of particular concern is the extent to which
these introduced fish have reduced populations or contributed
to the local or global extinction of native California fish. Evidence
for interference, reduction, and destruction of spawning and nursery
sites of native species, and the extirpation of native fish from
feeding grounds, has been found for introduced carp, catfish,
green sunfish and bluegill.
Little is known of pre-1850 Bay and Delta ecosystems by which to determine the diversity and density of the aboriginal aquatic biota, and thus assessments of whether introduced species replaced or displaced abundant native organisms are severely constrained. Stimpson (1857) implied (though he may have been speaking of echinoderms only) that the invertebrate fauna of the Bay was depauperate in both species and numbers of individuals, although it is possible that even by Stimpson's time the virtual elimination of a top level predator (the aboriginal Indian population) in the Bay Area had led to a top-down cascade of faunal changes; or that the elimination of a keystone species controlling habitat structure in the watershed (beaver), acting through effects on anadromous fish populations, could have similarly initiated a cascade effect (McEvoy, 1986). Nevertheless, despite the limitations on our knowledge of the Estuary's native fauna, it is clear that in certain habitats there were no native species in some taxonomic groups and trophic guilds.
Table 5 shows the patterns of spatial relationship between native and introduced invertebrates along the marine to freshwater gradient in the Estuary. These patterns suggest that at least for some invading species, resources were available that were not being comparably utilized by native taxa, perhaps facilitating the initial invasion and establishment of the exotic species. (The terms "open niche," "empty niche" or "vacant niche," sometimes applied to such situations, are misnomers. A "niche" refers to the living conditions of an existing species, not to imaginary ecologic space, open or otherwise; see Herbold & Moyle, 1986.)
The most common spatial pattern of invasion in the Estuary is
for introduced species to occupy regions partially or wholly upstream
of their apparent native counterpart species. These introduced
and native counterparts may compete where their ranges in the
Estuary overlap, but in many cases in at least part of its range,
the introduced species is free from such competition. An example
is the introduced Atlantic crab Rhithropanopeus harrisii
which exists in the upper Bay and Delta at salinities below the
3 ppt tolerance limit of the native crab Hemigrapsus oregonensis.
In turn, however, Hemigrapsus, through predation and possibly
through competitive interactions, may limit Rhithropanopeus'
downstream expansion (Jordan, 1989).
Table 5. Patterns of Invasion Along the Salinity
Gradient in the San Francisco Estuary and the Adjoining Coast
Native species are listed in normal type. Invading
species are listed in bold type.
Marine Mesohaline Oligohaline Fresh
PATTERN: UPSTREAM INVADERS
Microcionid Sponges:
Microciona microjoanna Microciona prolifera
None None
Halichondriid Sponges:
Halichondria panicea Halichondria None None
bowerbanki
Acontiate Anemones:
Metridium senile Metridium senile ? None None
Diadumene franciscana
Diadumene ?cincta
Diadumene leucolena
Diadumene lineata
Tubeworms (Serpulid Polychaetes):
Serpula "vermicularis" Ficopomatus Ficopomatus None
enigmaticus enigmaticus
Flattened, Nestling Slipper Shells:
Crepidula nummariaa Crepidula plana None None
Crepidula perforansa
Convex Slipper Shells
Crepidula adunca Crepidula convexa
None None
Muricid Snails:
Ocenebra circumtexta Urosalpinx cinerea None None
Ocenebra lurida
Mussels in the Genus Mytilus:
Mytilus californianus Mytilus trossulus None None
Mytilus galloprovincialis
Gem Clams:
Transennella confusa Transennella tantilla ? None None
Transennella tantilla Gemma gemma
Littleneck Clams:
Protothaca staminea Protothaca staminea None None
Venerupis
philippinarum
Table 5. Patterns of Invasion Along the Salinity
Gradient - continued
Marine Mesohaline Oligohaline Fresh
Macoma Clams:
Macoma secta Macoma nasuta Macoma petalum None
Macoma inquinata Macoma petalum
Macoma nasuta
Shipworms:
Bankia setacea Teredo navalis None None
Lyrodus pedicellatus
Teredo navalis
Barnacles:
Balanus crenatus Balanus glandula Balanus improvisus Balanus improvisus b
Balanus glandula Balanus improvisus
Cirolanid Isopods:
Cirolana harfordi Eurylana arcuata
None None
Hydroid-eating Idoteid Isopods:
Synidotea bicuspida Synidotea None None
Synidotea ritteri laevidorsalis
Tanaids:
Leptochelia dubiac
Sinelobus sp.
Mud Crabs:
Hemigrapsus nudus Hemigrapsus Rhithropanopeus Rhithropanopeus b
Hemigrapsus oregonensis harrisii harrisii
oregonensis
Entoprocts:
Barentsia gracilis Barentsia benedeni
None Urnatella gracilis
Arborescent Bryozoans in the Genus Bugula:
Bugula californica Bugula neritina None None
Bugula pacifica Bugula stolonifera
Bugula neritina
Phlebobranch Sea Squirts:
Ascidia ceratodes Ascidia sp. None None
Corella sp. Ciona intestinalis
Chelyosoma productum Ciona savignyi
Simple Stolidobranch Sea Squirts:
Styela truncata Styela montereyensis Molgula manhattensis None
Styela montereyensis Styela clava
Pyura haustor Molgula manhattensis
Table 5. Patterns of Invasion Along the Salinity
Gradient - continued
Marine Mesohaline Oligohaline Fresh
Gobies
Clevelandia ios Clevelandia ios Eucyclogobius Tridentiger bifasciatus
Coryphopterus nicholsii Eucyclogobius newberryi d Acanthogobius
newberryi d Lepidogobius lepidus flavimanus
Lepidogobius lepidus Tridentiger bifasciatus
Tridentiger Acanthogobius
trigonocephalus flavimanus
Gillichthys mirabilis
Acanthogobius
flavimanus
PATTERN: INSERTION INVADERS
Pileworms (Nereid Polychaetes):
Nereis vexillosa Nereis succinea Nereis
succinea Hediste limnicola
PATTERN: DOWNSTREAM INVADERS
Tube-dwelling Corophium Amphipods:
None Corophium acherusicum Corophium spinicorne Corophium spinicorne
Corophium alienense Corophium stimpsoni Corophium stimpsoni
Corophium insidiosum Corophium acherusicum
Corophium Corophium alienense
heteroceratum
OTHER PATTERNS OF INVASION
Palaemonid Shrimp:
None Palaemon Palaemon None
macrodactylus macrodactylus
Intertidal Mudsnails:
None Cerithidea californica e None None
Ilyanassa obsoleta
e
Intertidal Marsh Snails:
None Assiminea californica f Assiminea californica f None
Ovatella myosotis f
Ovatella myosotis f
Table 5. Patterns of Invasion Along the Salinity
Gradient - continued
Marine Mesohaline Oligohaline Fresh
NO INVADERS (WITH POTENTIAL FOR INSERTION INVADERS)
Gnorimosphaeromid Isopods:
Gnorimosphaeroma Gnorimosphaeroma None g Gnorimosphaeroma
oregonense oregonense insulare
Anisogammarid Amphipods:
Anisogammarus Anisogammarus None Anisogammarus
confervicolus confervicolus ramellus
a Crepidula nummaria and perforans may not be separate species.
b Regularly present but not reproducing.
c Cryptogenic.
d Formerly present, now extinct from the Estuary.
e Race (1982) demonstrated that competitive and other interactions sort these snails along a salinity/elevation gradient by mid-summer
f Berman & Carlton (1991) found little competitive interaction between these snails in Oregon marshes.
g The introduced Japanese estuarine
isopod, Gnorimosphaeroma rayi, is reported from Tomales
Bay (north of San Francisco), but is not yet known from San Francisco
Bay.
Other notable "upstream invaders" include the Atlantic barnacle Balanus improvisus, the most freshwater-tolerant barnacle in the world, whose range in the Estuary extends far upstream of the Bay's native barnacles; two Japanese gobies, Acanthogobius flavimanus and Tridentiger bifasciatus, which have become abundant in the upper Bay and Delta upstream of the native estuarine gobies, and have been transported south from the Delta in freshwater irrigation canals; the Australian serpulid worm Ficopomatus enigmaticus, the only tubeworm found in the brackish parts of the Bay and extending into quite low salinity water; and the shipworm Teredo navalis, which when it was introduced in the 1910s invaded upstream portions of the Estuary not previously entered by the Bay's existing native and exotic shipworms, and caused enormous damage to wooden maritime structures. In some cases, such as that of the freshwater entoproct Urnatella gracilis, the introduced species may live in such low salinity water that it never overlaps in range with its closest native, and more marine, counterparts.
A second spatial pattern, rarer and perhaps more difficult for an exotic species to successfully achieve, is that of an "insertion invader." An example was described by Oglesby (1965a), who pointed out that among nereid worms the introduced brackish water worm Nereis succinea occupies a geographic position in the estuary between the range of the native marine worm Nereis vexillosa and the range of the native freshwater worm Hediste limnicola. He argued that succinea, being more finely and narrowly adapted to the brackish water ecotone, may outcompete the more broadly adapted vexillosa and limnicola within this zone.
A third spatial pattern in the Estuary, uncommon and somewhat unexpected, is the "downstream invader" mode exhibited by the introduced amphipods in the tube-building genus Corophium. John Chapman has suggested that the native Corophium species may have been adapted to a specific flow and sedimentation regime, and that the dramatic human alteration of these parameters (due to hydraulic mining, soil-eroding agricultural practices, construction and roadbuilding, and the leveeing of channels on the one hand, and dam construction and water diversions on the other) that has occurred since the mid-19th century may have facilitated the invasion of the Estuary by at least three species of more marine-adapted Corophium.
Other spatial patterns of native-invader competition are also represented in the Estuary:
· In the case of the brackish-water, fouling-inhabiting Korean shrimp Palaemon macrodactylus, there are no apparent native counterparts, upstream or downstream, and thus no obvious competitors.
· The native marsh snail Assiminea californica and the Atlantic marsh snail Ovatella myosotis, occur in the same marsh areas and appear to be counterparts, but studies in Oregon on these two snails found little evidence of any competitive interactions between them (Berman & Carlton, 1991; while in the Estuary these snails apparently co-occur over their whole elevational range, in Oregon they co-occur only in the lower part of Ovatella's elevational range).
· The introduced Atlantic snail Ilyanassa obsoleta
now occupies the Bay mudflat areas formerly occupied by the native
snail Cerithidea californica. Each spring the two populations
of these snails collide, and by mid-summer the exotic Ilyanassa
restricts the native Cerithidea to high-marsh salt pannes
(an environment too high in salinity for Ilyanassa and
thus providing a habitat refuge for Cerithidea) through
egg-string predation and direct competitive interference (Race,
1982).
Along with competition, other interactions between native and
introduced species may also occur, potentially leading to changes
in community or habitat structure, or to the replacement, displacement
or local elimination of the native taxa. Examples are reviewed
in the sections below.
At the end of the 20th century, exotic species play a major
role in structuring or altering aquatic environments.
We have considered above the evidence for dramatic alterations in the food webs and energy flow in the San Francisco Bay and Delta ecosystem due to individual species and species guilds. With such evidence in hand, it is easy to overlook the fact that for many abundant species in the Bay and Delta, little or nothing is known about their ecological rolesótrophic or otherwiseóin the ecosystem. For such common introduced species as the marsh plants brass buttons (Cotula coronopifolia) and peppergrass (Lepidium latifolium), many of the freshwater fish, the mat-forming mussel Musculista, the bed-forming mussel Mytilus galloprovincialis, the soft-shell clam Mya, the littleneck clam Venerupis, and many of the introduced polychaetes, crustaceans, hydroids, sea anemones, tunicates and bryozoans, little or nothing is known of their competitive and potentially regulatory interactions with native species and with each other.
Certain observations and experimental data are available, however,
both in the Bay and elsewhere, to gain some insight into the additional
extensive community-level modifications that have taken or may
be taking place through competitive and other interactions of
nonindigenous species.
In subtidal and intertidal soft-bottom communities, dense beds (> 2,000 individuals/m2) of Potamocorbula amurensis appear to have mechanisms that prevent the successful establishment of other organisms, native or introduced. These mechanisms may include predation on the larvae of these organisms, more efficient filter feeding (Nichols et al. 1990) and direct spatial competition.
In the only experimental studies done to date in San Francisco
Bay on the interactions between benthic native and introduced
invertebrates, Race (1982) has shown experimentally that the introduced
mudsnail Ilyanassa obsoleta restricts the native mudsnail
Cerithidea californica to upper intertidal, high salinity
habitat through egg predation and direct interference.
Competitive interactions in Bay and Delta fouling communities
can be inferred from studies of the same or similar species in
other systems; the absence of such work in San Francisco Bay is
notable. Working in nearby Bodega Harbor, Standing (1976) experimentally
demonstrated that the hydroid Obelia "dichotoma",
also present in San Francisco Bay, decreases the settlement rate
of barnacles but increases the settlement rate of ascidians. By
interfering with barnacle recruitment, ascidian settlement is
enhanced, and dense aggregations of ascidians support a diverse
associated community. Working in North Carolina, Sutherland (1977,
1978) found that the bryozoan Schizoporella sp. (identified
as S. unicornis but perhaps not that species) and
the seasquirt Styela plicata (introduced from the Pacific
to the Atlantic, although this was not known to Sutherland) have
a stabilizing role in community structure: when dense, these two
dominant species exclude other species from invading, resulting
in patches with fewer species and less change over time. On a
greater time scale, however, Styela destabilizes the fouling
community through annual "sloughing off" of the large
summer individuals, taking the associated fouling community with
it. Both Styela species and Schizoporella unicornis
are common in San Francisco Bay. Sutherland's observations may
further aid in explaining the apparent replacement of mussel beds
(Mytilus edulis) in parts of New England by the
introduced Asian seasquirt Styela clava, a species
common throughout the Bay's fouling communities.
Competitive interactions in Bay marsh systems are poorly known.
At local sites, the introduced peppergrass Lepidium latifolium
may compete with native pickleweed Salicornia virginica,
and may also play a role in displacing rare native marsh plants
such as Lillaeopsis masoni (Trumbo, 1994). At a
site in San Pablo Bay, the introduced chenopod Salsola
soda also appears to be competing with Salicornia.
Despite existing populations of the native Spartina foliosa,
three species of the cordgrass Spartina have been intentionally
planted in San Francisco Bay salt marshes (Spicher and Josselyn,
1985). One of these, Spartina alterniflora, which
has converted 100s of acres of mudflats in Willapa Bay, Washington
into cordgrass islands, has become abundant in parts of San Francisco
Bay and may be competing with the native cordgrass. Spartina
alterniflora has broad potential for ecosystem alteration:
its larger and more rigid stems, greater stem density, and higher
root densities may substantially alter habitat for native wetland
animals and infauna. Dense stands of S. alterniflora
may change sediment dynamics, reduce benthic algal production
because of lower light levels below the cordgrass canopy, and
reduce shorebird feeding habitat through colonization of mudflats
(Callaway, 1990; Callaway & Josselyn, 1992). In British estuaries,
the invasion of mudflats by Spartina anglica has
produced adverse effects on shorebirds (Goss-Custard & Moser,
1990).
The Delta today hosts large populations of exotic species: the Asian clam Corbicula can form dense beds many meters in extent, the eastern American worm Manayunkia can occur in sediments in densities of 2,000 to 5,000/m2, introduced crayfish and fish are frequently the only crayfish or fish species encountered, and meadows of floating or rooted aquatic plants may dominate areas of formerly open water.
The introduced crayfish Orconectes, Procambarus and Pacifastacus, when dense, are capable of extensive local habitat alteration through burrowing activities and presumably play an important role in regulating their prey plant and animal populations. Some introduced bottom-feeding fish are similarly capable of structurally altering habitats; carp, for example, dig up the bottom, destroying rooted vegetation and rendering potentially productive areas unsuitable for use as spawning or nursery areas by other fish species.
Several introduced freshwater plants can become locally abundant.
These include the aquarium plant Egeria (=Elodea),
which has been responsible for clogging channels and boat berths,
and the water hyacinth (Eichhornia crassipes), which manifests
itself as a nuisance plant by blocking waterways, interfering
with vessel operations, and fouling pumps. Both of these plants
alter conditions of shading and cover and, in the case of water
hyacinth, may become dense enough in places to interfere with
fish migration (CDBW, 1994).
Some evidence exists that bio-erosion of the Bay and Delta land margins may be occurring at the "hands" of burrowers and borers among the exotic fauna. The introduced crayfish Procambarus clarkii excavates burrows 5 cm in diameter and as much as 100 cm deep in Delta levees and banks. Muskrats similarly create extensive burrow systems in the Delta. The recently introduced Chinese mitten crab Eriocheir is known to form extensive excavations along river banks.
However, the most numerous bio-eroder around the Bay margins is the New Zealand boring isopod Sphaeroma quoyanum. Carlton (1979b) has described portions of certain eastern and northern bay shores, characterized by many linear meters of fringing mud banks riddled with the one-half centimeter holes of this isopod, as "sphaeroma topography," a phenomenon illustrated by Barrows (1919) and Hannon (1976). Higgins (1956) concluded that this isopod plays "a major, if not the chief, role in erosion" of intertidal sandstone and tuff terraces along the south shore of San Pablo Bay, due to boring activity that weakens the rock and facilitates its removal by wave action. Hannon (1976) reported one estimate that Sphaeroma could "remove up to 10 meters of dike in one year", a number that appears excessive. Nevertheless, Sphaeroma has been burrowing into bay shores for over a century, and it would not be surprising to learn that the land/water margin has retreated at certain sites by a distance of at least several meters due to this isopod's activities.
Exceedingly valuable would be observational and experimental studies
in the Estuary that focus on the erosion rates of crayfish, muskrats,
isopods and, if they become abundant along channel, stream and
river banks, Chinese mitten crabs.
No estuarine or aquatic introduction in the San Francisco Bay
region has solely or indisputably led to the extinction of a native
species. Short of this, however, invasions in the Bay have led
to the complete habitat or regional extirpation of species, have
contributed to one global extinction of a California freshwater
fish, and are now strongly contributing to the further demise
of endangered marsh birds and mammals.
Introduced freshwater and anadromous fish have been directly implicated
in the regional reduction and extinction, and the global extinction,
of four native California fish. The introduced striped bass, largemouth
and smallmouth bass, bluegill and green sunfish, through predation
or through competition for food and breeding sites, have all been
associated with the regional elimination of the native Sacramento
perch from the Delta. The introduced inland silverside may be
a significant predator on the larvae and eggs of the native Delta
smelt. Expansion of the introduced smallmouth bass has been associated
with a decline in the native hardhead. Predation by striped bass,
largemouth and smallmouth bass may have been a major factor in
the global extinction of the thicktail chub.
The California clapper rail may serve as an example of how populations of an already endangered species may be further threatened by biological invasions. Despite the interest in clapper rails in San Francisco Bay, however, there has been little quantitative investigation of the impact of introduced species, suggesting fruitful avenues for investigation.
Norway rats, established in many areas of California by the mid-1880s,
have long been recognized as significant predators on clapper
rail, starting with early observations such as the following (de
Groot, 1927):
"the clapper rail has no more deadly enemy than this sinister
fellow. No rail dares nest on a marsh area which has been dyked,
for as surely as she does this vicious enemy will track her down
and destroy the eggs. Many nests have I found bearing mute evidence
of the fact that some luckless rail had gambled her skill at nest-hiding
against the cunning of the Norway rat, only to have her home destroyed."
Predation on both rail eggs and rail chicks is considered to be high, with as many as a third of rail eggs said to be taken by rats (Josselyn, 1983; BODC, 1994). The cordgrass zones of salt marshes support the highest clapper rail densities by providing cover and/or isolation from rats, raptors and feral predators (Josselyn, 1983), and thus the expansion of these zones by the introduced Atlantic cordgrass Spartina alterniflora could benefit rails. Alternatively, competitive replacement of native cordgrass by S. alterniflora could reduce preferred cover for the rails.
Although present inland in California since the 1870s, the red fox has appeared on the margins of San Francisco Bay, adding another critical clapper rail predator to the ecosystem a century after the appearance of the Norway rat. In California the red fox has preyed on the eggs and sometimes the young or adults, and disrupted nests or colonies, of the clapper rail (as well as other birds, including least tern, snowy plover, Caspian tern, black-necked stilt and avocet) (Forester & Takekawa, 1991; Takekawa, 1993; BDOC, 1994).
Reduction in clapper rail populations by exotic species through
processes other than direct predation may also have occurred.
De Groot (1927) reported, under the heading of "the invisible
foe," the following concerning the relationship of adult
rails to the Atlantic ribbed marsh mussel Arcuatula demissa:
"This apparently harmless little mussel has been another
of the rail's most relentless enemies, and the number of rail
deaths attributable to its activities is incredible...Countless
millions of these small mussels cover the edges and sometimes
the entire bottoms of the gutters and creeks of the west Bay marshes.
Up under the banks, where the rail so commonly feed and hide when
the tide is out, these death traps are found in great numbers...Along
comes a rail gingerly pecking into the soft mud (and it) rams
(its) beak into the open mussel and in an instant the trap is
sprung and the rail is helplessly and hopelessly trapped... shaking
and scraping and pulling are all in vain...(and) the poor rail
eventually (dies) by starvation"
De Groot further believed that "at least seventy-five percent"
of the adult rails of the Redwood marsh area in the South Bay
had lost toes by entrapment in mussel shells. He argued that this
led to the loss of juvenile birds as well:
"But while the adult rail generally escapes with merely the
loss of a toe or two, young birds must meet death frequently...(there
is) some basis for stating that probably one or two chicks in
every brood, if not more, meet an untimely end in this manner..."
More recent observers note that clapper rails in the Bay are frequently missing one or more toes (Moffitt, 1941; Josselyn, 1983; Takekawa, 1993) and Josselyn (1983, p.69) includes a photograph of an adult clapper rail missing one toe and with an Arcuatula clamped to another.
Unfortunately, accurate quantification of rail:mussel interactions
is lacking, and thus the impact (implied by de Groot to be approaching
one-third brood mortality at the valves of the mussel) on clapper
rails remains unknown. That the rail/mussel interaction may not
be all one sided, however, is suggested by Moffitt's (1941) study
of rail feeding, wherein he found in a sample of 18 birds that
66 percent of the animal food of the rail (and 57 percent of the
total food) consisted of Arcuatula.
Around the Bay and Delta, reduction and elimination of populations of other native species have occurred or appear to be in progress as the result of interactions with introduced species. Unfortunately, as with impacts on the clapper rail, and with the sole exception of impacts on native snails, no quantified data appear to be available. It has thus been suggested or observed that:
· the introduced Atlantic mudsnail Ilyanassa has displaced from mudflats to saltmarsh pannes and reduced the population of the native mudsnail Cerithidea;
· introduced green sunfish, bluegill, largemouth bass and the introduced American bullfrog may have contributed to the decline of native red-legged and yellow-legged frogs in the Bay and Delta region, largely through predation;
· introduced red fox, through predation, reduce or limit the recovery of populations of the endangered salt-marsh harvest mouse;
· introduced crayfish have displaced some native crayfish species and threaten others;
· introduced peppergrass (Lepidium latifolium) may displace rare native marsh plants, such as Lillaeopsis masoni.
The economic impacts of introduced marine, estuarine and aquatic organisms have been little studied and rarely quantified. It is clear, however, that these impacts have been substantial in the San Francisco Estuary.
These impacts are of several interrelated and intergraded types.
Positive impacts have included the value of food resources and
recreational (sportfishing) resources provided by some introductions
of fish and shellfish; the biological control of nuisance insect
populations (e. g. by mosquitofish); and fish and wildlife enhancements
such as the provision of food, habitat or other resources for
valued species (Table 6). Major negative impacts have included
the fouling and blocking of waterways and water delivery systems;
damage to or impairment of maritime structures and vessels (e.
g. damage to wharves, docks, ferry slips and ships' hulls by marine
wood-boring organisms; increased fuel and maintenance requirements
resulting from hull fouling); disruption or impairment of vital
services; damage to populations of economically important fish
and wildlife species; the costs (both direct and indirect) of
control efforts; and the inability, in the face of continuous
new introductions, to adequately manage the Estuary's ecosystem,
resulting in restrictions on activities in and near the Estuary
(Table 7). We discuss certain of these impacts below.
Skinner (1962) and Smith & Kato (1979) review the history of the fisheries in the Estuary. Although the introduced striped bass, American shad, white catfish, bullfrog, signal crayfish (Pacifastacus leniusculus) and soft-shell clam (Mya arenaria) all supported commercial fisheries in the Estuary in the past, only the crayfish is still commercially harvested today. These species and others, including many warm-water gamefish introduced to the Delta, continue to provide sport fisheries.
Striped bass and shad supported large commercial fisheries during the late 19th and first half of the 20th century. Striped bass were introduced in 1879 and sold in San Francisco markets by 1889. The annual catch topped 500 tons by 1899, peaked at 1,000 toms in 1903, and generally stayed over 500 tons until 1918. The commercial fishery then declined and was closed in 1935 to avoid competition with sport fishing (Skinner, 1962; Smith & Kato, 1979).
Shad were introduced in 1871, commercially harvested by 1874,
and glutting the market by 1880 (Skinner, 1962). From 1900 to
1945 the Bay Area catch was often over 500 tons, and peaked at
over 2,800 tons in 1917 (Skinner, 1962; Herbold & Moyle, 1989).
The fish were mainly sold fresh until 1912, and thereafter salted
and export to China, with the roe salted and canned; the size
of the fishery was said to be limited by demand rather than by
the abundance of shad. After 1945 the catch averaged around 300
tons until the fishery was eliminated in 1957 by a ban on gill-netting
inside the Golden Gate (Shebley, 1917; Skinner, 1962; Smith &
Kato, 1979).
Table 6. Positive Economic Impacts of Marine,
Estuarine and Aquatic Organisms Introduced into the San Francisco
Estuary
Details and references are provided in the species
descriptions in Chapter 3.
ORGANISMS CAUGHT FOR FOOD, FUR OR SPORT
· Striped bass, American shad and catfish supported commercial fisheries in the Estuary that were sometimes substantial, until commercial fishing for these species in the Estuary was banned.
· The above species, plus black bass, crappie, sunfish and carp support recreational fisheries in the Estuary.
· Crayfish are taken from the Delta both commercially and recreationally.
· The bullfrog Rana catesbeiana has been both raised in ponds and harvested from public waters in California.
· The Asian littleneck clam Venerupis philippinarum and sometimes the Atlantic soft-shell clam Mya arenaria are taken recreationally. Venerupis is harvested commercially in the Pacific northwest and sold in Bay Area markets as "Manila clams." A few other introduced molluscs are sometimes recreationally harvested from the Bay.
· The Asian freshwater clam Corbicula fluminea is sometimes taken recreationally from the Delta. Corbicula are harvested commercially from Lake Isabella in the southern end of the Delta's watershed.
· The Asian freshwater snail Cipangopaludina was imported and sold in Asian markets in the late 19th century, and was reportedly planted in the Bay Area and the Central Valley "to supply the markets of San Francisco Bay."
· Watercress is an edible green which no doubt is sometimes harvested recreationally.
· Muskrat are trapped for their fur.
BAIT
· The golden shiner and fathead minnow are commercially raised as legally-designated freshwater bait fish in California.
· The yellowfin goby is commercially and recreationally harvested for use as bait, primarily for the introduced striped bass.
· The freshwater Asian clam Corbicula is harvested commercially and recreationally for bait.
· Introduced crayfish and bullfrog are caught recreationally for use as freshwater bait.
· Various other introduced fish (e. g. inland
silverside) and invertebrates (e. g. the mussel Mytilus galloprovincialis)
are sometimes used for bait.
BIOCONTROL
· The mosquitofish Gambusia affinis contributes
to the control of mosquitoes. However, introductions of other
species for biocontrol purposes (e. g. blue catfish to control
the introduced clam Corbicula, South American Neochetina
weevils to control water hyacinth) appear to have had no significant
control effect, and have sometimes harmed desirable species (e.
g. inland silverside Menidia beryllina).
EROSION CONTROL
· According to one study, the Atlantic cordgrass
Spartina alterniflora may be reducing erosion at San Bruno
Slough.
Table 6. Positive Economic Impacts - continued
ENHANCEMENT OF ECONOMICALLY IMPORTANT FISH AND WILDLIFE
· The South African brackish-marsh plant brass buttons provides food for waterfowl and refuge; marshes are sometimes managed to encourage its growth.
· The Atlantic cordgrass Spartina alterniflora might provide much-needed cover for the endangered California clapper rail.
· Threadfin shad were introduced to provide forage for sport fish, although there is doubt about how useful they are as forage; to the extent that they do provide forage they may have simply replaced native species; and some researchers believe that they may in fact compete with young sport fish and reduce the populations of sport fish.
· Many pelagic and benthic marine invertebrates
form part of the trophic webs that support recreationally and
commercially important fish, but may have simply replaced native
invertebrates in this role.
White catfish were introduced in 1874. In 1875 the California Fish Commission predicted that they would support a commercial fishery by the following year, and in 1877 reported that they constituted an "important addition to the fish food supply of the city of Sacramento," further described in 1879 as "an immense supply of food" (Smith, 1896). By 1900 catfish were being exported to Mississippi. The Bay Area's reported annual catch of catfish ranged between 100 and 500 tons from 1905 to 1951 (Skinner, 1962), but the fishery was closed in 1953 due to declining numbers of fish(Miller, 1966a; Borgeson & McCammon, 1967).
The soft-shell clam was first collected in the Bay in 1874 and by the 1880s was the most common clam in Bay Area markets (Stearns, 1881), and public and private soft-shell clam beds were established and managed throughout the Bay (Bonnot, 1932). The annual catch in the Bay Area (including bays north to Bodega) was 500 to 900 tons in 1889-1899, 50-150 tons in 1917-1935, and then declined until the fishery closed in 1948, for reasons that are now unclear but could involve a decline in the resource or market competition from other clams (Skinner, 1962; Herbold et al., 1992). Several workers have suggested that the soft-shell clams' early abundance in San Francisco Bay was due to replacement of populations of the native bent-nose clam Macoma nasuta.
It is unclear when signal crayfish were introduced to California, but commercial harvest began in the Delta in 1970 to supply the Swedish market (after the native Swedish crayfish was decimated by an introduced North American crayfish disease). Initial landings of 50 tons rose to over 250 tons from 1975 to the 1980s (Osborne, 1977; Herbold & Moyle, 1989). The 1976 catch sold for a little over $300,000 (Osborne, 1978).
Striped bass has been the economically most important sport fish in the Estuary, accounting for a substantial transfer of funds, variously estimated, from those who do the fishing to those who help them fish. Skinner (1962, p. 172) reported that striped bass anglers were spending about $18 million per year on the sport. McGinnis (1984) reported that anglers took about 1 million striped bass in 1980, spending about $7 million in the process. Herbold et al. (1992) reported that the industries surrounding striped bass fishing (involving boats, marinas, and fishing equipment and supplies) were estimated to inject $45 million into local
economies.
Several small fish have been introduced to California in part
to provide forage for larger sport fish, including the threadfin
shad. However, there has been considerable disagreement over the
value of the threadfin as forage (ranging, according to different
authors, from "major" and "important" to "minor"
and "inadequate"), and its overall impact on sport fish
(involving competition with young sport fish for food), as reviewed
in Chapter 3.
Mare Island, in the upper part of San Francisco Bay, was chosen as the site for a naval base partly in order to get upstream of the Bay's marine wood-boring organisms. However, the introduction of the shipworm Teredo navalis, which tolerated much fresher water than did the Bay's existing wood borers, led to the destruction of some fifty major wharves, ferry slips and other structures in the northern part of the Estuary between 1919 and 1921, including several at Mare Island (Figure 8).
Neily (1927) reported the damage to amount to $25 million, which, escalated to current (1992) dollars (based on the Engineering News Record: General Construction Cost Index; US Commerce Dept., 1975, 1984, 1993) is $616 million dollars. Although this figure does not include collateral damage (such as loaded freight cars that fell into the Bay when a railroad dock collapsed), disrupted service and lost business, or the subsequent costs of constructing, treating and maintaining structures to be resistant to Teredo, nor does it include damage from Teredo since 1921 or in other parts of the Bay, it does provide some quantification of the scale of potential economic impact from a single introduced organism.
Other introduced wood-borers in the Bay are the shipworm Lyrodus
pedicellatus,, and the isopods Limnoria tripunctata
and L. quadripunctata, and Chelura terebrans.
Although modern, chemically-treated pilings, marine timbers and
marine wood products are considerably more resistant to borer
infestations than untreated wood, borer damage continues to occur
to the Bay's wooden pilings, docks and boat hulls. However, no
current estimates of this damage are available.
Hull fouling and other ship fouling have a large but generally
little-recognized economic impact. For example, Gordon & Mawatari
(1992) report estimates that a coating of slime 1 mm thick on
an otherwise clean hull can increase skin friction up to 80 percent
and reduce speed up to 15 percent, an estimate
Figure 8. Some Examples of Damage Caused by the
Wood-boring Shipworm Teredo navalis in the San
Francisco Estuary
From Neily, 1927.
(1) Failure of dock at Oleum, Contra Costa County,
Oct. 8, 1919, dumping several loaded freight cars into San Francisco
Bay.
(2) Collapse of the South Vallejo Ferry Slip, Solano
County, Nov. 4, 1920.
(3) Collapse of the Benicia Municipal Wharf and House, Oct. 7, 1920.
generally borne out by towing tests (WHOI, 1952). Ross & Emerson (1974) calculated that "a luxuriant growth of barnacles on a one-square-foot area of a ship may weigh as much as six pounds. On a large ship, the barnacles and other fouling organisms can add as much as three hundred tons to a ship's weight...a heavily fouled ship may need as much as 50 percent more fuel to move the same distance." In 1928 it was reported that U. S. shipping interests spent $100 million annually dealing with fouling (WHOI, 1952, citing Visscher, 1928). In the 1940s, the British Admiralty estimated that hull fouling on naval vessels increased fuel consumption by 35% to 50% after six months in temperate waters or after three months in tropical waters (WHOI, 1952). More recently, Haderlie (1984) reported that "all classes of [U. S.] naval ships show a ten percent average yearly increase in fuel consumption between dry dockings, and...most or all of this is due to increased drag caused by hull and propeller fouling." He further reported that in 1975 the U. S. Navy spent $15 million a year applying antifouling coatings to its vessels, but that despite this "the increased drag from hull fouling was adding over $150 million to the navy's annual fuel bill."
Hull fouling can thus result in a significant loss of maximum speed and maneuverability, increased fuel consumption and decreased range, as well as necessitating increased maintenance and more frequent drydockingsóissues of concern to all vessels but especially to military vessels (Haderlie, 1984). WHOI (1952) and Haderlie (1984) reported other impacts of ship fouling, including blocked fire mains; restricted or blocked flow to the main condensers serving the ship's engines, preventing the development of full power; other fouled seawater pipe systems, sometimes requiring the complete dismantling of these systems; fouled propellers causing increased vibration on board ship and loss of power; increased hull corrosion; fouled sonar domes causing degradation of performance due to reduced sound transmission and reception, increased self-noise due to turbulence, and interference with mechanical operation; and increased self-noise of the ship hull, a problem for military ships seeking to evade detection by enemy sonar.
Such considerations have lead to the development and widespread use of anti-fouling compounds containing tributyltin (TBT), copper, mercury, arsenic and other materials which are toxic both to fouling and to nontarget marine organisms, and to those working with these compounds. The cleaning and maintenance of TBT-coated hulls has contributed to the creation of toxic "hot spots" in the Estuary.
Though ships may be fouled by both native and non-native organisms,
virtually all of the common fouling organisms in San Francisco
Bay are introduced (e. g. Graham & Gay, 1945; Banta, 1963;
ANC & JTC, pers. obs.). Thus fouling impacts for vessels spending
much of their time in San Francisco Bay are largely due to introduced
species.
The fouling of Delta waterways by water hyacinth became serious
enough by the early 1980s to block ferry boats from reaching Bacon
Island and prevent the island's produce reaching the market. In
1982 the California Legislature passed a bill ordering the control
of water hyacinth in the Delta. Control efforts included setting
up barriers to keep masses of hyacinth out of navigation channels,
spraying
Table 7. Negative Economic Impacts of Introduced
Marine, Estuarine and Aquatic Organisms
A. Examples in the San Francisco Estuary
Details and references are provided in the species
descriptions in Chapter 3.
WATERWAY FOULING
| · Water hyacinth Eichhornia crassipes
· European milfoil Myriophyllum spicatum · Elodea Egeria densa | · Navigational and recreational impacts include blocking passage through navigable waterways and access to marinas and berths, and fouling propellers and the water intakes of boat engines; impacts have been serious eno
ugh to shut down marinas and bar ferry boats from their routes.
· Interference with salmon migration. · Costs of herbicide applications (including environmental and occupational health impacts). · Costs of biocontrol efforts. · Costs of mechanical removal and disposal. |
FOULING OF VESSELS AND MARITIME STRUCTURES
| · Many kinds of plants and animals, including seaweeds, sponges, hydroids, tubeworms, mussels, barnacles, bryozoans and sea squirts | · Increased frictional resistance of ship and boat hulls, resulting in slower speeds, increased transit times, increased fuel costs, reduced maneuverability, and reduced effectiveness of military vessels.
· Cost of anti-fouling coatings. · Costs of pollution from the use of anti-fouling compounds formulated with tributyltin, copper, mercury, creosote or other toxic materials. · Occupational health costs of manufacturing, applying and maintaining coatings of anti-fouling compounds formulated from toxic materials. · Other increased maintenance costs, including the cost of time spent in drydock rather than in service. |
WOOD BORING
|
· Shipworms Teredo navalis and Lyrodus pedicellatus · Isopods Limnoria spp. and Chelura terebrans | · Damage to wooden maritime structures and vessels.
· Disruption of service. · Increased maintenance costs. · Increased construction costs. · Impacts from the use of toxic anti-fouling compounds, as noted above. | |
BURROWING
| · Muskrat
· Crayfish Orconectes and Procambarus · Isopod Sphaeroma · Chinese mitten crab | · Damage to levees, the walls of ditches, stream banks and shorelines. |
| · Isopod Sphaeroma | · Damage to styrofoam flotation of marina docks. |
Table 7. Negative Economic Impacts - continued
FOULING OF WATER SYSTEMS
| · Corbicula , and to a minor degree, Urnatella and Cordylophora | · Increased sedimentation in canals reducing flow rates.
· Increased maintenance costs. |
| · Water hyacinth | · Fouled irrigation pumps and fish screens. |
PREDATION ON AND COMPETITION WITH ECONOMICALLY IMPORTANT SPECIES
| · Many species of fish
· Crayfish Orconectes virilis and Pacifastacus leniusculus · Bullfrog Rana catesbeiana | · Reduction of populations of commercial and sport fish.
· Elimination of the Sacramento perch Archoplites interruptus, a sport fish, from its native waters. · Reduction in populations of certain native fish, crayfish and frogs contributing to their listing or potential listing as threatened or endangered species, resulting in: - interference with water diversions, including restrictions on the location, timing and volume of diversions and on the construction of new diversion facilities; - interference with other construction and development projects, both inside and outside the Estuary, · Costs of control efforts, such as rotenone applications. · Kills of nontarget sport fish from rotenone applications. · Occupational and environmental health costs of rotenone use. |
| · Atlantic oyster drill Urosalpinx cinerea and odostomiid snail Boonea bisuturalis | · Predators or parasites on oysters, clams and mussels. |
PROMOTION OF UNDESIRABLE SPECIES
| · Parrot's feather Myriophyllum aquaticum | · Said to provide excellent mosquito habitat. |
CROP DAMAGE
| · Crayfish Orconectes virilis and Procambarus clarkii | · Eat rice shoots, as apparently does the recently introduced Chinese mitten crab Eriocheir sinensis in China. |
INTERFERENCE WITH WATER QUALITY MONITORING
|
· Mussel Mytilus galloprovincialis | · Fifteen years of estuarine water quality monitoring, based on comparing contaminant levels in the same species of mussel in different bays, may have been rendered questionable by the introduction of this second and virtually indistinguishable species of mussel which may take up and metabolize contaminants at a different rate. | |
Table 7. Negative Economic Impacts - continued
ECOSYSTEM INSTABILITY/MANAGEMENT UNCERTAINTY
|
· Continuous high rate of introductions | · New species continually being introduced into the Estuary's biota resulting in unmanageable fluctuations in populations of important species, in turn resulting in added restrictions on many activities (including wat er diversions, wastewater discharges, dredging, levee maintenance, construction) in and near the Estuary. | |
B. Some Examples from Elsewhere
FOULING
|
· Zebra mussel Dreissena polymorpha | · The European zebra mussel was introduced to the Great Lakes in ballast water in 1986 and rapidly spread to 14 states and 3 Canadian provinces.
- It has seriously fouled and in some cases caused the complete blockage of the water intakes for municipal water systems, industrial process water systems, and cooling water systems for power plants. It has incurred costs through the disrupt ion of services; increased monitoring and maintenance requirements; changes in operations; the retrofitting of existing facilities and added costs in the construction of new facilities to make them less vulnerable to mussel fouling; the construction of re dundant facilities to prevent service disruptions; the increased use of chlorine (with attendant occupational, public and environmental health costs). - It has interfered with commerce and recreation by fouling navigational buoys, maritime structures and vessels, with attendant costs. - It has fouled recreational beaches. In the past year, live zebra mussels have been found attached to boats entering California from the eastern states. | |
PREDATION ON ECONOMICALLY IMPORTANT SPECIES
|
· Green crab Carcinus maenas | · This European crab was introduced to the eastern United States in ship fouling and destroyed commercially valuable soft-shell clam (Mya arenaria) beds in New England and Maine in the 1950s. Control efforts in
cluded fencing, trapping and poisoning.
The green crab became established in San Francisco Bay in the late 1980s. | |
Table 7. Negative Economic Impacts - continued
| · Chinese mitten crab Eriocheir sinensis | · Introduced in ballast water, this catadromous, burrowing crab became phenomenally abundant in the rivers and upper estuaries of Germany in the 1930s, causing damage to trap and net fisheries and to river banks, leadi
ng to a government-sponsored control program that, at its peak, trapped and destroyed tens of millions of crabs per year.
The mitten crab became established in San Francisco Bay in the 1990s. |
| · Mnemiopsis leidyi | · Discovered to the Black and Azov seas in the early 1980s, this northwestern Atlantic ctenophore or 'comb jelly' became phenomenally abundant by 1988, decimating the zooplankton and virtually destroying the region's a nchovy and sprat fisheries. |
DISEASE
| · 'red tide'-forming dinoflagellates and other bloom-forming plankton | · Blooms of dinoflagellates that produce sometimes-lethal paralytic shellfish poisons (PSP) have resulted from introductions of these plankton to Australia and probably other parts of the world. |
| · Oriental lung fluke | · In China, the mitten crab Eriocheir sinensis is the second intermediate host of this debilitating human parasite; human hosts are infected by eating raw or inadequately cooked, infected crabs. With the mitten crab now established in the Bay Area, and snails available that are capable of serving as first intermediate hosts, the lung fluke could become established in California. |
| · cholera pathogen Vibrio cholerae | · In 1991 during the South American cholera epidemic, ships' ballast water from that continent arriving in U. S. ports in the Gulf of Mexico frequently carried the cholera pathogen, which was also found in fish and oys ters in those ports. |
herbicides, and releasing biocontrol agents, at a cost that reached
$400,000/year (L. Thomas, pers. comm., 1994), though it only partly
alleviated the problems.
The Asian freshwater clam Corbicula fluminea plugged condenser
tubes at the federal water project's pumping plant in the South
Delta, colonized the bed of the project's Delta-Mendota Canal
(trapping sediment and forming bars that reduced delivery capacity,
requiring the dewatering of the canal and the dredging of over
50,000 cubic yards of clam-bearing material), and in southern
California plugged underground pipes, turnout valves, and irrigation
sprinklers (Ingram, 1959; Hanna, 1966; Eng, 1979).
As discussed earlier in this chapter under "Bio-eroders,"
several introduced species burrow in and damage both natural banks
and man-made embankments, including muskrat, two species of crayfish
and the Chinese mitten crab in fresh and brackish areas, and the
isopod Sphaeroma quoyanum in the more saline waters of
the Bay. In addition, we have found the styrofoam blocks that
provide flotation for marina docks frequently riddled with Sphaeroma
burrows, and though no quantitative data are available, it seems
that this must substantially shorten their lifetime.
Several intentional introductions may have had the "side effect" of reducing populations of other economically important species. Economically important species in this context include both species that are hunted or fished, and species that, because of their declining populations, become listed or become candidates for listing under the state or federal endangered species act (or otherwise become species of special concern), triggering limitations on economic activities. Examples of such "side effects" suggested by various researchers include the following.
· In the 19th century, the destruction of water celery, a common duck food, by introduced carp might have reduced populations of canvasback and other ducks (Smith, 1896, citing Jordan & Gilbert, 1894).
· Shebley (1917) reported carp to be the principal cause of destruction of the Sacramento perch, by eating its eggs and digging up its nests. Moyle (pers. comm.) has suggested that predation by striped bass and black bass may have been the major cause of the elimination of Sacramento perch from the Delta. McGinnis (1984) suggests that competition with introduced sunfish was the cause.
· Several workers have suggested that threadfin shad compete with the fry of gamefish, including black bass (McConnell & Gerdes, 1961; Von Geldern & Mitchil, 1975), crappie (McConnell & Gerdes (1961) and striped bass (McGinnis, 1984).
· Inland silverside may compete with striped bass (McGinnis, 1984) and prey on the eggs and fry of the endangered Delta smelt (BDOC, 1994; Moyle, pers. comm.).
· The Shasta crayfish Pacifastacus fortis was proposed
for listing, in large part due to competition from the introduced
crayfish Orconectes virilis and Pacifastacus leniusculus
(Anon., 1987).
Substantial costs have been incurred through efforts to eradicate populations of two predaceous, nonindigenous fish present in the Delta watershedówhite bass and northern pikeóbefore they reach the Delta where it is feared they would reduce populations of endangered species and sport fish. For both fish, eradication efforts have centered around massive applications of the fish poison rotenone. The northern pike effort, for example, was preceded by three years of environmental review and litigation and a ban on fishing in the area (resulting in economic losses to the local economy), followed by the application of 12 semi-trailer loads of rotenone by 60 workers who were on site for over two weeks, with the cost of the on-site work alone totaling over a million dollars. The costs due to nontarget fish kills (which were substantial), other environmental health costs and occupational health costs are unknown.
The effort failed to eradicate northern pike from the watershed.
The greatest impact from introductions to the Estuary may be restrictions on the operation of the California water system. In recent years a combination of litigation, new legislation, and regulatory realignment has placed increasing environmental demands on the water agencies that store and divert water from the Estuary's watershed (DWR, 1993). Specifically, the agencies' ability to withdraw water increasingly depends on whether they can restore and sustain healthy populations of anadromous and native fish. This in turn will depend on the water agencies' and regulators' level of understanding of the ecosystem and their ability to figure out the necessary habitat conditions, including the amount and timing of instream flows needed, to maintain the fish.
However, the achievement of an adequate level of understanding
to reliably manage the Estuary is severely hampered by a rate
of introduction averaging (at least) one new species established
in the Estuary every 24 weeks. For example, the arrival, growth
and spread of the Asian clam Potamocorbula amurensis in
1986-87 appears to have fundamentally altered trophic relations
in the northern reach of the Estuary, and perhaps made models
and calculations based on pre-1987 data obsolete and irrelevant
(Nichols, 1985; Cohen, 1990; Alpine & Cloern, 1992; Cohen
& Carlton, 1995). A constantly changing species composition
may make the ecosystem even less stable, and major functional
shifts more common. Under such conditions, the reliable management
of the Estuary required of (and promised by) the water agencies
may be impossible. Since water from the Estuary's watershed supports
much of California's population, industry and agriculture, the
costs of failure could be substantial.
Some organisms introduced to the Estuary might possibly be harvested
and marketed. The European green crab Carcinus maenas,
the Chinese mitten crab Eriocheir sinensis, and the yellowfin
goby are commercially harvested for food in parts of their native
range (Cohen et al., 1995). The Asian sea squirt Styela clava
is harvested and eaten in Korea (Abbott & Newberry, 1980).
Water hyacinth leaves are sold as a vegetable in markets in the
Philippines (Ladines & Lontoc, 1983).
Hallegraeff and his coworkers have demonstrated that toxic dinoflagellates that produce paralytic shellfish poisons (PSP) were introduced to Australia from Japan in ballast water sediments (Hallegraeff et al., 1989; Hallegraeff & Bolch, 1991). The introduction of toxic dinoflagellates to the northeastern Pacific could have costly impacts. In the Philippines, three outbreaks in five years of a PSP-producing dinoflagellate previously unreported from the region cost the local mussel industry about $15 million, poisoned over a thousand people and killed at least thirty-four (Corrales & Gomez, 1989). In San Francisco Bay clams and mussels are commonly collected for food in a poorly monitored and largely unregulated sport fishery (Sutton, 1981). Although there is no commercial shellfishery in the Bay, dinoflagellates that arrive there in ballast water could be readily carried by coastal currents or by coastal transport of ballast water to commercial shellfish beds to the north.
In July, 1991 during the South American cholera epidemic, the
U. S. Food and Drug Administration discovered the causative organism
of cholera, Vibrio cholerae, in oysters and fish from Mobile
Bay, Alabama. Subsequently sampling of ballast water from nine
ships arriving in Alabama and Mississippi from South America revealed
Vibrio cholerae in one third of them (US Federal Register,
1991). It has been suggested that cholera could have initially
reached South America via ballast water (Ditchfield, 1993).
Many transport vectors releasing exotic species into the San Francisco
Estuary remain active, and new invasions are certain to occur.
These fall into eight categories discussed below, for each of
which we give examples of potential invaders. In addition, at
least 36 species of introduced aquatic plants, snails, fish, and
one turtle are established in regions adjacent to the greater
Bay-Delta system (Table 9), some of which will undoubtedly spread
into the Estuary.
Ships release in ballast water scores if not hundreds of new species on a monthly basis into the San Francisco Estuary (Table 10). That this highly successful vector remains active in the Estuary is indicated both by the number of new invasions now occurring (Table 1) and by the continual appearance but uncertain establishment of both small and large crustaceans in the Bay (Table 8).
Around the world there have been a number of important invasions,
linked to ballast water release, whose temperate climate biology
suggest that these species could become established in the San
Francisco Estuary. Ballast water from Japan could include the
larvae of the carnivorous North Pacific Sea Star Asterias amurensis
and several species of Japanese dinoflagellates not yet established
in San
Table 8. Recent Records of Nonindigenous Species
in the San Francisco Estuary whose Establishment is Uncertain
Native Date
Species Range Collected Comments (references)
| Invertebrates | |||
| Mollusca: Gastropoda | |||
| Prosobranchia | |||
| Littorina littorea | 14 collected at Alameda & Bay Farm islands in the northern South Bay in 1968-70, 6 collected at Selby on the east shore of San Pablo Bay in 1976-77 (Carlton, 1969, 1979a). ANC collected one specimen on the San Francisco shore in 1995. | ||
| Arthropoda: Crustacea | |||
| Isopoda | |||
| Ianiropsis serricaudis | Oakland Estuary (Carlton, 1979a). | ||
| Munna sp. A | (J. Chapman. pers. comm., 1995). | ||
| Sphaeroma sp. | (J. Chapman. pers. comm., 1995). | ||
| Amphipoda | |||
| Ampithoe sp. | (J. Chapman. pers. comm., 1995). | ||
| Calliopiella sp. | (J. Chapman. pers. comm., 1995). | ||
| Dulichia monocantha | (M. Kellogg, pers. comm., 1995). | ||
| Listriella goleta | (M. Kellogg, pers. comm., 1995). Collected in Los Angeles Harbor in the late 1980s. | ||
| Synchelidium miraculum | (G. Gillingham, M. Kellogg, H. Peterson, pers. comm., 1995). Collected in Los Angeles Harbor in the late 1980s. | ||
| Decapoda | |||
| Exopalaemon carinicauda | One specimen (L. Holthuis, pers. comm., 1993). | ||
| Exopalaemon sp. | One specimen, possibly E. carinicauda (K. Hieb, pers. comm., 1995). | ||
| unidentified Pandalid shrimp | One specimen (R. Van Syoc, pers. comm., 1995). | ||
| Vertebrates | |||
| Fish | |||
| Anguilla anguilla | European Eel, one specimen (Skinner, 1971). | ||
| Anguilla rostrata |
| American Eel, one specimen caught in each of 1964 & 1994. A fourth and unidentified eel, dated 1987, estimated 1 m length, is preserved at the Skinner Fish Facility in the Delta (Skinner, 1971; S. Walker, pers. comm., 1 994). | |
| Lepisosteus spatula | Alligator Gar, one specimen, 146 cm long (Raquel, 1992). |
Francisco Bay which, however, have become important invaders in southern Tasmania in a similar climatic regime (Carlton et al., 1995). Water from bays and estuaries of the American mid-Atlantic coast could include the Atlantic comb jelly Mnemiopsis leidyi, which has become a devastating zooplankton and larval fish predator in the Black and Azov Sea ecosystems (Shushkina & Musayeva, 1990; Mutlu et al., 1994) and the Japanese crab Hemigrapsus sanguineus, which was collected in 1988 in New Jersey (McDermott, 1991) and has now spread from North Carolina to Cape Cod (G. Ruiz, pers. comm., 1995; JTC, pers. obs.). The appearance of several Atlantic coast invertebrates in the San Francisco Estuary over the past 15 years (discussed under "Transport Mechanisms" in Chapter 5) suggests that the transport of additional organisms from the Atlantic is not unlikely. Ballast water from Europe could transport the freshwater-oligohaline gammarid amphipod Corophium curvispinum, a major fouling organism (Carlton et al., 1996).
These are clearly only a few out of scores of examples of known
invaders that have become established elsewhere and which, should
they hop on the ballast water conveyor belt, would be rapidly
transported to the Estuary. In addition, we expect there are many
organisms which have not invaded regions outside of their native
range, but which could yet become potent invaders (as was the
case with the Chinese clam, Potamocorbula amurensis, which
entered the Estuary in 1986).
Coastal ship traffic plays an unknown but potentially important role in transporting invasions that have established elsewhere on the Pacific coast to the Estuary. Examples include the transport of ballast water from the Columbia River (potentially transporting the Asian copepod Pseudodiaptomus inopinus, now well established there; Cordell et al., 1992) and from Pacific Northwest bays (which could include whole floating plants of the Japanese eelgrass Zostera japonica, which now occurs from Coos Bay to British Columbia). The arrival of the Atlantic oligochaete Lumbricillus lineatus in the Bay is also predictable, and should be specifically looked for in enriched sediments. Coates and Ellis (1980) have noted its establishment in pulp mill effluent sites in northern Vancouver Island, where it was introduced by international ship traffic.
Ballast water transport or ship fouling could play the central role in bringing to San Francisco Bay a number of species of Asian and Atlantic seasquirts that have become established in the harbors of southern California since the 1980s (G. Lambert, pers. comm., 1995). Indeed, ship fouling from these harbors is probably how the Japanese seasquirt Ciona savignyi arrived in San Francisco Bay, having previously become established in southern California. Coastal ship traffic from the south or the north may similarly have carried the Japanese seaweed Sargassum muticum as hull fouling into the Bay.
Similarly, coastal ship traffic may transport introduced organisms
now established in the San Francisco Estuary, including many known
in the northeastern Pacific only from the Estuary (Appendix 4),
to other sites along the coast. The Estuary has likely operated
in the past, and will likely continue to operate in the future,
as the port of entry for many invasions of the Pacific coast.
In Table 10 we list additional evidence for five additional vectors
for ongoing inoculations into the Estuary. These are (3) the live
bait and lobster industries (releasing not only the subject organisms
but the living seaweed used as packing material and numerous associated
invertebrates); (4) the herring-roe-on-kelp fishery (transporting
live Macrocystis kelp and associated invertebrates into
the Bay); (5) live bait releases of bait fish; (6) private party
releases of fish and shellfish; and (7) releases from home or
school aquaria. Each of these mechanisms is known to have resulted
in the at least temporary establishment of one or more non-native
species in the Estuary. There are few regulatory mechanisms in
place to manage the extent or minimize the impact of these vectors.
Table 10. Examples of Ongoing Inoculations of
Nonindigenous Species into the San Francisco Estuary
MECHANISM: Species Inoculated
BALLAST WATER:
Includes a wide variety of planktonic estuarine organisms
from many parts of the globe. Common types of organisms include
the adult or larvae of calanoid, cyclopoid and harpacticoid copepods,
spionid, polynoid and other polychaete worms, diatoms, barnacles,
bivalves, snails, flatworms, decapods, chaetognaths, tintinnids,
mysid shrimp, isopods, bryozoans, phoronid worms, amphipods, dinoflagellates,
hydroids and other taxa (Carlton & Geller, 1993).
BAIT WORM SHIPMENTS:
Includes a variety of organisms from the Maine coast,
including the baitworms Nereis virens and Glycera dibranchiata;
the seaweeds used for packing them, especially Ascophyllum
nodosum; and epiphytic seaweeds and small intertidal and epiphytic
invertebrates found on the Ascophyllum. Recent examinations
of such shipments arriving at bait shops in the Bay Area found
large numbers of live snails, bivalves, amphipods, isopods, harpacticoid
copepods, marine mites, insect larvae, polychaetes, oligochaetes,
nematodes and forams (Lau, 1995; ANC & JTC, pers. obs.). This
mechanism is likely responsible for the recent establishment of
one Atlantic periwinkle in the Bay and the occasional presence
of another. New bait worms now beginning to be marketed in California,
such as the Asian worm Namalycastis abiuma, may become
established in the Estuary or carry with them additional, yet
unknown, organisms.
HERRING-ROE-ON-KELP FISHERY:
Includes the kelp Macrocystis pyrifera collected
from the Channel Islands in southern California and placed in
San Francisco Bay as a substrate for herring spawning (Moore &
Reilly, 1989; Oda, 1989), and organisms found on Macrocystis.
Although it had been thought that M. pyrifera would not
reproduce and become established in the Bay, it has been found
attached, and therefore reproducing, in the Bay (L. Solarzano,
pers. comm., 1994; ANC & JTC, pers. obs.).
LIVE BAIT FISH:
Includes probable ongoing "bait bucket"
releases of the red shiner Notropis lutrensis into the
fresh waters of the Estuary and its tributaries (McGinnis, 1984;
Jennings & Saiki, 1990).
Table 10. Examples of Ongoing Inoculations - continued
PRIVATE PARTY RELEASES OF FISH OR SHELLFISH TO ESTABLISH FOOD OR SPORT RESOURCES:
In recent years these types of releases probably
account for the white bass established in the San Joaquin River
drainage and northern pike established in the Feather River drainage,
both likely to spread downstream to the Delta; Chinese mitten
crab established in San Francisco Bay and tributary streams and
likely to spread into the Delta and Central Valley rivers; blue
crab collected from the Delta, the Bay, and nearby coastal waters,
but not established; and possibly the alligator gar and Atlantic
eels collected but not established in the Delta. Nonindigenous
organisms currently imported alive to Bay Area markets, and thus
readily available for release into the Estuary along with any
parasites and epizoics they carry, include green-lipped mussels
from New Zealand, blue crabs from Chesapeake Bay and American
lobsters from Maine. The packing materials for these shellfish,
sometimes discarded into the Bay from dockside restaurants and
distribution and repacking centers, may contain yet additional
organisms. For example, the seaweed (Ascophyllum nodosum) used
to pack Atlantic lobsters was found, on arrival in the Bay Area,
to contain at least 29 other species of invertebrates and 7 other
species of seaweed from the Atlantic (Miller, 1969).
RELEASES FROM AQUARIA:
Can introduce and establish a variety of organisms,
which in the past have likely included plants (and the oligochaetes
and entoprocts living on them), snails, fish and turtles.
Recreational vessels entering the San Francisco Bay and Delta
from northern or eastern states have the potential to transport
with them, on their hulls or in incidental water aboard the vessel,
a broad variety of aquatic pest species, including aquatic weeds
(such as Hydrilla), snails (such as the New Zealand snail
Potamopyrgus antipodarum, introduced to the Middle Snake
River system of southern Idaho, and sometimes occurring in densities
of 100s of 1,000s of snails per square meter; Carlton et al.,
1996), and, especially, Eurasian zebra mussels (Dreissena polymorpha
and Dreissena bugensis), which between 1993 and 1995 have
been intercepted at the California border on recreational boats
coming from the Midwest and the Great Lakes.
Our certainty that there will be additional invasions of the Estuary stands in contrast to our limited ability to predict exactly which species (or even which trophic guilds) will invade and when they will invade. Carlton (1996b) discusses six scenarios, none mutually exclusive, that seek to explain why invasions may occur when they do; these include changes in the donor region, new donor regions, environmental changes in the recipient region, changes in the dispersal vector, the phenomenon of invasion windows, and stochastic inoculation events. All of these pertain to potential invasions of the San Francisco Estuary. A recent example of a combination of several of these processes apparently led to the successful invasion and subsequent persistence of the Asian clam Potamocorbula amurensis in the Bay (as discussed in Chapter 3).
Predicting specific guilds of invaders is often an elusive endeavor.
However, we note as an example the absence of certain truly euryhaline-oligohaline
taxa from the Estuary where native marine and freshwater counterparts
exist. Oglesby's (1965a) proposal that the Atlantic worm Nereis
succinea was successful in the Bay because it inserted itself
in this intermediate microhabitatóthat is, that it was
an "insertion invader"ósuggests that similar
opportunities may be available for other taxa. We note two such
examples (Table 4) among Bay isopods and amphipods. Also to be
expected are further warmer-water species as new colonists in
the Bay. The Bay has had a continuous history of such southern
species establishing on warm bay margins, including the barnacle
Balanus amphitrite, the tubeworm Ficopomatus and
the bryozoan Zoobotryon.
Consideration of the biological invasions of the San Francisco
Bay and Delta ecosystem has required examination of the records
and status of over 400 species. Documented plant and animal invasions
in the Estuary now number 212 species. An additional 123 species
are listed as cryptogenicónot clearly native or introducedóa
number that might represent less than half of the number of candidate
cryptogenic taxa. An additional 40 nonnative species were either
reported previously or have been recently discovered but are not
known to have become established in the Estuary, while another
36 nonnative species are established in adjacent aquatic ecosystems.
· Nonindigenous aquatic animals and plants have had a profound
impact on the ecology of this region. No shallow water habitat
now remains uninvaded by exotic species and, in some regions,
it is difficult to find any native species in abundance. In some
regions of the Bay, 100% of the common species are introduced,
creating "introduced communities." In locations ranging
from freshwater sites in the Delta, through Suisun and San Pablo
Bays and the shallower parts of the Central Bay to the South Bay,
introduced species account for the majority of the species diversity.
· 212 introduced species are now recognized in the Estuary.
Sixty-nine percent of these are invertebrates, 4 percent protists,
15 percent are fish and other vertebrates, and 12 percent are
vascular plants. Marine introductions are dominated by species
from the Western North Atlantic (41 percent), the Western North
Pacific (33 percent) and the Eastern North Atlantic (15 percent).
Continental introductions are dominated by species from North
America (54 percent, mostly fish) and from Eurasia (29 percent,
mostly plants).
· In addition to the 212 introductions reported, 123 species
are reported as cryptogenic (not clearly native or introduced),
and the total number of cryptogenic taxa in the Estuary might
well be twice that. Thus simply reporting the documented introductions
and assuming that all other species in a region are nativeóas
virtually all previous studies have doneóseverely underestimates
the impact of marine and aquatic invasions on a region's biota.
· Despite issues related to data quality that may frustrate
efforts to detect refined temporal patterns of invasions, the
first collection records of over 50 non-native species in the
Estuary since 1970 appear to reflect a significant new pulse of
invasions. In the period since the beginning of introductions
(here taken to be 1850), the Estuary has been invaded by an average
of one new species every 36 weeks. Since 1970, the rate has been
at least one new species every 24 weeks.
· The major bloom-creating, dominant phytoplankton species
are cryptogenic. Because of the poor state of taxonomic and biogeographic
knowledge, it remains possible that many of the Estuary's major
primary producers that provide the phytoplankton-derived energy
for zooplankton and filter feeders, are in fact introduced.
· Introduced species are abundant and dominant among the
zooplankton in the northern part of the Estuary, and throughout
the benthic and fouling communities of San Francisco Bay. On the
intertidal and sublittoral soft-bottom floors of the Bay these
include 10 species of introduced bivalves, most of which are abundant
to extremely abundant. Introduced filter-feeding polychaetes and
crustaceans may occur by the thousands per square meter. On subtidal
hard substrates, the mussel Mytilus galloprovincialis is
abundant, while sublittoral substrates (such as float fouling
communities) support large populations of introduced filter feeders,
including bryozoans, sponges and seasquirts. The holistic role
of the entire nonindigenous filter-feeding guildóincluding
clams, mussels, bryozoans, barnacles, seasquirts, spionid worms,
serpulid worms, sponges, hydroids, and sea anemonesóin
altering and controlling the trophic dynamics of the Bay-Delta
system remains unknown. The potential role of just one species,
the Atlantic ribbed marsh mussel Arcuatula demissa, as
a biogeochemical agent in the economy of Bay salt marshes is striking.
· Introduced benthic clams are capable of filtering the entire
volume of the South Bay and Suisun Bay once a day; indeed it now
appears that the primary mechanism controlling phytoplankton biomass
during summer and fall in South San Francisco Bay is "grazing"
(filter feeding) by the introduced clams Gemma, Venerupis,
and Musculista. This remarkable process thus has a significant
impact on the standing phytoplankton stock in the South Bay, and
since these stocks are now being utilized almost entirely by introduced
filter feeders, passing the energy through a non-native benthic
fraction of the biota may have fundamentally altered the energy
available for native biota
· Drought year control of phytoplankton by introduced clamsóresulting in the failure of the summer diatom bloom to appear in the northern reach of the Estuaryóis a remarkable phenomenon. The introduced soft-shell clams (Mya) alone were estimated to be capable at times of filtering all of the phytoplankton from the water column on the order of once per day. Phytoplankton blooms occurred only during higher flow years, when the populations of Mya and other introduced benthic filter feeders retreated downstream to saltier parts of the Estuary. However, phytoplankton populations in the northern reach of the Estuary may now be continuously and permanently and controlled by introduced clams. Arriving by ballast water and first collected in the Estuary in 1986, by 1988 the Asian clam Potamocorbula reached and has since sustained average densities exceeding 2,000/m2. Since the appearance of Potamocorbula, the summer diatom bloom has disappeared, presumably because of increased filter feeding by this new invasion. The Potamocorbula population in the northern reach of the Estuary can filter the entire water column over the channels more than once per day and over the shallows almost 13 times per day, a rate of filtration which exceeds the phytoplankton's specific growth rate and approaches or exceeds the bacterioplankton's specific growth rate.
Potamocorbula feeds at multiple levels in the food chain,
consuming bacterioplankton, phytoplankton, and zooplankton (copepods),
and so may substantially reduce copepod populations both by depletion
of the copepods' phytoplankton food source and by direct predation.
In turn, under such conditions, the copepod-eating native opossum
shrimp Neomysis may suffer a near-complete collapse in
the northern reach. It was during one such pattern that mysid-eating
juvenile striped bass suffered their lowest recorded abundance.
This example and the linkages between introduced and native species
may provide a direct and remarkable example of the potential impact
of an introduced species on the Estuary's food webs.
· As with the guild of filter feeders, the overall picture
of the impact of introduced epibenthic and shallow-infaunal grazers
and deposit feeders in the Estuary is incompletely known. The
Atlantic mudsnail Ilyanassa is likely playing a significantóif
not the most importantórole in altering the diversity,
abundance, size distribution, and recruitment of many species
on the intertidal mudflats of San Francisco Bay.
· The arrival and establishment of the green crab Carcinus
maenas in San Francisco Bay signals a new level of trophic
change and alteration. The green crab is a food and habitat generalist,
capable of eating an extraordinarily wide variety of animals and
plants, and capable of inhabiting marshes, rocky substrates, and
fouling communities. European, South African, and recent Californian
studies indicate a broad and striking potential for this crab
to significantly alter the distribution, density, and abundance
of prey species, and thus to profoundly alter community structure
in the Bay.
· Nearly 30 species of introduced marine,
brackish and freshwater fish are now important carnivores throughout
the Bay and Delta. Carp, mosquitofish, catfish, green sunfish,
bluegills, inland silverside, largemouth and smallmouth bass,
and striped bass are among the most significant predators, competitors,
and habitat disturbers throughout the brackish and freshwater
reaches of the Delta, with often concomitant impacts on native
fish communities. The introduced crayfish Procambarus and
Pacifastacus may play an important role, when dense, in
regulating their prey plant and animal populations.
· Native waterfowl in the Estuary consume some introduced
aquatic plants (such as brass buttons) and native shorebirds feed
extensively on introduced benthic invertebrates.
· Spartina alterniflora, which has converted 100s
of acres of mudflats in Willapa Bay, Washington, into cordgrass
islands, has become locally abundant in San Francisco Bay, and
is competing with the native cordgrass. Spartina alterniflora
has broad potential for ecosystem alteration. Its larger and more
rigid stems, greater stem density, and higher root densities may
decrease habitat for native wetland animals and infauna. Dense
stands of S. alterniflora may cause changes in sediment
dynamics, decreases in benthic algal production because of lower
light levels below the cordgrass canopy, and loss of shorebird
feeding habitat through colonization of mudflats.
· The Australian-New Zealand boring isopod Sphaeroma quoyanum
creates characteristic "Sphaeroma topography"
on many Bay shores, with many linear meters of fringing mud banks
riddled with its half-centimeter diameter holes. This isopod may
arguably play a major, if not the chief, role in erosion of intertidal
soft rock terraces along the shore of San Pablo Bay, due to their
boring activity that weakens the rock and facilitates its removal
by wave action. Sphaeroma has been burrowing into Bay shores
for over a century, and it thus may be that in certain regions
the land/water margin has retreated by a distance of at least
several meters due to this isopod's boring activities.
· Introduced freshwater and anadromous fish have been directly
implicated in the regional reduction and extinction, and the global
extinction, of four native California fish. The bluegill, green
sunfish, largemouth bass, striped bass, and black bass, through
predation and through competition for food and breeding sites,
have all been associated with the regional elimination of the
native Sacramento perch from the Delta. The introduced inland
silverside may be a significant predator on the larvae and eggs
of the native Delta smelt. Expansion of the introduced smallmouth
bass has been associated with the decline in the native hardhead.
Predation by largemouth bass, black bass and striped bass may
have been a major factor in the global extinction of the thicktail
chub in California.
· The situation of the California clapper rail may serve
as a model to assess how an endangered species may be affected
by biological invasions. The rail suffers predation by introduced
Norway rats and red fox; it may both feed on and be killed by
introduced mussels; and it may find refuge in introduced cordgrass,
although this same cordgrass may compete with native cordgrass,
perhaps preferred by the rail. Other potential model study systems
include introduced crayfish and their displacement of native crayfish;
introduced gobies and their relationship to the tidewater goby;
and the combined role that introduced green sunfish, bluegill,
largemouth bass, and American bullfrog may have played in the
dramatic decline of native red-legged and yellow-legged frogs.
· Though some of the fish intentionally introduced into the
Estuary by government agencies supported substantial commercial
food fisheries, these fisheries all declined after a time and
are now closed. The signal crayfish from Oregon, whose means of
introduction is unclear, supports the Estuary's only remaining
commercial food fishery based on an introduced species.
· The striped bass sport fishery has resulted in a substantial
transfer of funds from anglers to those who supply anglers' needs,
variously estimated, between 1962 and 1992, between $7 million
and $45 million per year. However, striped bass populations and
the striped bass sport fishery have declined dramatically in recent
years.
· Government introductions of organisms for sport fishing,
as forage fish and for biocontrol have frequently not produced
the intended benefits, and have sometimes had harmful "side
effects," such as reducing the populations of economically
important species.
· Few nonindigenous organisms that were introduced to the
Estuary by other than government intent have produced economic
benefits. The clams Mya and Venerupis, bothaccidentally
introduced with oysters, have supported commercial harvesting
in the Bay or elsewhere on the Pacific coast, and a small amount
of recreational harvesting in the Bay (though these clams may
have, to some extent, replaced edible native clams); the Asian
clam Corbicula is commercially harvested for food and bait
in California on a small scale; the Asian yellowfin goby is commercially
harvested for bait; muskrat are trapped for furs; and the South
African marsh plant brass buttons provides food for waterfowl.
There do not appear to be any other significant economic benefits
that derive from nongovernmental or accidental introductions to
the Estuary.
· A single introduced organism, the shipworm Teredo navalis,
caused $615 million (in 1992 dollars) of structural damage to
maritime facilities in 3 years.
· The economic impacts of hull fouling and other ship fouling
are clearly very large, but are not documented or quantified for
the Estuary. Most of the fouling incurred in the Estuary is due
to nonindigenous species. Indirect impacts due to the use of toxic
anti-fouling coatings may also be substantial.
· Waterway fouling by introduced water hyacinth has become
a problem in the Delta over the last fifteen years, with other
introduced plants beginning to add to the problem in recent years.
Hyacinth fouling has had significant economic impacts, including
interference with navigation.
· Perhaps the greatest economic impacts may derive from the
destabilizing of the Estuary's biota due to the introduction and
establishment of an average of one new species every 24 weeks.
This phenomenal rate of species additions has contributed to the
failure of water users and regulatory agencies to manage the Estuary
so as to sustain healthy populations of anadromous and native
fish, resulting in increasing limitations and threats of limitations
on water diversions, wastewater discharges, channel dredging,
levee maintenance, construction and other economic activities
in and near the Estuary, with implications for the whole of California's
economy.
Much remains unknown in terms of the phenomena, patterns, and
processes of invasions in the Bay and Delta, and thus large gaps
remain in the knowledge needed to establish effective management
plans. The following are examples of important research needs
and directions:
As discussed in Chapter 3, only a few of the hundreds of invaders
in the Estuary have been the subject of quantitative experimental
studies elucidating their roles in the Estuary's ecosystem and
their impacts on native biota. Such studies should receive the
highest priority.
Urgently required is a San Francisco Bay Shipping Study which
both updates the 1991 data base available and expands that data
base to all Bay and Delta ports. A biological and ecological study
of the nature of ballast water biota arriving in the Bay/Delta
system is urgently required. Equally pressing is a study of the
fouling organisms entering the Estuary on ships' hulls and in
ships' seachests, in order to assess whether this mechanism is
now becoming of increasing importance and in order to more adequately
define the unique role of ballast water. A Regional Shipping Study
would provide critical data for management plans.
Studies are required on the mechanisms and the temporal and spatial
scales of the distribution of introduced species by human vectors
after they have become established. Such studies will be of particular
value in light of any future introductions of nuisance aquatic
pests.
Our work has identified a major, unregulated vector for exotic
species invasions in the Bay: the constant release of invertebrate-laden
seaweeds from New England in association with bait worm (and lobster)
importation. In addition a new trade in exotic bait has commenced,
centered around the importation of living Vietnamese nereid worms,
and both the worms and their substrate deserve detailed study.
These studies are urgently needed to address the attendant precautionary
management issues at hand.
The application of modern molecular genetic techniques has already
revealed the cryptic presence of previously unrecognized invaders
in the Bay: the Atlantic clam Macoma petalum, the Mediterranean
mussel Mytilus galloprovincialis, and the Japanese jellyfish
Aurelia "aurita." Molecular genetic studies of
the Bay's new green crab (Carcinus) population may be of
critical value in resolving the crab's geographic origins and
thus the mechanism that brought it to California. Molecular genetic
studies of worms of the genus Glycera and Nereis
in the Bay may clarify if New England populations have or are
becoming established in the region as a result of ongoing inoculations
via the bait worm industry. Molecular analysis of other invasions
will doubtless reveal, as with Macoma and Mytilus,
a number of heretofore unrecognized species.
Fishery, bait, and other utilization studies should be conducted
on developing or enlarging the scope of fisheries for introduced
bivalves (such as Mya, Venerupis, and Corbicula),
edible aquatic plants, smaller edible fish (such as Acanthogobius),
and crabs (Carcinus and Eriocheir).
Studies are needed on the potential distribution, abundance and
impacts of zebra mussels (Dreissena polymorpha and/or D.
bugensis) in California, to support efforts to control their
introduction and to design facilities (such as water intakes and
fish screens) that will continue to function adequately should
the mussels become established.
The economic impacts of wood-boring organisms (shipworms and gribbles)
and of fouling organisms (on commercial vessels, on recreational
craft, in ports and marinas, and in water conduits) are clearly
very large in the San Francisco Estuary, but remain largely undocumented
and entirely unquantified. A modern economic study of this phenomenon,
including the economic costs and ecological impacts of control
measures now in place or forecast, is critically needed.
Largely qualitative data suggest that the economic, ecological,
and geological impacts of the guild of burrowing organisms that
have been historically and newly introduced have been or are forecast
to potentially be extensive in the Estuary. Experimental, quantitative
studies on the impacts of burrowing and bioeroding crustaceans
and muskrats in the Estuary are clearly now needed to assess the
extent of changes that have occurred or are now occurring, and
to form the basis for predicting future alterations in the absence
of control measures.
While primary attention must be paid to preventing future invasions,
studies should begin on examining the broad suite of potential
post-invasion control mechanisms, including biocontrol, physical
containment, eradication, and related strategies. A Regional Control
Mechanisms Workshop for past and anticipated invasions could set
the foundation for future research directions.